Loading metrics
Open Access
Peer-reviewed
Research Article

Associations of maternal quitting, reducing, and continuing smoking during pregnancy with longitudinal fetal growth: Findings from Mendelian randomization and parental negative control studies
Roles Conceptualization, Formal analysis, Methodology, Writing – original draft
Affiliations Clinical Epidemiology and Biostatistics, School of Medical Sciences, Örebro University, Örebro, Sweden, MRC Integrative Epidemiology Unit, University of Bristol, Bristol, United Kingdom, Population Health Science, Bristol Medical School, University of Bristol, Bristol, United Kingdom
Roles Conceptualization, Data curation, Writing – review & editing
Affiliations Generation R Study Group, Erasmus University Medical Center Rotterdam, Rotterdam, the Netherlands, Department of Pediatrics, Sophia Children’s Hospital, Erasmus University Medical Center Rotterdam, Rotterdam, the Netherlands
Roles Data curation, Funding acquisition, Writing – review & editing
Affiliations MRC Integrative Epidemiology Unit, University of Bristol, Bristol, United Kingdom, Population Health Science, Bristol Medical School, University of Bristol, Bristol, United Kingdom, Bradford Institute for Health Research, Bradford Royal Infirmary, Bradford, United Kingdom
Affiliation Bradford Institute for Health Research, Bradford Royal Infirmary, Bradford, United Kingdom
Roles Conceptualization, Data curation, Funding acquisition, Writing – review & editing
Roles Data curation, Writing – review & editing
Affiliations Generation R Study Group, Erasmus University Medical Center Rotterdam, Rotterdam, the Netherlands, Department of Pediatrics, Sophia Children’s Hospital, Erasmus University Medical Center Rotterdam, Rotterdam, the Netherlands, Department of Epidemiology, Erasmus University Medical Center Rotterdam, Rotterdam, the Netherlands
Roles Conceptualization, Methodology, Writing – review & editing
Affiliations MRC Integrative Epidemiology Unit, University of Bristol, Bristol, United Kingdom, Population Health Science, Bristol Medical School, University of Bristol, Bristol, United Kingdom
Roles Conceptualization, Funding acquisition, Methodology, Writing – original draft
* E-mail: [email protected]
Affiliations MRC Integrative Epidemiology Unit, University of Bristol, Bristol, United Kingdom, Population Health Science, Bristol Medical School, University of Bristol, Bristol, United Kingdom, National Institute for Health Research Bristol Biomedical Research Centre, Bristol, United Kingdom
- Judith S. Brand,
- Romy Gaillard,
- Jane West,
- Rosemary R. C. McEachan,
- John Wright,
- Ellis Voerman,
- Janine F. Felix,
- Kate Tilling,
- Deborah A. Lawlor

- Published: November 13, 2019
- https://doi.org/10.1371/journal.pmed.1002972
- Peer Review
- Reader Comments
Maternal smoking during pregnancy is an established risk factor for low infant birth weight, but evidence on critical exposure windows and timing of fetal growth restriction is limited. Here we investigate the associations of maternal quitting, reducing, and continuing smoking during pregnancy with longitudinal fetal growth by triangulating evidence from 3 analytical approaches to strengthen causal inference.
Methods and findings
We analysed data from 8,621 European liveborn singletons in 2 population-based pregnancy cohorts (the Generation R Study, the Netherlands 2002–2006 [ n = 4,682]) and the Born in Bradford study, United Kingdom 2007–2010 [ n = 3,939]) with fetal ultrasound and birth anthropometric measures, parental smoking during pregnancy, and maternal genetic data. Associations with trajectories of estimated fetal weight (EFW) and individual fetal parameters (head circumference, femur length [FL], and abdominal circumference [AC]) from 12–16 to 40 weeks’ gestation were analysed using multilevel fractional polynomial models. We compared results from (1) confounder-adjusted multivariable analyses, (2) a Mendelian randomization (MR) analysis using maternal rs1051730 genotype as an instrument for smoking quantity and ease of quitting, and (3) a negative control analysis comparing maternal and mother’s partner’s smoking associations. In multivariable analyses, women who continued smoking during pregnancy had a smaller fetal size than non-smokers from early gestation (16–20 weeks) through to birth ( p- value for each parameter < 0.001). Fetal size reductions in continuing smokers followed a dose-dependent pattern (compared to non-smokers, difference in mean EFW [95% CI] at 40 weeks’ gestation was −144 g [−182 to −106], −215 g [−248 to −182], and −290 g [−334 to −247] for light, moderate, and heavy smoking, respectively). Overall, fetal size reductions were most pronounced for FL. The fetal growth trajectory in women who quit smoking in early pregnancy was similar to that of non-smokers, except for a shorter FL and greater AC around 36–40 weeks’ gestation. In MR analyses, each genetically determined 1-cigarette-per-day increase was associated with a smaller EFW from 20 weeks’ gestation to birth in smokers ( p = 0.01, difference in mean EFW at 40 weeks = −45 g [95% CI −81 to −10]) and a greater EFW from 32 weeks’ gestation onwards in non-smokers ( p = 0.03, difference in mean EFW at 40 weeks = 26 g [95% CI 5 to 47]). There was no evidence that partner smoking was associated with fetal growth. Study limitations include measurement error due to maternal self-report of smoking and the modest sample size for MR analyses resulting in unconfounded estimates being less precise. The apparent positive association of the genetic instrument with fetal growth in non-smokers suggests that genetic pleiotropy may have masked a stronger association in smokers.
Conclusions
A consistent linear dose-dependent association of maternal smoking with fetal growth was observed from the early second trimester onwards, while no major growth deficit was found in women who quit smoking early in pregnancy except for a shorter FL during late gestation. These findings reinforce the importance of smoking cessation advice in preconception and antenatal care and show that smoking reduction can lower the risk of impaired fetal growth in women who struggle to quit.
Author summary
Why was this study done.
- Maternal smoking during pregnancy is an established risk factor for low infant birth weight. Understanding when and which parameters of fetal growth are affected by different smoking behaviours is important for strengthening and focusing clinical and public health guidelines.
- The importance of smoking cessation in early pregnancy and the extent to which fetal growth restriction can be prevented or minimised by lowering cigarette consumption in women who find quitting difficult is also uncertain.
What did the researchers do and find?
- We analysed data from 8,621 white European liveborn singletons from 2 population-based pregnancy cohorts to assess the associations of maternal quitting, reducing, and continuing smoking during pregnancy with the longitudinal growth of different fetal parameters (weight, head circumference, femur length, and abdominal circumference). We compared results across 3 different analytical approaches (conventional multivariable, Mendelian randomization, and parental negative control analyses) to strengthen confidence in our findings.
- We found that pre-pregnancy smokers who continued smoking during pregnancy had a reduced fetal size from early gestation (12–16 weeks) onwards. Associations of maternal smoking with each fetal parameter followed a dose-dependent pattern, with fetal size reductions increasing in magnitude with the number of cigarettes smoked.
- While all fetal parameters were affected in women who continued smoking during pregnancy, size reductions were most pronounced for femur length. In pre-pregnancy smokers who gave up smoking early in pregnancy, no overall growth deficit was observed, except for a smaller femur length towards the end of pregnancy.
- The association of maternal smoking with reduced fetal growth was consistent across all 3 methods, thus providing stronger support that the association is causal, in comparison to current evidence, which relies solely on multivariable regression.
What do these findings mean?
- Our findings reinforce existing advice promoting and supporting smoking cessation in preconception and antenatal care services; they provide strong support for these recommendations.
- The consistent results across methods for a linear dose-dependent association of maternal smoking with reduced fetal growth from early gestation in women who continue smoking during pregnancy provide evidence to support reducing smoking amounts in those who struggle to quit.
Citation: Brand JS, Gaillard R, West J, McEachan RRC, Wright J, Voerman E, et al. (2019) Associations of maternal quitting, reducing, and continuing smoking during pregnancy with longitudinal fetal growth: Findings from Mendelian randomization and parental negative control studies. PLoS Med 16(11): e1002972. https://doi.org/10.1371/journal.pmed.1002972
Academic Editor: Cosetta Minelli, Imperial College London, UNITED KINGDOM
Received: June 2, 2019; Accepted: October 21, 2019; Published: November 13, 2019
Copyright: © 2019 Brand et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Data Availability: The data underlying the results presented here are from 2 different studies with different data publishing policies. The individual-level data are not available through a public repository due to ethical and legal restrictions. Both studies have an open collaboration policy and the datasets generated and analysed for this study are available upon request. Applications to use the BiB dataset can be made directly to the BiB executive group ( https://borninbradford.nhs.uk/research/how-to-access-data/ ) and applications to use the GenR dataset can be made to the Generation R Study Management Team ( [email protected] ), subject to local rules and regulations.
Funding: The Born in Bradford study (BiB) receives core funding from the Wellcome Trust (WT101597MA) a joint grant from the UK Medical Research Council (MRC) and UK Economic and Social Science Research Council (ESRC) (MR/N024397/1) and the National Institute for Health Research (NIHR) under its Collaboration for Applied Health Research and Care (CLAHRC) for Yorkshire and Humber and the NIHR Clinical Research Network (CRN). The general design of the Generation R Study (GenR) is made possible by financial support from the Erasmus University Medical Center, Rotterdam, Erasmus University, Rotterdam, the Netherlands Organisation for Health Research and Development (ZonMw), the Netherlands Organisation for Scientific Research (NWO), the Dutch Ministry of Health, Welfare and Sport and the Dutch Ministry of Youth and Families. This project also received funding from the European Union's Horizon 2020 research and innovation programme under grant agreements No 633595 (DynaHEALTH) and No 733206 (LIFECYCLE). The research presented in this paper is supported by the British Heart Foundation (CS/16/4/32482 and AA/18/7/34219), US National Institute of Health (R01 DK10324), the European Research Council under the European Union’s Seventh Framework Programme (FP7/2007-2013) / ERC grant agreement No 669545 (the latter also provides J.S.B.’s salary), the European Union’s Horizon 2020 research and innovation programme under grant agreement No 733206 (LIFECYCLE) and the NIHR Biomedical Centre at the University Hospitals Bristol, the NHS Foundation Trust and the University of Bristol. JSB., KT. and DAL work in a unit that receives UK Medical Research Council (MRC) funding (MC_UU_00011/3 and MC_UU_00011/6) and DAL is an NIHR senior investigator (NF-SI-0611-10196). RG received funding from the Dutch Heart Foundation (2017T013), the Dutch Diabetes Foundation (2017.81.002) and the Netherlands Organisation for Health Research and Development (ZonMw) (543003109). The funders had no role in the design of the study, the collection, analysis, or interpretation of the data; the writing of the manuscript, or the decision to submit the manuscript for publication.
Competing interests: I have read the journal's policy and the authors of this manuscript have the following competing interests: DAL has received support from several national and international government and charitable funders, and from Medtronic Ltd and Roche Diagnostics for research unrelated to that presented here. The other authors report no conflicts.
Abbreviations: AC, abdominal circumference; BiB, Born in Bradford study; CRL, crown–rump length; EFW, estimated fetal weight; FL, femur length; GenR, Generation R Study; HC, head circumference; MR, Mendelian randomization; OR, odds ratio
Introduction
Maternal smoking during pregnancy is one of the most important modifiable determinants of low infant birth weight and other adverse perinatal outcomes [ 1 ]. Whilst the prevalence of pregnancy smoking has declined, it remains high in the US and Western Europe, where approximately 15%–20% of all pregnant women smoke [ 2 ]. According to the US Surgeon General’s reports [ 3 , 4 ], there is substantial evidence supporting a direct link between pregnancy smoking and low infant birth weight, with this evidence being consistent across a multitude of studies using conventional multivariable analyses of observational data. However, these results from conventional observational approaches may be explained by residual confounding given that women who smoke during pregnancy are more likely to be socioeconomically disadvantaged and to engage in other risky health behaviours that may lead to low birth weight. More recent findings from quasi- experimental studies comparing populations with different tobacco control policies [ 5 – 7 ], and from observational studies using Mendelian randomization (MR) [ 8 ], parental negative control [ 9 ], and discordant sibling [ 10 ] designs, support an intrauterine effect of maternal smoking during pregnancy on infant birth weight.
Still, evidence on the critical smoking exposure window and timing of smoking-related fetal growth restriction is limited. Previous studies have mainly focused on size at birth only, with few studies exploring when during pregnancy smoking starts to affect growth, or whether there are differences in its effect on different fetal growth parameters. Pregnancy smoking has been associated with a reduction in first trimester crown–rump length (CRL) [ 11 ], but studies using repeat ultrasound measures have failed to identify an association with early second trimester fetal size, and the magnitude of reported associations with different fetal parameters later in pregnancy varies considerably [ 12 ]. This inconsistency largely reflects between-study heterogeneity in longitudinal fetal growth assessment and adjustment for confounding factors, which hamper the ability to determine causal intrauterine effects from early fetal life [ 12 ]. There is also uncertainty about the impact of smoking cessation in early pregnancy and the extent to which fetal growth restriction can be prevented or minimised by lowering cigarette consumption. Obtaining stronger causal evidence of when in pregnancy smoking influences fetal growth, how quitting affects this, and whether there is a dose response in those who continue smoking will provide stronger evidence to inform updated guidelines and to help women make more informed decisions. Furthermore, understanding whether smoking reduces birth weight by universally reducing growth across all growth parameters or has varying effects could provide insights into the mechanisms through which smoking affects fetal growth.
The aim of this study was to determine the associations of maternal quitting, reducing, and continuing smoking during pregnancy with longitudinal trajectories of different fetal growth parameters (head circumference [HC], femur length [FL], abdominal circumference [AC], and estimated fetal weight [EFW]) in a joint analysis of data from 2 population-based pregnancy cohorts. To improve causal inference, we triangulated findings from 3 approaches with differing sources of bias (multivariable regression, MR, and parental negative control) [ 13 ]. This study provides novel insights into the impact of quitting or reducing smoking during pregnancy on fetal growth that can be used to tailor advice and support to individual women.
We identified cohorts from the MR-PREG consortia, a collaboration of cohorts used to explore causes and consequences of different pregnancy complications and outcomes. To contribute to this study, cohorts had to have repeat fetal ultrasound scan measurements. We used parental and offspring data from the Generation R Study (GenR) [ 14 ] and the Born in Bradford study (BiB) [ 15 ], 2 population-based pregnancy cohorts including participants from multi-ethnic urban populations (see S1 Text for a detailed description of both cohorts). GenR is based in Rotterdam (the Netherlands) and consists of 9,778 women (response rate at baseline = 61%) who had an expected delivery date between April 2002 and January 2006. Most participants ( n = 8,880, 91%) were recruited during pregnancy. BiB enrolled 12,450 women residing in Bradford (response rate > 80%), a city in the north of England. Women participating in BiB had an expected delivery date between March 2007 and December 2010 and were mainly recruited at their oral glucose tolerance test (OGTT) appointment at 26–28 weeks’ gestation (all pregnant women booked to deliver in Bradford are offered an OGTT). For the present analysis, we included women who gave birth to singletons without known fetal/birth congenital anomalies and with fetal growth and maternal smoking data (see S1 Fig ). We further restricted analyses to participants of white European origin, as smoking behaviours and fetal growth trajectories differ considerably by ethnicity, and for other ethnic groups, numbers were too small for reliable estimates. This resulted in a study population of 8,621 liveborn singletons (GenR, n = 4,682; BiB, n = 3,939). Of these, 6,527 had maternal genotype data (GenR, n = 3,604; BiB, n = 2,923) and were included in the MR analyses, and 5,537 had partner smoking data (GenR, n = 4,206; BiB, n = 1,331) for inclusion in the negative control study.
All study participants gave written informed consent, and the study was approved by the local medical ethical committees (GenR: MEC 198.782/2001/31; BiB: ref 06/Q1202/48).
Parental smoking
Full details of how parental smoking was assessed are provided in S1 Text . Based on the available data in both cohorts, maternal smoking during pregnancy was categorised as follows: (1) non-smokers (i.e., women who had never smoked or stopped smoking several months [>6 months in GenR and >3 months in BiB] prior to pregnancy); (2) pre-pregnancy smokers who quit in early pregnancy (i.e., women who reported smoking in the months before becoming pregnant but not after the first trimester); and (3) pre-pregnancy smokers who continued smoking through pregnancy (i.e., women who reported smoking in the months before becoming pregnant, in the first trimester, and during mid and/or late pregnancy).
We also extracted information on the number of cigarettes smoked by women who continued to smoke through pregnancy. This was done by taking the mean of the smoking quantity reported during the first trimester and during mid and/or late pregnancy. Since each cohort used different cutoffs to categorise self-reported smoking quantity (intensity), these data were grouped into ‘light’ (1–4 [GenR] or 1–5 [BiB] cigarettes per day), ‘moderate’ (5–9 [GenR] or 6–10 [BiB] cigarettes per day), and ‘heavy’ smoking (≥10 [GenR] or >10 [BiB] cigarettes per day).
To strengthen causal inference, we used information on partner smoking during pregnancy as a negative control [ 13 , 16 ]. Under the assumption that confounding would be similar for maternal and partner smoking, stronger estimates for the association of fetal growth parameters with maternal than partner smoking can be interpreted as support for a causal intrauterine effect of maternal smoking, while similar effect estimates would be suggestive of unmeasured shared familial confounding [ 13 ]. We used the same categories of partner smoking quantity (light, moderate, and heavy smoking) to those used for maternal smoking.
Instrument selection and genotyping for MR
We selected the rs1051730 single nuclear polymorphism (SNP) in the α-nicotinic acetylcholine receptor (CHRNA3/5) gene cluster as an instrumental variable for our MR analysis because this variant has been shown to robustly (in genome-wide discovery and replication studies [ 17 , 18 ]) relate to smoking intensity and ease of quitting, and also to relate to these smoking traits in pregnancy [ 19 ]. Full details of genotyping and quality control are provided in S1 Text and S1 Table .
Fetal growth assessment
Anthropometric measurements at birth (HC and weight in both cohorts and AC in BiB only) were obtained from obstetric medical records and combined with fetal anthropometric data to estimate trajectories of fetal growth. In both cohorts, the median number of repeat weight estimates per singleton was 3.
Other variables
The following variables were considered as potential confounders in multivariable analyses, including for partner smoking associations: age, parity (at the time of index pregnancy; mothers only), height, body mass index, education, and alcohol intake. In both cohorts, these variables were mainly derived from the baseline questionnaire administered at enrolment at a median gestational age of 13 weeks (GenR) or 26 weeks (BiB). Full details of their assessment and inclusion in the analysis models are provided in S1 Text .
Statistical analysis
A draft analysis plan was written by JSB, KT, and DAL in December 2017, and the final analysis plan was agreed upon by all investigators in February 2018 after discussions with the GenR team (see S2 Text ). Analyses commenced in June 2018, and 2 changes were made after analyses had begun. Preliminary results showed that maternal rs1051730 genotype was associated with maternal age, and we decided to explore the impact of this by repeating all MR analyses with adjustment for maternal age. In January 2019, in response to suggestions from one of the co-authors, it was agreed that we would also estimate and report proportional differences in mean fetal size as we might expect absolute differences to increase as the fetus grows. No further changes to the analysis plan were made.
Fetal growth trajectories were derived using multilevel fractional polynomial models with 2 levels (i.e., measurements within occasions [level 1] within individuals [level 2]) as described in detail elsewhere [ 21 ]. A variable for cohort (GenR versus BiB) and one for its interaction with gestational age were entered in all models to account for between-study differences in fetal growth. In pooling the data in a single analysis model, we assume that both cohorts are from the same underlying population for which inferences can be made. To test this assumption, we compared estimates obtained from this single analysis model with those observed in GenR and BiB separately and further tested for between-study heterogeneity by adding a 3-way interaction term (between study, smoking exposure, and gestational age) to each analysis model. More details on model specification can be found in S1 Text , including supportive data for the growth trajectories fitted ( S2 and S3 Tables; S2 Fig ). We also compared the fetal growth results with associations observed for birth weight using linear regression. Because fetal growth trajectories, by definition, are conditioned on gestational age, we checked the extent to which birth weight differences changed with adjustment for gestational age.
Multivariable and parental negative control analyses
Associations of maternal and partner smoking exposures were analysed by adding these variables as main effects and as interactions with gestational age to the multilevel models. From these models, global p- values for these coefficients were derived to assess overall differences in fetal growth (i.e., to test the null hypothesis that the growth trajectories across pregnancy for different parameters do not differ, for example comparing smokers to non-smokers). This global approach is recommended to avoid multiple testing. Differences in mean fetal size associated with each smoking exposure were estimated at 4-week intervals from 12–16 weeks onwards in absolute original units (i.e., millimetres and grams) and proportionally as the ratio of the observed differences to the mean at each time point. To provide an estimate of the timing of fetal growth restriction, we also report the earliest gestational age (based on 4-week intervals) at which the 95% CI for a difference in mean fetal size did not include 0. It should, however, be noted that this estimate is conservative as it is guided by statistical significance only, and the actual process of growth restriction will have started prior to this point in time.
Fetal size differences by parental (maternal/partner) smoking status were estimated using maternal/partner non-smokers as the reference group. Associations with parental smoking are presented with adjustment for cohort only (model 1) and with adjustment for cohort, infant sex, parity (for maternal smoking only), and respective parental age, height, BMI, education level, and alcohol use during pregnancy (model 2). In the parental negative control comparison, associations were additionally mutually adjusted for the smoking behaviour of the other parent (model 3).
Missing covariate data were imputed using multiple imputation stratified by cohort (see details in S1 Text ), and we also present results based on complete case data.
MR analysis
Fetal size differences by maternal rs1051730 genotype were modelled per risk allele (T) increase (i.e., assuming an additive genetic effect). As rs1051730 genotype has been associated with smoking quantity in individuals who smoke (with each T allele increase corresponding to approximately 1 additional cigarette per day [ 17 , 24 ]), we would anticipate an association in women continuing to smoke during pregnancy, and would expect this association to be weaker in those who quit smoking and null in non-smokers. An association with fetal growth in non-smokers would be indicative of a horizontal pleiotropic effect of the variant (i.e., not acting through smoking intensity or inability to quit smoking) and would suggest that our results in smokers may be biased [ 25 ]. In our main analyses we compared the associations of the genetic variant with fetal growth in these 3 groups (pre-pregnancy smokers who continued smoking through pregnancy, pre-pregnancy smokers who quit in early pregnancy, and non-smokers). Stratifying pre-pregnancy smokers into those quitting and continuing smoking could, however, introduce collider bias [ 26 ], as the rs1051730 T allele has previously been associated with the ability to quit smoking [ 19 ]. We therefore repeated the MR analysis in just 2 groups: pre-pregnancy smokers and non-smokers. We examined associations of maternal rs1051730 genotype with potential confounders of the smoking–fetal growth association to test the independence assumption. To address the possibility of an association of maternal rs1051730 being mediated through inheritance rather than a causal intrauterine effect, we performed a sensitivity analysis with adjustment for fetal rs1051730 genotype. Fetal genotype data were available for 4,457 singletons.
All analyses were undertaken using MLwiN version 2.4 run in Stata/MP version 15.
Participant characteristics
Distributions of characteristics of participants included in the MR and negative control analyses were very similar to those included in the main analysis of maternal smoking effects ( S4 Table ). In GenR, 1,221 women (26%) reported smoking prior to pregnancy, including 423 who quit early in pregnancy (9%) and 798 (17%) who continued smoking through pregnancy. Corresponding percentages of pre-pregnancy smoking (41%) and continued smoking (30%) were higher in BiB, whereas the proportion of women quitting in early pregnancy was similar to that observed in GenR (11%) ( Table 1 ). Overall, 44% and 35% of partners smoked in GenR and BiB, respectively. In both cohorts, mothers who continued smoking during pregnancy were younger, shorter, less educated, and more often multiparous, and gave birth to infants of lower gestational age and birth weight ( S5 Table ). In BiB, mothers who continued smoking also had a lower BMI ( S5 Table ). Similar associations with these characteristics were observed for partner smoking status during pregnancy ( S6 Table ).
- PPT PowerPoint slide
- PNG larger image
- TIFF original image
https://doi.org/10.1371/journal.pmed.1002972.t001
Associations of maternal smoking with fetal growth
Differences in mean fetal size across gestation by maternal smoking during pregnancy are presented in Fig 1 , with estimates from multivariable adjusted analyses shown in S7 Table . Overall, trajectories of fetal growth varied according to maternal smoking status ( p < 0.001 for each fetal parameter). From the early second trimester through to term, fetuses of women who continued to smoke weighed less than those of non-smokers. More specifically, at 20 weeks, the predicted difference (95% CI) in mean EFW was −2.6 g (−5.1 to −0.1), and this absolute difference increased to −207 g (−231 to −182) at 40 weeks’ gestation. A similar pattern was observed for HC, FL, and AC, with absolute differences in each fetal parameter widening with gestational age. Fetal FL reductions were observed from 16 weeks, and fetal HC and AC reductions were observed from 20 and 24 weeks, respectively. The EFW trajectory of women who quit smoking early in pregnancy was similar to that of non-smokers ( Fig 1 ; S7 Table ). However, quitters had a shorter FL and greater AC than non-smokers towards the end of pregnancy, though these differences were smaller in magnitude than those observed for continuing smokers.
Predicted differences in mean head circumference (mm), femur length (mm), abdominal circumference (mm), and estimated fetal weight (g) across gestation comparing pre-pregnancy smokers who quit in early pregnancy and those who continued smoking during pregnancy with non-smokers (reference category). Predicted mean differences (with 95% confidence intervals) in the pooled Generation R Study and Born in Bradford study cohort by analysis model: model 1 adjusting for cohort only (in white) and model 2 adjusting for cohort, infant sex, and maternal age, parity, height, body mass index, education level, and alcohol use during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.g001
Fetal size differences by smoking quantity in mothers who continued smoking during pregnancy are shown in Fig 2 and S8 Table . Dose–response associations with EFW and individual fetal parameters (HC, FL, and AC) were observed from early gestation through to birth ( p < 0.001 for all fetal parameters). Compared to non-smokers, the difference (95% CI) in mean EFW for light, moderate, and heavy smoking in women who continued smoking was −144 g (−182 to −106), −215 g (−248 to −182), and −290 g (−334 to −247) at 40 weeks, respectively. As was the case for smoking status, associations of smoking quantity with fetal parameters appeared to be most pronounced for FL. Results for predicted differences as a proportion of the mean revealed a similar pattern as for mean differences in absolute original units: proportional differences in fetal parameters observed with maternal smoking status and smoking quantity followed an increasing pattern with increasing gestational age ( S9 and S10 Tables).
Predicted differences in mean head circumference (mm), femur length (mm), abdominal circumference (mm), and estimated fetal weight (g) across gestation comparing pre-pregnancy smokers who continued smoking through pregnancy by smoking intensity with non-smokers (reference category). Predicted mean differences (with 95% confidence intervals) in the pooled Generation R Study and Born in Bradford study cohort by analysis model: model 1 adjusting for cohort only (in white) and model 2 adjusting for cohort, infant sex, and maternal age, parity, height, body mass index, education level, and alcohol use during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.g002
Association of maternal rs1051730 genotype with smoking and fetal growth—MR analysis
Maternal rs1051730 genotype was associated with the ability to quit smoking among pre-pregnancy smokers (per T allele odds ratio [OR] for continuing versus quitting smoking during pregnancy = 1.10 [95% CI 0.96 to 1.25]). This association was mainly driven by GenR (OR = 1.17 [95% CI 0.96 to 1.43]) as rs1051730 was not clearly associated with the ability to quit smoking in BiB (OR = 1.03 [95% CI 0.86 to 1.24]) ( S11 Table ). In both cohorts, maternal rs1051730 genotype was associated with the number of cigarettes smoked in women who continued smoking during pregnancy (per T allele OR for heavy smoking versus light/moderate smoking = 1.24 [95% CI 1.04 to 1.47]), but not in those who quit smoking in early pregnancy (OR = 1.02 [95% CI 0.79 to 1.33]). The SNP was not associated with being a smoker (per T allele OR for being a pre-pregnancy smoker = 1.00 [95% CI 0.92 to 1.08]), confirming genome-wide association results that rs1051730 is not associated with smoking initiation [ 18 ].
Associations of maternal rs1051730 genotype with fetal growth differed by smoking status ( Fig 3 ; S12 Table ). Amongst women who continued to smoke across pregnancy, EFW growth differed by rs1051730 genotype ( p = 0.01). At 20 weeks’ gestation, each additional T allele was associated with a lower EFW (−3.7 g [95% CI −7.0 to −0.4]), and this absolute reduction in fetal weight increased in magnitude with gestational age (at 40 weeks it was −45 g [95% CI −81 to −10]). A similar pattern of fetal growth restriction was observed for individual fetal parameters (HC, FL, and AC) in these women, though differences in mean fetal AC were less precise and close to null in late gestation. In women classified as non-smokers, each additional T allele at rs1051730 was associated with a higher EFW between 32 weeks and 40 weeks of gestation ( p for overall difference in growth = 0.03), with similar patterns of associations for HC, AC, and FL. No association between maternal rs1051730 genotype and fetal growth was observed in women who quit smoking in early pregnancy, but these results need to be interpreted with caution because of the small number of individuals in this group. Proportional differences in mean fetal size with each additional T allele at rs1051730 followed a similar pattern as for the absolute differences observed ( S13 Table ). Effect estimates were not materially different after combining pre-pregnancy smokers who quit and those who continued smoking ( S14 Table ; S3 Fig ). With the exception of maternal age, the SNP was not associated with confounders ( S15 Table ). Maternal age in continuing smokers increased with each additional T allele; there was no association with age in non-smokers and those who quit smoking in early pregnancy. Results were not altered with adjustment for maternal age ( S4 and S5 Figs) or fetal rs1051730 genotype (Figs 3 and S3 – S5 ).
Predicted differences in mean head circumference (mm), femur length (mm), abdominal circumference (mm), and estimated fetal weight (g) per maternal rs1051730 T allele increase in non-smokers, pre-pregnancy smokers who quit smoking in early pregnancy, and pre-pregnancy smokers who continued smoking during pregnancy. Predicted mean differences (with 95% confidence intervals) by analysis model: model 1 adjusting for cohort (in white) and model 2 adjusting for cohort and fetal rs1051730 genotype (in black).
https://doi.org/10.1371/journal.pmed.1002972.g003
Association of maternal versus mother’s partner smoking with fetal growth—Parental negative control analysis
In unadjusted analyses, partner smoking was associated with slower growth of all parameters ( S6 Fig ), but these associations were considerably weaker than those for maternal smoking and were only apparent later in pregnancy (from 32 weeks onwards). The partner smoking associations were attenuated to the null after multivariable adjustment for confounders and mutual adjustment for maternal smoking (Figs 4 and S6 ; S16 and S17 Tables). Results for partner smoking quantity showed similar patterns ( S7 Fig ).
Predicted differences in mean head circumference (mm), femur length (mm), abdominal circumference (mm), and estimated fetal weight (g) across gestation associated with maternal smoking (i.e., comparing maternal continued smoking through pregnancy with no maternal smoking during pregnancy [reference category]) and mother’s partner’s smoking (comparing partner smoking during pregnancy with no partner smoking during pregnancy [reference category]). Predicted mean differences associated with maternal smoking (in white) and partner smoking (in black) are adjusted for cohort, infant sex, parity (for maternal smoking only), and respective parental age, height, BMI, education level, and alcohol use during pregnancy, and mutually adjusted for the smoking behaviour of the other parent.
https://doi.org/10.1371/journal.pmed.1002972.g004
Additional analyses
Effect estimates of the association between mother’s and mother’s partner’s smoking status during pregnancy with fetal growth were not materially different in analyses restricted to those with complete covariate data ( S8 – S12 Figs). Associations were very similar in GenR and BiB, with no statistical evidence for any differences between the 2 cohorts ( p interaction > 0.05 for all analysis models; S7 – S10 and S12 – S17 Tables). Also, associations of maternal smoking with birth weight were consistent in direction and effect size with the fetal size differences observed during late gestation, and conditioning on gestational age did not strongly influence these associations ( S18 Table ).
In this study we found that infants of pre-pregnancy smokers who continued smoking during pregnancy had a reduced fetal size from early second trimester to term, and that this association followed a dose-dependent pattern (with reductions in fetal size being present in light smokers but of lower magnitude than in moderate or heavy smokers) and was independent of observed confounding factors. Furthermore, the lack of an equivalent association for partner’s smoking with fetal growth and the results from MR, which showed evidence of a linear association of increasing cigarette consumption with fetal growth restriction, provide support that these associations represent an intrauterine effect and are not explained by unmeasured residual confounding factors. Pre-pregnancy smokers who gave up smoking before the second trimester had broadly similar fetal growth to non-smokers except for a shorter FL and greater AC between 36 and 40 weeks of gestation.
Strengths and limitations
A strength of our study is the large population-based sample with detailed information on smoking (including dose–response data) and on potential confounders. We triangulated evidence from conventional multivariable regression analyses in mothers with evidence from parental negative control and genetic MR analyses. The consistent findings across these 3 methods—which have differing key sources of bias (residual confounding in the multivariable analyses, unanticipated causal effect of partner smoking in the negative control, and genetic horizontal pleiotropy in MR)—strengthen confidence in our conclusions [ 13 ]. Multiple sensitivity analyses suggested that the multivariable regression analyses were not biased by selection due to missing covariate data, and that stratified genetic analyses were not notably influenced by collider bias. An assumption of MR analysis is that the genetic instrument influences the outcome only through the exposure of interest. One theoretical source of violation of this assumption is via fetal genotype, but we found no difference in the maternal SNP–fetal growth associations with adjustment for fetal genotype. The positive association of maternal rs1051730 genotype with EFW between 32 and 40 weeks of gestation in non-smokers suggests that genetic pleiotropy may have resulted in violation of the assumption that the genetic instrument influences fetal growth only via maternal smoking. Previous MR studies using the rs1051730 genotype have demonstrated that smoking is causally related to a lower BMI in smokers and higher BMI, and waist and hip circumference, in non-smokers [ 27 , 28 ]. This raises the possibility that the association of the variant with offspring EFW in non-smokers is mediated through an increase in maternal body mass and that in smokers associations may be stronger (i.e., there may have been some masking of the association by a pleiotropic path through maternal BMI). Our MR results are consistent with those from a large study of 26,241 mother–infant pairs, which found a negative association of rs1051730 with birth weight in smokers but no strong evidence of an association in non-smokers (difference in mean birth weight per T allele = 5 g [95% CI −4 to 14]) [ 8 ].
The parental negative control study assumes that factors related to maternal smoking are similar to those related to their partner’s smoking (i.e., that confounding is the same for the ‘real’ and ‘negative control’ exposure), which is plausible given that risk factors for smoking, such as low socioeconomic position, have not been shown to be sex-specific. This approach also assumes that it is not plausible that the negative control influences the outcome. Whilst partner smoking, and that of others whom the mother is exposed to, could have an intrauterine effect via passive smoking, we would expect this effect to be considerably smaller than what is seen with maternal smoking. The fact that we see in our analyses presented here a maternal-specific association with fetal growth parameters, as has been shown for birth weight [ 9 ], supports an intrauterine mechanism that is not explained by shared familial confounding.
In both GenR and BiB, pregnancies were dated using fetal biometry using CRL and biparietal diameter. Although this approach is superior to the use of women’s self-report of last menstrual period, it assumes no variation in fetal size at the time of dating, which may have led to an underestimation of smoking associations. Moreover, as there is some evidence of maternal smoking being associated with reduced first trimester CRL [ 11 ], systematic underestimation of gestational age in women who smoke during pregnancy could have further limited our ability to detect (stronger) associations. Since we modelled longitudinal fetal growth, and since ultrasound fetal anthropometrics tend to correlate less with dating measures as pregnancy progresses, we expect this to have resulted in an underestimation of fetal size differences only during early gestation. We further note that associations of maternal smoking with fetal AC attenuated towards term, most likely because of limited power as AC was not measured at birth in GenR. Information about smoking during pregnancy was collected by questionnaire. Several studies using cotinine as a biochemical measure of smoking exposure have demonstrated underreporting of smoking among pregnant women [ 29 , 30 ]. It is therefore likely that not all women in our study admitted smoking and when reporting smoking may have systematically underestimated the number of cigarettes smoked or falsely reported having quit. In the multivariable regression analyses, this misreporting could have resulted in associations with whether mothers smoked or not being biased towards the null, and associations with light/moderate smoking and smoking cessation being biased away from the null. In MR analyses, this misclassification would be expected to potentially bias results for non-smokers and quitters towards the results for smokers (i.e., as the group of non-smokers and quitters may have included some smokers), but not to bias causal effect estimates for smoking quantity within smokers.
Partners can be difficult to recruit to pregnancy cohorts, and data on behaviours like smoking is sometimes collected from the pregnant woman (as was the case for most partners in GenR) or there is a smaller proportion of partners with data (as was the case for BiB) [ 31 ]. The fact that we found consistent results for partner smoking in GenR, where information on partner smoking was based predominantly on maternal report (which may be biased towards the maternal association), and BiB, in which only a small proportion of partners responded (where there may be selection bias), suggests that the null findings for partner smoking are robust. Finally, as this study included women of white European origin only, results may not necessarily generalise to other populations.
Comparison with other studies
Our results are consistent with those from a systematic review, which reported lower third trimester fetal size and second trimester reductions in some fetal parameters in women who continued smoking during pregnancy [ 12 ]. That review, however, could not assess fetal growth trajectories across pregnancy from early gestation to term, or explore dose-dependent patterns or the impact of quitting in detail, and it did not include methods we used to explore causality. Our MR estimates of EFW reductions of 37 g (95% CI 14 to 60) and 45 g (95% CI 10 to 81) per genetically determined 1-cigarette-per-day increase at 36 and 40 weeks’ gestation in women who continued smoking beyond the first trimester are broadly consistent with the 24-g (95% CI 3 to 45) reduction in birth weight reported in a previous MR analysis [ 8 ]. Our genetic results are also comparable with those from a randomized smoking cessation trial showing a reduction of approximately 5.4 cigarettes per day and a 92-g heavier birth weight in the intervention compared to the control group (giving an average increase of 17.9 g per 1 less cigarette) [ 5 ]. Of note, rs1051730 genotype is an instrument for both smoking quantity (in current smokers, i.e., pre-pregnancy smokers who continue smoking during pregnancy) and smoking cessation (in pre-pregnancy smokers), 2 independent traits that are strongly correlated. As we found similar associations in analyses including all pre-pregnancy smokers, our results support the importance of smoking reduction and cessation to prevent fetal growth restriction. The absence of an independent association of mother’s partner’s smoking with fetal growth provides additional support for maternal smoking influencing early fetal growth through an intrauterine mechanism and is consistent with a previous negative control study investigating parental smoking associations with birth weight [ 9 ].
Different fetal parameters have different peak periods of growth during gestation as reflected by the shapes of their growth trajectories, i.e., linear growth of HC and FL is highest during early pregnancy and starts to level off at approximately 20 weeks of gestation, while the peak growth of AC is observed between approximately 20 and 32 weeks (reflecting the predominant accumulation of fat and lean tissue during the third trimester) [ 32 ]. These differences in fetal growth patterns may affect the timing at which associations with each fetal parameter can be observed. Differences in smoking effects on fetal growth parameters may also reflect developmental plasticity and the ‘protection’ of some organs and tissues over others. In our study, associations of maternal smoking with fetal growth were first noted for FL, with differences in FL being detectable from as early as 16 weeks’ gestation. Compared to non-smokers, pre-pregnancy smokers who quit smoking before the second trimester had a shorter fetal FL at 36–40 weeks’ gestation, whereas no differences in size were observed for other growth parameters. If the toxic effects of smoking affected the growth of different tissues and organs similarly and the main cause of differences between them was related to timing of peak growth, we would have expected similar patterns between FL and HC, which is not what we find.
A predominant association of maternal smoking with FL has been reported before by other studies including GenR [ 20 , 33 – 35 ] and animal experiments [ 36 ], and for many years, leg length has been proposed to be the most sensitive growth parameter to adverse early-life exposures [ 37 ]. A Swedish register-based study found that infants of women who quit smoking during pregnancy had a similar birth weight and HC to infants of non-smokers, but a shorter crown–heel length [ 38 ]. Since birth crown–heel length is a measure of linear growth that is closely correlated to FL [ 39 ], these data are consistent with our findings. Taking our findings together with those from human and animal experimental studies, it appears that fetal skeletal linear growth is particularly susceptible to maternal cigarette smoking. It seems plausible that in the presence of reduced nutrient and oxygen supply to the fetus, as a result of smoking [ 40 ], key organs, such as the brain and liver, are preserved as much as possible at the expense of skeletal growth. We further observed a greater fetal AC towards the end of gestation in women who quit smoking early in pregnancy compared to non-smokers. Smoking cessation is often accompanied by weight gain and changes in central fat tissue [ 41 , 42 ], most likely through an increased appetite and lower basal metabolic rate upon nicotine withdrawal. This could potentially explain the larger fetal AC found among quitters, but because of the small number of quitters in this study, we were unable to explore the causal nature of this association further in MR analysis.
Implications
Harmful effects of maternal smoking during pregnancy are well known. However, many women who smoke find it difficult to quit before or during pregnancy [ 43 ], as evidenced by the relatively high rates of smoking prior to pregnancy (26% and 41%) and low quit rates in early pregnancy (35% and 27% of pre-pregnancy smokers) in the 2 contemporary cohorts of white European women in our study. Our study is the first to our knowledge to provide robust support for an association between the number of cigarettes smoked and fetal size from as early as 20 weeks’ gestation in women who continue smoking after the first trimester. There are 2 key implications of these findings. First, even at low doses, cigarette smoking adversely affects fetal growth, and continued efforts to prevent women of reproductive age from smoking, and to support those who do take it up to quit, are paramount. In this context, our findings reinforce existing recommendations [ 44 – 46 ] that encourage strategies to reduce initiation, improve detection, and promote cessation of tobacco use including cessation counselling as part of preconception health services and antenatal care. Simultaneously, the absence of major fetal growth deficits observed with smoking cessation early in pregnancy should comfort women with unplanned pregnancies who are able to quit smoking early after realising they are pregnant, but who may have experienced anxiety because of smoking in that early period of pregnancy. Second, for women who do not manage to quit smoking before or during pregnancy, smoking reduction will have some benefit in a linear fashion, i.e., the greater the reduction in smoking, the smaller the likelihood of fetal growth restriction; therefore, support to lower the number of cigarettes smoked in these women should be promoted. Currently, few guidelines suggest this, perhaps because of fear that it may be interpreted as condoning light smoking in pregnancy. We believe our results provide sufficient evidence to update guidelines that currently promote only quitting, not reducing, smoking: For women who find it too difficult to quit smoking before or during pregnancy, support to reduce the amount they smoke should be provided. We acknowledge that fetal growth restriction, despite being a predictor of neonatal morbidity and mortality, is only one of the adverse outcomes associated with pregnancy smoking, and it is unknown whether lowering cigarette consumption in those who cannot quit will be beneficial for other perinatal outcomes. Finally, our findings should stimulate more research to further explore the effectiveness and safety of currently available interventions that might reduce smoking in pregnancy [ 47 ]. The potential effect of electronic cigarettes and other smokeless tobacco products, which women may use as a means to quit smoking or as potentially safer alternatives to cigarette smoking, on fetal growth and perinatal outcomes requires investigation. However, as these alternatives have only recently become available, we did not have information on their use in either of these cohorts, and it will take some time before other cohorts with sufficient information on these will be available for analyses.
By triangulating findings from different analytical approaches, this study provides strong support for a dose-dependent effect of cigarette smoking on fetal growth from the second trimester onwards in women who continue smoking during pregnancy, while only minor deficits in fetal growth are seen in women who quit smoking early in pregnancy. Collectively, these findings reinforce the importance of smoking cessation counselling in preconception health services and antenatal care. They demonstrate the importance of quitting smoking early in pregnancy, which is knowledge that could also help reduce anxiety in women who quit after learning they are pregnant. Lastly, our findings indicate that support and advice to reduce smoking during pregnancy in women who find it impossible to quit has some benefit in lowering the risk of fetal growth restriction.
Supporting information
S1 fig. flow chart of the study population..
https://doi.org/10.1371/journal.pmed.1002972.s001
S2 Fig. Best-fitting growth trajectories for each fetal parameter identified by multilevel fractional polynomial models in GenR and BiB.
Average growth trajectories of fetal HC, FL, AC, and EFW from 12–16 to 40 weeks’ gestation predicted by best-fitting multilevel fractional polynomial models in GenR and BiB.
https://doi.org/10.1371/journal.pmed.1002972.s002
S3 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation per risk allele increase at rs1051730 in pre-pregnancy smokers and non-smokers.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation per maternal rs1051730 T allele increase in pre-pregnancy smokers and non-smokers. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort (in white) and model 2 adjusting for cohort and fetal rs1051730 genotype (in black).
https://doi.org/10.1371/journal.pmed.1002972.s003
S4 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation per risk allele increase at rs1051730 in non-smokers, pre-pregnancy smokers who quit in early pregnancy, and pre-pregnancy smokers who continued smoking during pregnancy—Analysis with adjustment for maternal age.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation per maternal rs1051730 T allele increase in non-smokers, pre-pregnancy smokers who quit smoking before the second trimester, and pre-pregnancy smokers who continued smoking during pregnancy. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort and maternal age (in white) and model 2 adjusting for cohort, maternal age, and fetal rs1051730 genotype (in black).
https://doi.org/10.1371/journal.pmed.1002972.s004
S5 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation per risk allele increase at rs1051730 in pre-pregnancy smokers and non-smokers—analysis with adjustment for maternal age.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation per maternal rs1051730 T allele increase in pre-pregnancy smokers and non-smokers. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort and maternal age (in white) and model 2 adjusting for cohort, maternal age, and fetal rs1051730 genotype (in black).
https://doi.org/10.1371/journal.pmed.1002972.s005
S6 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing mothers’ partners who smoked with non-smoking partners (reference category).
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation comparing partners who smoked during pregnancy to those who did not smoke during pregnancy (reference category). Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort only (in white); model 2 adjusting for cohort, infant sex, and partner age, height, body mass index, education level, and alcohol use during pregnancy (in grey); and model 3 adjusting for cohort; infant sex; partner age, height, body mass index, education level, and alcohol use during pregnancy; and smoking during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.s006
S7 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing different categories of smoking intensity in mothers’ partners who smoked with non-smoking partners (reference category).
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation comparing partners who smoked during pregnancy by smoking intensity with those who did not smoke during pregnancy (reference category). Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort only (in white); model 2 adjusting for cohort, infant sex, and partner age, height, body mass index, education level, and alcohol use during pregnancy (in grey); and model 3 adjusting for cohort; infant sex; partner age, height, body mass index, education level, and alcohol use during pregnancy; and maternal smoking during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.s007
S8 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing pre-pregnancy smokers who quit in early pregnancy and pre-pregnancy smokers who continued smoking through pregnancy with non-smokers (reference category)—Complete case analysis.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation comparing pre-pregnancy smokers who quit smoking in early pregnancy and those who continued smoking during pregnancy with non-smokers (reference category)—complete case analysis. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort only (in white) and model 2 adjusting for cohort, infant sex, and maternal age, parity, height, body mass index, education level, and alcohol use during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.s008
S9 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing different categories of smoking intensity in pre-pregnancy smokers who continued smoking through pregnancy with non-smokers (reference category)—Complete case analysis.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation comparing pre-pregnancy smokers who continued smoking through pregnancy by smoking intensity with non-smokers (reference category)—complete case analysis. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort only (in white) and model 2 adjusting for cohort, infant sex, and maternal age, parity, height, body mass index, education level, and alcohol use during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.s009
S10 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing mothers’ partners who smoked with non-smoking partners (reference category)—Complete case analysis.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation comparing partners who smoked during pregnancy with those who did not smoke during pregnancy (reference category)—complete case analysis. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort only (in white); model 2 adjusting for cohort, infant sex, and partner age, height, body mass index, education level, and alcohol use during pregnancy (in grey); and model 3 adjusting for cohort; infant sex; partner age, height, body mass index, education level, and alcohol use during pregnancy; and maternal smoking during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.s010
S11 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing different categories of smoking intensity in mothers’ partners who smoked with non-smoking partners (reference category)—Complete case analysis.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation comparing partners who smoked during pregnancy by smoking intensity with those who did not smoke during pregnancy (reference category)—complete case analysis. Predicted mean differences (with 95% confidence intervals) in the pooled GenR and BiB cohort by analysis model: model 1 adjusting for cohort only (in white); model 2 adjusting for cohort, infant sex, and partner age, height, body mass index, education level, and alcohol use during pregnancy (in grey); and model 3 adjusting for cohort; infant sex; partner age, height, body mass index, education level, and alcohol use during pregnancy; and maternal smoking during pregnancy (in black).
https://doi.org/10.1371/journal.pmed.1002972.s011
S12 Fig. Predicted differences in mean fetal size (with 95% CIs) across gestation comparing mothers and mothers’ partners who smoked to non-smokers (reference category)—Complete case analysis.
Predicted differences in mean HC (mm), FL (mm), AC (mm), and EFW (g) across gestation associated with maternal smoking (i.e., comparing maternal continued smoking through pregnancy with no maternal smoking during pregnancy [reference category]) and mother’s partner’s smoking (comparing partner smoking during pregnancy with no partner smoking during pregnancy [reference category])—complete case analysis. Predicted mean differences associated with maternal smoking (in white) and partner smoking (in black) are adjusted for cohort, infant sex, parity (for maternal smoking only), and respective parental age, height, BMI, education level, and alcohol use during pregnancy, and mutually adjusted for the smoking behaviour of the other parent.
https://doi.org/10.1371/journal.pmed.1002972.s012
S1 Table. Genotype quality control measures in GenR and BiB.
https://doi.org/10.1371/journal.pmed.1002972.s013
S2 Table. Descriptive statistics of the repeat ultrasound and birth anthropometric measurements in GenR and BiB.
https://doi.org/10.1371/journal.pmed.1002972.s014
S3 Table. Comparison of observed means of fetal size with those predicted by the multilevel fractional polynomial model in GenR and BiB.
https://doi.org/10.1371/journal.pmed.1002972.s015
S4 Table. Participant characteristics by availability of maternal rs1051730 genotype and partner smoking data.
https://doi.org/10.1371/journal.pmed.1002972.s016
S5 Table. Participant characteristics by maternal smoking status during pregnancy.
https://doi.org/10.1371/journal.pmed.1002972.s017
S6 Table. Participant characteristics by partner smoking status during pregnancy.
https://doi.org/10.1371/journal.pmed.1002972.s018
S7 Table. Maternal smoking during pregnancy and predicted differences in mean fetal size (with 95% CIs) across gestation, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s019
S8 Table. Maternal smoking intensity in continuing smokers and predicted differences in mean fetal size (with 95% CIs) across gestation, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s020
S9 Table. Maternal smoking during pregnancy and predicted differences in mean fetal size across gestation as a proportion of the mean, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s021
S10 Table. Maternal smoking intensity in continuing smokers and predicted differences in mean fetal size across gestation as a proportion of the mean, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s022
S11 Table. Association of maternal rs1051730 genotype with maternal smoking variables.
https://doi.org/10.1371/journal.pmed.1002972.s023
S12 Table. Predicted differences in mean fetal size (with 95% CIs) across gestation per risk allele increase at rs1051730 in non-smokers, pre-pregnancy smokers who quit in early pregnancy, and pre-pregnancy smokers who continued smoking through pregnancy, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s024
S13 Table. Predicted differences in mean fetal size across gestation as a proportion of the mean per risk allele increase at rs1051730 in non-smokers, pre-pregnancy smokers who quit in early pregnancy, and pre-pregnancy smokers who continued smoking through pregnancy, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s025
S14 Table. Predicted differences in mean fetal size (with 95% CIs) across gestation per risk allele increase at rs1051730 in pre-pregnancy smokers and non-smokers, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s026
S15 Table. Association of maternal rs1051730 genotype with potential confounders.
https://doi.org/10.1371/journal.pmed.1002972.s027
S16 Table. Parental smoking during pregnancy and predicted differences in mean fetal size (with 95% CIs) across gestation, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s028
S17 Table. Parental smoking during pregnancy and predicted differences in mean fetal size across gestation as a proportion of the mean, overall and stratified by cohort.
https://doi.org/10.1371/journal.pmed.1002972.s029
S18 Table. Predicted differences in mean infant birth weight in grams (with 95% CIs) associated with maternal smoking during pregnancy and smoking intensity in continuing smokers.
https://doi.org/10.1371/journal.pmed.1002972.s030
S1 Text. Supplemental methods.
https://doi.org/10.1371/journal.pmed.1002972.s031
S2 Text. Analysis plan.
https://doi.org/10.1371/journal.pmed.1002972.s032
S3 Text. STROBE checklist.
https://doi.org/10.1371/journal.pmed.1002972.s033
Acknowledgments
BiB is only possible because of the enthusiasm and commitment of the children and parents. We are grateful to all the participants, practitioners, and researchers who have made BiB happen.
GenR is conducted by Erasmus University Medical Center Rotterdam, in close collaboration with the School of Law and Faculty of Social Sciences of the Erasmus University Rotterdam; the Municipal Health Service for Rotterdam area, Rotterdam; the Rotterdam Homecare Foundation Rotterdam; and the Stichting Trombosedienst & Artsenlaboratorium Rijnmond (STAR-MDC), Rotterdam. We gratefully acknowledge the contribution of participating children and mothers, parents, general practitioners, hospitals, midwives, and pharmacies in Rotterdam. The generation and management of genome-wide association study (GWAS) genotype data was performed by the Human Genotyping Facility of the Genetic Laboratory of the Department of Internal Medicine, Erasmus MC, Rotterdam, the Netherlands. We thank Pascal Arp, Mila Jhamai, Marijn Verkerk, Lizbeth Herrera, and Marjolein Peters for their help in the creation, management, and quality control of the GWAS database, and Karol Estrada and Carolina Medina-Gomez for their support in the creation and analysis of imputed data. We would like to thank Anis Abuseiris, Karol Estrada, Dr. Tobias A. Knoch, and Rob de Graaf as well as their institution, Biophysical Genomics, Erasmus MC, Rotterdam, the Netherlands, for their help in creating GRIMP, BigGRID, MediGRID, and Services@MediGRID/D-Grid for access to their grid computing resources.
- View Article
- PubMed/NCBI
- Google Scholar
- 3. Office of the Surgeon General, Office on Smoking and Health. The health consequences of smoking: a report of the Surgeon General. Atlanta: Centers for Disease Control and Prevention; 2004.
- 4. Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. How tobacco smoke causes disease—the biology and behavioral basis for smoking-attributable disease: a report of the Surgeon General. Atlanta: Centers for Disease Control and Prevention; 2010.
- 22. Royal College of Obstetricians and Gynaecologists. Routine ultrasound screening in pregnancy: protocol. London: RCOG Press; 2000.
- 44. World Health Organization. WHO recommendations for the prevention and management of tobacco use and second-hand smoke exposure in pregnancy. Geneva: World Health Organization; 2013.
- 46. National Institute for Health and Care Excellence. Smoking: stopping in pregnancy and after childbirth. Public health guideline [PH26]. London: National Institute for Health and Care Excellence; 2010 [cited 2019 Oct 23]. Available from: http://www.nice.org.uk/PH26 .
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Population Study Article
- Open access
- Published: 09 February 2023
Association of maternal snuff use and smoking with Sudden Infant Death Syndrome: a national register study
- Anna Gunnerbeck ORCID: orcid.org/0000-0003-1808-1091 1 , 2 ,
- Cecilia Lundholm 1 ,
- Samuel Rhedin 1 , 3 ,
- Ayoub Mitha 4 , 5 ,
- Ruoqing Chen 6 , 7 ,
- Brian M. D’Onofrio 1 , 8 &
- Catarina Almqvist 1 , 9
Pediatric Research volume 94 , pages 811–819 ( 2023 ) Cite this article
4783 Accesses
4 Citations
257 Altmetric
Metrics details
The aim was to study whether non-combustible nicotine (Swedish snuff) use in pregnancy is associated with elevated risk of post neonatal mortality, Sudden Infant Death Syndrome (SIDS), and Sudden Unexpected Infant Death (SUID) and to study how cessation before the antenatal booking influenced these risks.
This was a population-based register study of all infants with information on tobacco exposure in early pregnancy born in Sweden 1999–2019, n = 2,061,514. Self-reported tobacco use in early pregnancy was categorized as nonuse, snuff use, and moderate and heavy smoking. Multiple logistic regression models were used to estimate crude and adjusted odds ratios (aORs) with 95% confidence intervals (CIs).
Maternal snuff use was associated with increased risks of post neonatal mortality, SIDS, and SUID. The risks of snuff use and moderate smoking were of similar magnitude. Heavy smoking was associated with the highest risks. Cessation of smoking and snuff use before the antenatal booking was associated with lower risks of SIDS and SUID compared to that of continuous usage.
Conclusions
Maternal snuff use was associated with increased risks of post neonatal mortality, SIDS, and SUID. Nicotine is the common substance in cigarette smoke and snuff. These findings support the hypothesis that nicotine contributes to an elevated risk of SIDS.
Maternal snuff use and smoking in early pregnancy were associated with increased risks of post neonatal mortality, SIDS, and SUID.
Cessation of smoking and snuff use before the first antenatal visit was associated with reduced risks of SIDS and SUID.
The common substance in cigarette smoke and snuff is nicotine. Our findings suggest that nicotine contributes to an elevated risk of SIDS and SUID.
The implication of our findings is that all forms of nicotine should be avoided in pregnancy.
Similar content being viewed by others

Racial differences in the impact of maternal smoking on sudden unexpected infant death
Association between maternal smoke exposure and congenital heart defects from a case–control study in China
Predictive factors and adverse perinatal outcomes associated with maternal smoking status, introduction.
Maternal smoking is one of the most important preventable risk factors for infant morbidity and mortality and associated with increased risk of Sudden Infant Death Syndrome (SIDS). 1 , 2 SIDS is a cause assigned to infant deaths that cannot be explained after a thorough case investigation including autopsy, a scene investigation and a review of clinical history. Due to more advanced forensic techniques in recent years, there has been a diagnostic shift in many countries with increased reporting of accidental suffocation and strangulation in bed and a subsequent decline of reported cases of SIDS. 3 Thus, the term Sudden Unexpected Infant Death (SUID) is frequently used, including SIDS, unspecified death, and accidental suffocation and/or strangulation. 4 Although the risk of SIDS is multifactorial, maternal smoking, following sleeping position, remains the strongest modifiable risk factor for SIDS in developed countries. 1 , 4 SIDS is suggested to be related to a disturbed cardiorespiratory control and a blunted response to hypoxia with impaired arousal. 1 Animal studies of prenatal nicotine exposure have shown similar disturbances in the cardiorespiratory control as seen in infants exposed to smoking in pregnancy, which suggests that nicotine is involved in the mechanisms behind smoking-related risks of SIDS. 5
With increasing knowledge about the hazardous effects of smoking on women’s and children’s health, smoking in pregnancy has declined in Sweden from 13% to <5% between 1999 and 2019. 6 Non-combustible nicotine products are regarded less harmful than cigarette smoking, and as a consequence the use of smokeless tobacco and electronic nicotine delivery systems (ENDS) has increased during the past decades. 7 Swedish snuff is an oral moist powder and contains equally high levels of nicotine as cigarettes, but no combustion products and only low levels of tobacco-specific nitrosamines. 8 Snuff use among women has increased substantially the past decades and daily snuff use in Sweden is approximately 7% of women in reproductive age.
However, among women aged 16–29 years snuff use has tripled from 3% in 2018 to 9% in 2021. 9 Approximately 1.8% of Swedish women used snuff in pregnancy in 2021. 9 Whereas sufficient knowledge about nicotine exposure from electronic cigarettes in pregnancy still are lacking, 10 , 11 maternal snuff use has been associated with increased risk of stillbirth, preterm birth, neonatal apnea, and disturbed heart rate variability. 12 , 13 , 14 , 15 However, in contrast to smoking, snuff use has not been associated with increased risk of early neonatal mortality. 14 Whether nicotine, in the form of Swedish snuff, increases the risk of post neonatal mortality and SIDS has to our knowledge not previously been studied.
The aim of this study was to investigate the associations of snuff use and smoking in early pregnancy with post neonatal mortality and SIDS. Because of the increased use of the terminology SUID in many countries, we chose to study both SIDS and SUID. Further, we wanted to study how cessation of snuff use or smoking in very early pregnancy, before the first antenatal visit, influenced these associations.
Study population
Based on the Swedish Medical Birth Register (MBR), we included all live-born infants, born between 1999 and 2019, with information on gestational age, birth weight, and mothers’ individual identification number, n = 2,181,021 births. Infants who died during the first year of life, but with invalid date of death, were excluded, n = 2. The eligible study population was 2,181,019. We excluded infants of mothers who had missing information on tobacco use in early pregnancy ( n = 117,865). Dual users (users of both cigarettes and snuff) were too few ( n = 1640) to be analyzed separately and were also excluded. A total of n = 2,061,514 births were included in the analyses with tobacco use in early pregnancy as exposure, Fig. 1 .
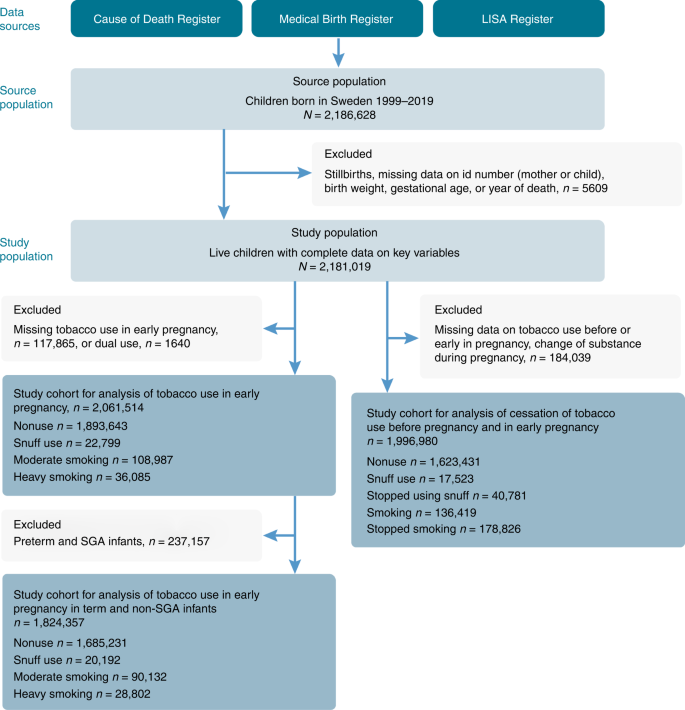
Flow chart of the source population, exclusions, the final study population as well as categorization of exposures.
The MBR is a national register containing information on the demography, maternal reproductive history, pregnancy, delivery, and neonatal period for 98% of all births in Sweden. By means of the unique personal identification number, the MBR can be linked to other data sources such as the Cause of Death Register and the Longitudinal integrated database for health insurance and labor market studies (LISA) and the Register of Total Population. The Cause of Death Register includes all deaths of Swedish citizens with annual updates, while LISA includes information on education for 98% of Swedish residents. 16 Since 1997, diagnoses in the MBR and the Cause of Death Register are classified according to the 10th Revision of the WHO International Classification of Diseases (ICD-10).
Snuff use and smoking in pregnancy
Self-reported data on maternal smoking have been registered in the MBR since 1982 and maternal snuff use since 1999. Information on present tobacco use and use 3 months before pregnancy was collected at the antenatal booking, which generally occurs between gestational weeks 8 and 12. Maternal tobacco use was categorized as nonuser (no tobacco use), moderate smoker (1–9 cigarettes /day), heavy smoker (≥10 cigarettes/day), and snuff user (yes/no). Information on the number of pouches of snuff used daily is lacking in the MBR, which only allowed us to categorize women as user or nonuser.
To investigate whether cessation of tobacco use in very early pregnancy, before the antenatal booking, influenced the risk of post neonatal mortality, SIDS, and SUID, we combined information on tobacco use 3 months before pregnancy with use in early pregnancy. Exact quit date of tobacco use was not possible to obtain. Mothers who neither used tobacco before nor in early pregnancy were categorized as nonusers. Snuff users 3 months before pregnancy and in early pregnancy were categorized as persistent snuff users. Smokers 3 months before pregnancy and in early pregnancy were categorized as persistent smokers. Mothers who used snuff or smoked before pregnancy but not at the antenatal booking were categorized as quitters of snuff or smoking, respectively. We excluded infants of mothers lacking information on tobacco use before and/or in early pregnancy, dual users, and mothers who changed substance of tobacco or who started to use tobacco during pregnancy ( n = 184,039). A total of 1,996,980 live births were included when studying cessation of tobacco use before the antenatal booking (Fig. 1 ).
Post neonatal death, SIDS, and SUID
Post neonatal mortality was defined as death between 28 and 364 days. SUID is a term used to describe any sudden and unexpected death occurring before 1 year of age. In Sweden, unexpected infant death is thoroughly investigated with anamnesis, medical history, examination, and laboratory testing. However, in contrast to some others countries crime scene investigations are not routinely performed in Sweden. 17 Autopsy of the infant is routinely performed by the forensic pathologist and the diagnosis reported to the Cause of Death Register. 18 After case investigation, if SUID cannot be explained otherwise, it is categorized as death before 365 days from birth and including SIDS (R95), unspecified causes (R99), or strangulation/suffocation in bed (W75). SIDS was categorized as death before 365 days and ICD-10 R95.
Covariates were chosen based on their association with both tobacco use in pregnancy and neonatal death in previous research. 2 Preterm or small for gestational age (SGA) birth is highly associated with both tobacco use, post neonatal mortality, and SIDS and potentially on the causal pathway between the exposure and the outcome. Thus, preterm and SGA births were considered possible mediators for the associations of tobacco use and risks of post neonatal mortality, SIDS, and SUID. SGA was defined as birth weight below the tenth percentile for gestational age according to population-based birth weight standards. Preterm birth was defined as birth before 37 gestational weeks. We retrieved information on maternal age, parity, co-habitant with father-to-be, the child’s gestational age and birth weight from the MBR, and information on mother’s level of education from LISA. Mother’s country of birth was obtained from the Register of Total Population. The categorization of covariates are shown in Table 1 .
Statistical analyses
The rates for post neonatal mortality and cause of death were calculated as the number of deaths divided by the number of infants. Chi 2 test was used to calculate p values for maternal and birth characteristics. The logistic model was used to estimate crude and adjusted odd ratios (aORs) with 95% confidence interval (CI) for the association between tobacco use in early pregnancy and post neonatal mortality, SIDS, and SUID, as well as for the association between cessation of tobacco use before the antenatal booking and post neonatal mortality, SIDS, and SUID. Crude ORs were estimated on births with complete data on all covariates used in the first adjusted model, n = 1,883,726, (91% of the population). The first adjusted model included the covariates maternal age, parity, co-habitant with father-to-be, mother’s level of education at birth, and mother’s country of birth. We also wanted to investigate whether possible risks related to tobacco use were explained by preterm birth and SGA births. Therefore, in the second adjusted model, we additionally adjusted for gestational age and birth weight adjusted for gestational age. Further, we repeated our analyses with only term and non-SGA births included to study if potential associations were independent of effects on length of gestation or fetal growth.
To examine the potential effect of missing information on tobacco use, we also conducted analyses assuming the extreme cases that all missing values were either related to tobacco users or nonusers. All analyses were performed using SAS version 9.4 (Statistical Analysis Software version 9.4, SAS Institute, Inc, Cary, NC).
In the study, 145,072 (7%) of the mothers smoked and 22,799 (1.1%) used snuff in early pregnancy. More than 50% of smokers 3 months prior to pregnancy and almost 70% of previous snuff users had stopped smoking or using snuff at their first antenatal visit (data not shown). Maternal tobacco use, and especially heavy smoking, was more common among young mothers (≤20 years at delivery), mothers not co-habitant with the father-to-be, and mothers who were multiparous and had a low level of education (<12 years of schooling). Maternal smoking was more common among mothers born outside a Nordic country, whereas snuff use was more common among mothers born in a Nordic country. Preterm birth was more common among smoking and snuff using mothers, and SGA births were more common among mothers who smoked (Table 1 ).
The post neonatal mortality rate was 0.8 per 1000 live-born infants. The highest rates of post neonatal mortality, SIDS, and SUID were found among infants of mothers with low maternal age (<20 years at delivery), who were multiparous, not co-habitant with father-to-be, and with low level of education (<12 years of schooling). Further, post neonatal mortality, SIDS, and SUID were more common among preterm and SGA infants. Post neonatal mortality rates decreased with increasing gestational age. Of all the post neonatal deaths, almost 32% were preterm and 28% were SGA births (Table 2 ). Median age of post neonatal death was 3 months (interquartile range (IQR) 1–6). The median age of death due to SIDS was 2 months (IQR 1–3) and due to SUID 1 month (IQR 0–3) (Data not shown).
Both maternal snuff use and smoking were associated with higher risk of post neonatal mortality. Snuff use in pregnancy was associated with 70% higher risk of post neonatal mortality in comparison to that of nonusers, aOR 1.71 (95% CI: 1.16–2.53). The highest risk of post neonatal mortality compared to that of nonusers was found among infants of heavy smokers, aOR 2.72 (95% CI: 2.17–3.40) (Table 3 ).
There were a total of 611 cases of SUID. Of these, 60% were due to SIDS ( n = 381), 11 cases of accidental strangulation in bed, and 219 death by unspecified cause. Frequency plot of SIDS, unspecified death, and accidental strangulation in bed by years is shown in Fig. 2 . In Sweden, the frequency of reporting unspecified death or accidental strangulation cannot fully explain the fluctuations in SIDS cases. The total number of cases of SIDS and SUID has varied substantially over the years. There has been a gradual decrease, however, and since 2015, the number of SUID cases has been below 30 and SIDS cases below 20.
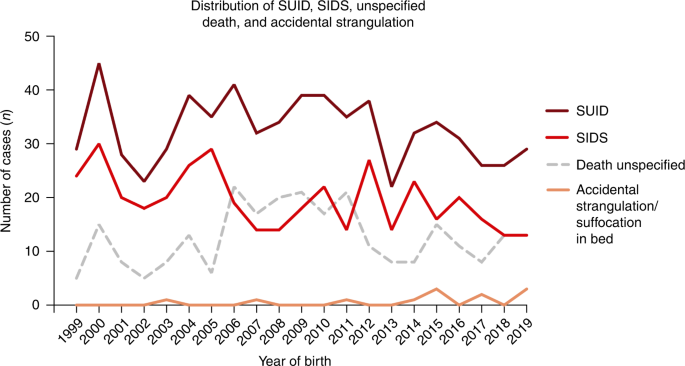
Presentation of number of deaths/year in 1999–2019 of Sudden Unexpected Infant Death (SUID) as well as presentation of deaths of Sudden Infant Death Syndrome (SIDS), accidental strangulation/suffocation and unspecified death of the newborn.
Snuff use in early pregnancy was associated with a more than tripled risk of SIDS, aOR 3.70 (95% CI: 2.06–6.65) compared to that of nonuse. Maternal smoking was also associated with higher risk of SIDS, aORs were 4.31 (95% CI: 3.26–5.70) and 7.03 (95% CI: 5.04–9.79) for moderate and heavy smoking, respectively. Maternal snuff use was associated with an almost tripled risk of SUID, aOR 2.90 (95% CI: 1.75–4.80). Maternal smoking was also associated with higher risk of SUID, with aORs 3.20 (95% CI: 2.53–4.05) and 5.44 (95% CI: 4.10–7.22) for moderate and heavy smoking, respectively. The associations were attenuated after adjustment for possible confounders in all analyses and were most pronounced in the analyses of maternal smoking (Table 3 ). The associations between maternal snuff use or smoking and post neonatal mortality, SIDS, and SUID in the subgroup analysis of only term and non-SGA infants ( n = 1,824,357) were similar to the analyses of the full cohort (Table S1 ).
In infants of mothers who stopped using snuff or stopped smoking before the antenatal booking, there were no overall statistically significant higher risks of post neonatal mortality. However, among infants of mothers who had stopped using snuff before the antenatal booking the risk of SIDS was lower but still higher compared to that of nonusers, aOR 2.13 (95% CI: 1.04–4.34). In infants of mothers who had stopped smoking before the antenatal booking, the aOR was 1.35 (95% CI: 0.89–2.05) compared to that of nonusers. In infants of mothers who had stopped using snuff or smoking before the antenatal booking, risks of SUID was substantially lower, aOR 1.37 (95% CI: 0.73–2.58) and aOR 1.29 (95% CI: 0.95–1.74), respectively (Table 4 ). Results did not substantially differ for any of the outcomes regardless of whether individuals with missing information on tobacco use in pregnancy were assumed to be all tobacco users or all non-tobacco users (data not shown).
In this population-based cohort study, including >2 million infants, we found that maternal snuff use in pregnancy was associated with a >70% higher risk of post neonatal death and a tripled risk of SIDS compared to that of nonusers. The risks of snuff use and moderate smoking were of similar magnitude. The highest risks were found among infants of heavy smokers, with a more than sevenfold higher risk of SIDS compared to that of nonusers. Both maternal snuff use and smoking were associated with higher risk of SUID. In our population, the majority of SUID cases among tobacco users were due to SIDS. Cessation of smoking and snuff use before the antenatal booking was associated with lower risks of SIDS and SUID.
Despite a study population of more than two million infants, the number of SIDS cases was small. In the cohort, only 0.2 per 1000 live-born infants died of SIDS and <7% of women smoked and only 1% used snuff in pregnancy. However, despite small numbers, the estimates and confidence intervals are in agreement with international studies on smoking, post neonatal mortality, SIDS and SUID. 19 , 20 , 21 , 22 Our results are in agreement with Anderson et al. who showed that infants of mothers who had stopped smoking in early pregnancy ran a reduced risk of SUID compared to those whose mothers continued to smoke. 20 To speculate, the lower, but still elevated, risk observed in infants of mothers who had stopped using snuff before the antenatal booking may be due to residual confounding or environmental tobacco exposure. It is also likely that a proportion of women who used snuff or smoked pre-pregnancy and quit before pregnancy restarted later in pregnancy or in the postpartum period. It is also possible that among women who stopped using snuff or smoking as soon as they knew they were pregnant, the fetus had been exposed to snuff or cigarette smoking in very early pregnancy. As nicotine-acetylcholine receptors (nAChRs) are present as early as gestational week 4–5 in fetal development, before most women are aware of their pregnancy, nicotine may carry out teratogenic effects by binding these receptors, induce apoptosis, and affect cell programming. 23
The associations between maternal snuff use and SIDS remained fairly stable after adjusting for possible confounders, whereas the associations with especially heavy maternal smoking were considerably attenuated in the adjusted analyses. Snuff use is not to the same extent as smoking associated with low socioeconomic status. 24 Further, snuff use in pregnancy is associated with increased risk of preterm birth, but only with modest effects on birth weight, 25 which may explain why the risk of SIDS associated with snuff use was only slightly attenuated when adjusting for preterm and SGA births. By stratifying for gestational age and birth weight, we may have introduced collider stratification bias, suggesting those results must be interpreted with caution. However, the findings were similar to those of the full cohort, indicating that preterm birth and birth weight may not fully explain the association between nicotine use and SIDS.
The risk of SIDS is multifactorial, and prone sleeping position, bed sharing, and prenatal and postnatal exposure to tobacco smoke are regarded as important risk factors. 1 , 26 Most SIDS deaths occur at around 2–4 months of age when the brain and cardiorespiratory systems mature. 27 It has been suggested that SIDS is related to disturbed autonomous control and disturbances in the serotonergic neural development. 1 Maternal smoking has been associated with similar disturbances in the cardiorespiratory and serotonergic system. 1 , 28 Further, animal studies of prenatal nicotine suggest that nicotine is involved in the mechanisms behind smoking-related risks of SIDS. 5 , 23 Maternal snuff use has been associated with neonatal apnea and disturbed heart rate variability. 12 , 15 The common substance in cigarette smoke and snuff is nicotine. The peak plasma concentration between cigarettes and snuff is similar. However, whereas smoking generates short peaks (30 min) of high plasma concentration intermittently during the day, snuff has a slower release and a longer peak duration, 1.5–2 h. 29 Snuff use leads to higher accumulated nicotine levels and a more continuous exposure on the nicotine receptors, compared with short, intermittent exposure in smoking, which may explain differences between effects of smoking and snuff use. 29 , 30 Our findings support the hypothesis that nicotine is involved in the mechanisms behind the increased risk of post neonatal mortality and SIDS in infants of smokers.
Strengths and limitations
The major strength with our study is the large study population of two million births and the nationwide design. Further, the prospectively collected tobacco information in early pregnancy precludes recall bias. This is, to our knowledge, the first study exploring snuff use in pregnancy and risk of post neonatal mortality, SIDS, and SUID. By linking several Swedish national registers, we were able to control for several important confounders, including socioeconomic status, parity, and maternal age.
Nicotine replacement therapy (NRT) is often regarded a safe alternative in pregnancy. However, safety studies of NRT use in pregnancy have not been conclusive due to lack of compliance to NRT. Cessation of smoking has not proved successful at used doses of NRT in randomized studies. The Cochrane systematic review by Clair et al. 31 concluded that studies with higher doses of nicotine were needed to study safety. The metabolism of nicotine is elevated in pregnancy, which may partly explain why smoke cessation in pregnancy is difficult to obtain with NRT. 32 The widespread use of snuff in Sweden, also in pregnancy, provides a unique opportunity to specifically study the effect of nicotine separated from the effect from other combustion products in smoking.
There are also some limitations of the study. First, the diagnoses were collected from the Cause of Death Register, without further validation through the medical records. However, all infant deaths are thoroughly investigated in Sweden and the Cause of Death Register has been validated. 18 Nevertheless, crime scene investigations are not routinely performed in Sweden following SIDS/SUID 17 , 33 and potentially cases of accidental and strangulation in bed could have been misclassified as SIDS in the current study. 33 Second, the self-reported tobacco information was not validated by cotinine levels. However, self-reported smoking and snuff use in pregnancy has been proved a valid proxy for nicotine levels in blood. 34 , 35 In addition, we lacked information on the number of snuff pouches consumed daily. However, a previous validation study, based on a sub-cohort of the women included in the MBR register, showed that the mean nicotine dose of women who continued to use snuff during pregnancy was 46 mg/day in early pregnancy (corresponds to 5–6 pouches/daily) and 36 mg/day (corresponds to 3–4 pouches daily) among those who quit in early pregnancy. 35 The validation study also showed that women using NRT in pregnancy continued to consume snuff. Among the women who stopped using snuff, none was using NRT. 35 This is in agreement with other studies on NRT use in pregnancy and smoke cessation lacking strong evidence for successful smoke cessation with NRT use in pregnancy. 31 , 36 Third, we had information on tobacco use 3 months before pregnancy and at the antenatal booking, but we lack information on when in that time span the women stopped their tobacco use. Finally, we cannot make definitive causal conclusions, given the lack of information on important confounding factors, including alcohol and substance use, and prenatal and postnatal exposure to environmental tobacco smoke. Missing mandatory education, as well as being a teenage mother, was highly associated with both tobacco use in pregnancy, especially heavy smoking, and risk of postnatal death and SIDS. It is not possible to disentangle the risk of smoking in pregnancy from other possible neonatal health hazards in this group of women. Sibling comparisons or other family design analyses may have helped account for bias due to unmeasured and residual confounding shared by siblings but were not possible to conduct due to limited power. 37
The adverse effects of nicotine on pregnancy and the developing fetus are often neglected in discussions of smokeless tobacco or ENDS as means of smoke cessation and harm reduction. 38 In the US, e-cigarettes are now more popular than conventional cigarettes among young adults. 7 With growing knowledge about the detrimental effects of non-combustible nicotine in the form of snuff, caution should be recommended regarding use of other sources of nicotine in pregnancy as well. No safe level of prenatal nicotine has been established. The use of new tobacco products and other nicotine sources place high demands on society and health care professionals regarding information and preventive strategies.
Snuff use and smoking in pregnancy were associated with increased risks of post neonatal mortality, SIDS, and SUID. The risks of snuff use and moderate smoking were of similar magnitude. Cessation of smoking and snuff use before the antenatal booking was associated with lower risks of SIDS and SUID. Our findings support the hypothesis that nicotine contributes to elevated risk of SIDS. Prevention strategies should focus on all forms of nicotine use in pregnancy. Nicotine in all forms should be avoided in pregnancy.
Data availability
Restrictions apply to the availability of these data. Data were obtained from the Swedish National Board of Health and Welfare and are available from the authors with the permission of the Swedish National Board of Health and Welfare.
Kinney, H. C. & Thach, B. T. The sudden infant death syndrome. N. Engl. J. Med. 361 , 795–805 (2009).
Article CAS PubMed PubMed Central Google Scholar
Cnattingius, S. The epidemiology of smoking during pregnancy: smoking prevalence, maternal characteristics, and pregnancy outcomes. Nicotine Tob. Res. 6 (Suppl 2), S125–S140 (2004).
Article PubMed Google Scholar
American Academy of Pediatrics Task Force on Sudden Infant Death Syndrome. The changing concept of sudden infant death syndrome: diagnostic coding shifts, controversies regarding the sleeping environment, and new variables to consider in reducing risk. Pediatrics 116 , 1245–1255 (2005).
Article Google Scholar
Moon, R. Y. & Task Force on Sudden Infant Death Syndrome. SIDS and other sleep-related infant deaths: evidence base for 2016 updated recommendations for a safe infant sleeping environment. Pediatrics 138 , e20162940 (2016).
Hafstrom, O., Milerad, J., Sandberg, K. L. & Sundell, H. W. Cardiorespiratory effects of nicotine exposure during development. Respir. Physiol. Neurobiol. 149 , 325–341 (2005).
The National Board of Health and Welfare, Sweden Pregnancies, Deliveries and Newborn Infants:The Swedish Medical Birth Register 1973–2017 (The National Board of Health and Welfare; 2019).
CDC. E-Cigarette Use Among Youth and Young Adults. A Report of the Surgeon General (Centers for Disease Control and Prevention, 2016).
Rutqvist, L. E., Curvall, M., Hassler, T., Ringberger, T. & Wahlberg, I. Swedish snus and the GothiaTek(R) standard. Harm Reduct. J. 8 , 11 (2011).
Article PubMed PubMed Central Google Scholar
Public Health Agency of Sweden. Hälsa på lika vilkor. https://www.folkhalsomyndigheten.se/folkhalsorapportering-statistik/om-vara-datainsamlingar/nationella-folkhalsoenkaten (2021).
Calder, R. et al. Vaping in pregnancy: a systematic review. Nicotine Tob. Res. 23 , 1451–1458 (2021).
DeVito, E. E. et al. Electronic nicotine delivery systems (ENDS) use and pregnancy II: Perinatal outcomes following ENDS use during pregnancy. Curr. Addict. Rep. 8 , 366–379 (2021).
Gunnerbeck, A., Wikstrom, A. K., Bonamy, A. K., Wickstrom, R. & Cnattingius, S. Relationship of maternal snuff use and cigarette smoking with neonatal apnea. Pediatrics 128 , 503–509 (2011).
Dahlin, S., Gunnerbeck, A., Wikstrom, A. K., Cnattingius, S. & Edstedt Bonamy, A. K. Maternal tobacco use and extremely premature birth - a population-based cohort study. BJOG 123 , 1938–1946 (2016).
Article CAS PubMed Google Scholar
Baba, S., Wikstrom, A. K., Stephansson, O. & Cnattingius, S. Influence of snuff and smoking habits in early pregnancy on risks for stillbirth and early neonatal mortality. Nicotine Tob. Res. 16 , 78–83 (2014).
Nordenstam, F. et al. Prenatal exposure to snus alters heart rate variability in the infant. Nicotine Tob. Res. 19 , 797–803 (2017).
Ludvigsson, J. F., Svedberg, P., Olen, O., Bruze, G. & Neovius, M. The longitudinal integrated database for health insurance and labour market studies (LISA) and its use in medical research. Eur. J. Epidemiol. 34 , 423–437 (2019).
The National Board of Health and Welfare, Sweden. Plötslig Oväntad död hos Spädbarn (The National Board of Health and Welfare, 2016).
Brooke, H. L. et al. The Swedish cause of death register. Eur. J. Epidemiol. 32 , 765–773 (2017).
Friedmann, I., Dahdouh, E. M., Kugler, P., Mimran, G. & Balayla, J. Maternal and obstetrical predictors of sudden infant death syndrome (SIDS). J. Matern. Fetal Neonatal Med. 30 , 2315–2323 (2017).
Anderson, T. M. et al. Maternal smoking before and during pregnancy and the risk of sudden unexpected infant death. Pediatrics 143 , e20183325 (2019).
MacDorman, M. F., Cnattingius, S., Hoffman, H. J., Kramer, M. S. & Haglund, B. Sudden infant death syndrome and smoking in the United States and Sweden. Am. J. Epidemiol. 146 , 249–257 (1997).
Mitchell, E. A. et al. Risk factors for sudden infant death syndrome following the prevention campaign in New Zealand: a prospective study. Pediatrics 100 , 835–840 (1997).
Slotkin, T. A. Fetal nicotine or cocaine exposure: which one is worse? J. Pharmacol. Exp. Ther. 285 , 931–945 (1998).
CAS PubMed Google Scholar
Overland, S., Tjora, T., Hetland, J. & Aaro, L. E. Associations between adolescent socioeducational status and use of snus and smoking. Tob. Control 19 , 291–296 (2010).
Baba, S., Wikstrom, A. K., Stephansson, O. & Cnattingius, S. Changes in snuff and smoking habits in Swedish pregnant women and risk for small for gestational age births. BJOG 120 , 456–462 (2013).
Alm, B. et al. A case-control study of smoking and sudden infant death syndrome in the Scandinavian countries, 1992 to 1995. The Nordic Epidemiological SIDS Study. Arch. Dis. Child. 78 , 329–334 (1998).
Trachtenberg, F. L., Haas, E. A., Kinney, H. C., Stanley, C. & Krous, H. F. Risk factor changes for sudden infant death syndrome after initiation of Back-to-Sleep campaign. Pediatrics 129 , 630–638 (2012).
Moon, R. Y., Horne, R. S. & Hauck, F. R. Sudden infant death syndrome. Lancet 370 , 1578–1587 (2007).
Benowitz, N. L. Systemic absorption and effects of nicotine from smokeless tobacco. Adv. Dent. Res. 11 , 336–341 (1997).
Benowitz, N. L., Zevin, S. & Jacob, P. 3rd Sources of variability in nicotine and cotinine levels with use of nicotine nasal spray, transdermal nicotine, and cigarette smoking. Br. J. Clin. Pharmacol. 43 , 259–267 (1997).
Claire, R. et al. Pharmacological interventions for promoting smoking cessation during pregnancy. Cochrane Database Syst. Rev. 3 , CD010078 (2020).
PubMed Google Scholar
Benowitz, N. L. & Jacob, P. 3rd Individual differences in nicotine kinetics and metabolism in humans. NIDA Res. Monogr. 173 , 48–64 (1997).
Mollborg, P., Wennergren, G., Almqvist, P. & Alm, B. Bed sharing is more common in sudden infant death syndrome than in explained sudden unexpected deaths in infancy. Acta Paediatr. 104 , 777–783 (2015).
Mattsson, K. et al. Cotinine validation of self-reported smoking during pregnancy in the Swedish Medical Birth Register. Nicotine Tob. Res. 18 , 79–83 (2016).
Gunnerbeck, A., Raaschou, P., Cnattingius, S., Edstedt Bonamy, A. K. & Wickstrom, R. Maternal snuff use and cotinine in late pregnancy-a validation study. Acta Obstet. Gynecol. Scand. 97 , 1373–1380 (2018).
Coleman, T. et al. A randomized trial of nicotine-replacement therapy patches in pregnancy. N. Engl. J. Med. 366 , 808–818 (2012).
D’Onofrio, B. M., Lahey, B. B., Turkheimer, E. & Lichtenstein, P. Critical need for family-based, quasi-experimental designs in integrating genetic and social science research. Am. J. Public Health 103 (Suppl 1), S46–S55 (2013).
England, L. J. et al. Developmental toxicity of nicotine: a transdisciplinary synthesis and implications for emerging tobacco products. Neurosci. Biobehav. Rev. 72 , 176–189 (2017).
Download references
Acknowledgements
We thank Aki Tuuliainen for excellent data management and technical support.
This work was supported by the Swedish Research Council (project grant 2018-02640), grants provided by the Stockholm County Council (ALF-projects), the Swedish Heart-Lung Foundation (project grant 20210416), and Stiftelsen Frimurare Barnhuset i Stockholm. A.G. and S.R. were supported by Region Stockholm (clinical post doctoral appointments). Open access funding provided by Karolinska Institute.
Author information
Authors and affiliations.
Department of Medical Epidemiology and Biostatistics, Karolinska Institutet, Solna, Sweden
Anna Gunnerbeck, Cecilia Lundholm, Samuel Rhedin, Brian M. D’Onofrio & Catarina Almqvist
Neuropediatric Unit, Department of Women’s and Children’s Health, Karolinska Institutet, Solna, Sweden
Anna Gunnerbeck
Sach’s Children and Youth Hospital, Stockholm, Sweden
Samuel Rhedin
Clinical Epidemiology Division, Department of Medicine Solna, Karolinska Institutet, Solna, Sweden
Ayoub Mitha
Université de Paris, Epidemiology and Statistics Research Center/CRESS, INSERM (U1153 - Obstetrical, Perinatal and Pediatric Epidemiology Research Team (EPOPé)), INRA, Hôpital Tenon, Bâtiment Recherche, Paris, France
School of Public Health (Shenzhen), Sun Yat-sen University, Guangzhou, China
Ruoqing Chen
Institute of Environmental Medicine, Karolinska Institutet, Solna, Sweden
Department of Psychological and Brain Sciences, Indiana University, Bloomington, IN, USA
Brian M. D’Onofrio
Pediatric Allergy and Pulmonology Unit, Astrid Lindgren Children’s Hospital, Karolinska University Hospital, Solna, Sweden
Catarina Almqvist
You can also search for this author in PubMed Google Scholar
Contributions
A.G. conceptualized and designed the study, carried out the initial analyses, drafted the initial manuscript, and reviewed and revised the manuscript. C.L. contributed with statistical expertise and critically reviewed the manuscript. A.M., R.C., S.R., and B.M.D. critically reviewed the manuscript for important intellectual content. C.A. conceptualized and designed the study, coordinated and supervised analyses, and critically reviewed the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Correspondence to Anna Gunnerbeck .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Ethics approval and consent to participate
The study was approved by the Regional Ethical Review Board in Stockholm, Sweden, Dnr 2018/1697-31/1, Dnr 2013/862-31/5. Patient consent was not required. The data were aggregated and anonymized.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplemental table 1, rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Gunnerbeck, A., Lundholm, C., Rhedin, S. et al. Association of maternal snuff use and smoking with Sudden Infant Death Syndrome: a national register study. Pediatr Res 94 , 811–819 (2023). https://doi.org/10.1038/s41390-022-02463-4
Download citation
Received : 09 June 2022
Revised : 03 November 2022
Accepted : 21 December 2022
Published : 09 February 2023
Issue Date : August 2023
DOI : https://doi.org/10.1038/s41390-022-02463-4
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
Vaping during pregnancy: a systematic review of health outcomes.
- Michael Ussher
- Joy Fleming
- Leonie Brose
BMC Pregnancy and Childbirth (2024)
Separation and identification of snuff constituents by using GC–MS and ICP-OES as well as health risk assessment of some existing heavy metals
- Safoura Javan
- Mahboube Eskandari
- Amir M. Ramezani
Environmental Monitoring and Assessment (2023)
Ventilatory response to added dead space in infants exposed to second-hand smoke in pregnancy
- Allan Jenkinson
- Nadja Bednarczuk
- Anne Greenough
European Journal of Pediatrics (2023)
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
- Research article
- Open access
- Published: 13 March 2015
Maternal smoking and the risk of still birth: systematic review and meta-analysis
- Takawira C Marufu 1 ,
- Anand Ahankari 1 , 2 ,
- Tim Coleman 3 &
- Sarah Lewis 1
BMC Public Health volume 15 , Article number: 239 ( 2015 ) Cite this article
16k Accesses
141 Citations
44 Altmetric
Metrics details
Smoking in pregnancy is known to be associated with a range of adverse pregnancy outcomes, yet there is a high prevalence of smoking among pregnant women in many countries, and it remains a major public health concern. We have conducted a systematic review and meta-analysis to provide contemporary estimates of the association between maternal smoking in pregnancy and the risk of stillbirth.
We searched four databases namely MEDLINE, EMBASE, Psych Info and Web of Science for all relevant original studies published until 31 st December 2012. We included observational studies that measured the association between maternal smoking during pregnancy and the risk of stillbirth.
1766 studies were screened for title analysis, of which 34 papers (21 cohorts, 8 case controls and 5 cross sectional studies) met the inclusion criteria. In meta-analysis smoking during pregnancy was significantly associated with a 47% increase in the odds of stillbirth (OR 1.47, 95% CI 1.37, 1.57, p < 0.0001). In subgroup analysis, smoking 1-9 cig/day and ≥10 cig/day was associated with an 9% and 52% increase in the odds of stillbirth respectively. Subsequently, studies defining stillbirth at ≥ 20 weeks demonstrated a 43% increase in odds for smoking mothers compared to mothers who do not smoke, (OR 1.43, 95% CI 1.32, 1.54, p < 0.0001), whereas studies with stillbirth defined at ≥ 24 weeks and ≥ 28 weeks showed 58% and 33% increase in the odds of stillbirth respectively.
Our review confirms a dose-response effect of maternal smoking in pregnancy on risk of stillbirth. To minimise the risk of stillbirth, reducing current smoking prevalence in pregnancy should continue to be a key public health high priority.
Peer Review reports
Smoking in pregnancy is a major public health problem in the developed countries [ 1 ]. The World Health Organisation (WHO) predicts that this will reach ‘epidemic’ proportions in developing countries in the near future [ 1 ]. Within the current challenging economic climate in many countries, smoking in pregnancy imposes a significant burden on population health and resources, and is associated with a range of poor outcomes for both mother and child, such as ectopic pregnancy, miscarriage, placental abruption, preterm birth and low birth weight [ 2 , 3 ]. The harmful effects of tobacco smoke exposure in pregnancy can be avoided [ 4 ] and it is one of the most prevalent modifiable risk factors for adverse pregnancy outcomes [ 5 , 6 ].
In many developed countries, the rates of smoking in pregnancy have been declining over recent decades [ 7 ] with current prevalence estimates between 10% – 19% [ 8 ], and data from the UK suggesting that one out of eight women smoke throughout the pregnancy [ 9 ]. Moreover, this decline has not been consistent across all social classes; lower rates of decline have been noted across less advantaged socioeconomic groups [ 10 ]. Smoking in pregnancy in developed countries tends to be higher among women who have low income and poor education [ 11 ].
Stillbirth rates widely vary across low, middle and high-income countries [ 12 - 14 ]. The lowest rates have been observed in Finland with 2.0 per 1000 live births and in the developing world rates are much higher at more than 40 per 1000 live births in countries like Nigeria and Pakistan [ 6 ]. Two previous systematic reviews and meta-analyses have estimated the size of effect of smoking during pregnancy and the risk of stillbirth. A report by the United States Surgeon General [ 15 ] showed a relative risk of stillbirth of 1.2-1.8 within smokers versus non-smokers. The analysis was based on three studies [ 16 - 18 ] conducted in two countries, the United States and Sweden. Recent stillbirth statistics by Flenady et al [ 8 ] was based on a systematic review from high-income countries; and four studies were included in the meta-analysis of the association between any smoking in pregnancy and the risk of stillbirth yielding an odds ratio of 1.36 (95% CI 1.27, 1.46). Results from these published reviews were limited to literature from developed countries; yet stillbirth rates are much higher in developing countries, and it is therefore imperative to conduct a comprehensive review, which reflects the impact of smoking in pregnancy on stillbirth. We have conducted a systematic review and meta-analysis to provide contemporary estimates of the association between maternal smoking in pregnancy and the risk of stillbirth.
A detailed electronic search was performed through four databases namely MEDLINE, EMBASE, Psych Info and Web of Science. All relevant published studies with sufficient data on maternal cigarette or tobacco smoking during pregnancy with the outcome of stillbirth were included. Due to the nature of the research objective, there were no randomised control trials (RCTs) identified, and observational studies (such as cohort design, case control studies and cross sectional surveys) were considered using the standard guidelines of Meta-analysis of Observational Studies in Epidemiology (MOOSE) [ 19 ]. All relevant studies published in the English language, up to 31 st December 2012 were included in the review. Case reports, non-English publications and those, which only involved passive or environmental smoke information, were excluded. We carefully considered studies where the study populations were similar to avoid duplication of cases; where studies were based on exactly the same population during the same time period, but there were differences in sample sizes, we selected the results from the publication with the larger sample size for inclusion in the meta-analysis, though those excluded from quantitative analysis were included in a narrative synthesis.
Search strategy
We developed search terms based on medical subject headings (MeSH), free text words and words in the title or abstract. We combined search terms of exposure (smoking during pregnancy) with the outcome of the study (stillbirth). The MeSH terms used included combinations of the terms: stillbirth, maternal smoking, and pregnancy, also applied with special characters ($, *), wherever required. The study protocol was agreed within the team, however it was not published prior to commencement of the review. We also checked the reference list of all identified papers and the most recent similar reviews for any additional studies. A complete online search strategy along with search terms is attached (Additional file 1 ).
Study selection and outcome definition
The electronic search was performed with a three-stage approach; first titles were screened by TM and AA, then abstracts were screened by two authors independently (TM and SL). For studies, which appeared to be eligible, full texts were obtained and reviewed independently by two authors who further performed data extraction independently on pre-piloted forms, and discrepancies were resolved by consensus. We also obtained full texts for those studies where a decision could not be made based on title and abstract. Data extraction forms included followings; study design, exposure validation, definition of exposure (smoking) and outcome (stillbirth), confounders adjustments, sample size and study location.
Since stillbirth is defined differently in different countries and different studies, stillbirth for this review was defined as fetal loss or death at 20 weeks gestation and above [ 20 ]; this included both early (20-28 weeks gestation) and late (after 28 weeks) stillbirth enabling inclusion of a wider range of international studies. Sensitivity analysis was done using alternative definitions of stillbirth:
Stillbirth at ≥ 20 weeks of gestation
Stillbirth at ≥ 24 weeks of gestation
Stillbirth at ≥ 28 weeks of gestation
Where both unadjusted and adjusted measures were reported, the latter were extracted. Quality assessment was based on a Newcastle Ottawa Scale (NOS). Cohort and case control studies were awarded up to a maximum quality score of nine and cross sectional studies were given up to a maximum score of seven [ 21 ]. A definition of ‘high quality’, was given to cohort and case control studies with a score of 7 or above or cross-sectional studies with a score of 5 or above; the rest were deemed ‘low quality’ [ 21 ]. Each quality assessment (NOS) was conducted by at least by two authors independently and then findings were verified.
Statistical analysis
Meta-analysis was performed using Rev-Man 5.3 [ 22 ] with a random effects model. The I 2 statistic was applied to calculate heterogeneity (I 2 more than 75% was considered high heterogeneity, more than 50% moderate and 25% was considered as low heterogeneity) [ 22 ]. The odds ratio for the overall effect of smoking in pregnancy from each study was used, which was the main outcome reported in included literature. The ratio was calculated from the available data, wherever feasible. Studies included in the meta-analysis were further considered for subgroup analysis based on study quality, definition of stillbirth and cigarette consumption (1-9 cig/day and ≥10 cig/day). The funnel plot method was used to assess publication bias. Studies without statistical data presentation, and those where cases were potentially overlapping between publications, were considered in a narrative synthesis.
The initial database search produced 2,934 papers. We found 1,168 duplicate studies; meaning the same study was obtained from more than one of the four databases used in the review. After removal of duplicates, 1,766 study titles were screened, 94 were found eligible for abstract screening and 35 papers were considered for full text analysis. Out of 35 full texts sought, 34 were obtained within the available research timelines, and of these, 29 were included in the systematic review [ 12 - 14 , 16 - 18 , 23 - 45 ]. Eight full text studies, which did not have sufficient qualitative or quantitative data relevant to this review, were excluded [ 46 - 53 ]. An additional five relevant studies [ 54 - 58 ] were identified from references listed in the full text eligible studies. Details of the electronic search are explained in Figure 1 . Out of the 34 included studies, 24 were included in the meta-analysis [ 12 - 14 , 17 , 23 , 24 , 26 - 29 , 31 - 41 , 54 - 56 ] and the other remaining ten in the qualitative synthesis [ 16 , 18 , 25 , 30 , 42 - 45 , 57 , 58 ]. One of the studies, [ 23 ] used two separate study designs, case control and bidirectional crossover methodology; therefore the study was considered in the meta-analysis separately as two observations resulting in 25 studies included in the quantitative synthesis. Two studies references [ 30 , 31 ] were derived from the same dataset (Missouri, USA) conducted in the same timeframe (1978-1997). Study [ 31 ] with a higher sample size was included in the meta-analysis and the other [ 30 ] was considered in the narrative synthesis. Four studies [ 16 , 17 , 25 , 29 ] used the same study population (Swedish National registry data) with differences in sample size and study methodology. Studies [ 17 , 25 ] had the same time frame, with study [ 17 ] having a higher sample size. Studies [ 16 , 17 , 29 ] had overlapping but not the exact dataset time frames. Study [ 16 ] time period (1983 – 1985) was exceeded by study 17 (1983-1989) which also had a higher sample size. Study [ 29 ] used the data for 1984 and 1991 for comparative purposes. To avoid having any woman’s data being counted twice in the meta-analysis, only the estimates obtained for 1991 data [ 29 ] were used in the meta-analysis. Two studies [ 17 , 29 ] were included in the meta-analysis whilst the other two studies [ 16 , 25 ] were included in the narrative synthesis only.
Flow diagram of included and excluded studies.
Included studies are further detailed in Table 1 .
Out of the 34 eligible studies (Table 1 ), eight were case control studies [ 23 - 28 , 54 , 55 ], five cross sectional [ 16 , 18 , 40 , 41 , 45 ] and twenty-one were cohort studies [ 12 - 14 , 17 , 29 - 39 , 42 - 44 , 56 - 58 ]. Fourteen were conducted in Europe, [ 13 , 14 , 16 , 17 , 25 , 27 , 29 , 32 , 36 - 39 , 43 , 54 ], four in Australia [ 34 , 41 , 56 , 58 ], fourteen in North America [ 12 , 18 , 23 , 26 , 28 , 30 , 31 , 33 , 35 , 42 , 44 , 45 , 55 , 57 ], one in Asia [ 40 ] and one in South America [ 24 ]. The largest sample size observed was 7,792,990 [ 45 ] with the smallest being 202 [ 25 ]. Ten studies [ 13 , 16 , 25 , 26 , 30 - 32 , 35 , 54 , 55 ] did not present their results with odds ratios but had sufficient data to calculate the ratio using STATA 12, which was further used in the meta-analysis. Seven studies [ 18 , 42 - 45 , 57 , 58 ] did not present estimates or sufficient data to calculate an odds ratio; therefore they were included in the narrative synthesis. Based on NOS scale, overall study quality was moderately satisfactory. Ten cohort studies [ 12 , 14 , 17 , 32 , 35 - 39 , 42 ] and four case control studies [ 23 , 24 , 54 , 55 ] were considered of high quality with seven or more points, and only two cross sectional studies [ 16 , 45 ] were of high quality having five or more points. Scores ranged from 2 to 9 and the median score was 6.
The type of exposure
Eleven studies clearly defined the level of exposure (smoking) [ 14 , 16 , 17 , 25 , 29 , 31 , 33 , 34 , 38 , 54 , 56 ]. Most of the studies used categories of 1-9cig/day and ≥10 cig/day to categorise participants according to the number of cigarettes or packs consumed daily, but the Robson study [ 41 ] used slightly different categories. Seventeen studies did not define smoking according to level of cigarette consumption [ 12 , 13 , 24 , 26 - 28 , 30 , 32 , 35 , 36 , 40 , 41 , 43 , 45 , 55 , 57 , 58 ] but according to the smoking status (yes/no). Fifteen studies collected exposure information during pregnancy [ 12 - 14 , 16 , 17 , 27 , 29 , 30 , 32 , 33 , 38 , 42 , 43 , 54 , 56 ] and the rest of the studies [ 18 , 23 - 26 , 31 , 34 , 35 , 39 - 41 , 44 , 45 , 55 , 57 , 58 ] obtained the information after delivery. None of the studies reported biochemical validation of smoking status such as salivary cotinine assessments.
Data collection on outcome
The definition of stillbirth varied across studies. Twenty-one out of thirty-four studies included stillbirth from early gestational age ≥ 20 weeks [ 18 , 23 , 30 , 31 , 33 , 34 , 36 , 37 , 41 - 43 , 45 , 56 - 58 ], ≥22 weeks [ 54 ], ≥23 weeks [ 35 ], and four studies used birth weight to estimate the gestational week of stillbirth (Birth weight ≥ 500 g, [ 24 , 26 , 28 , 44 ]. The rest of the studies used late gestational ages as the cut off values for stillbirth, ≥24 weeks in four studies [ 12 , 13 , 27 , 32 ], ≥28 weeks in nine studies [ 14 , 16 , 17 , 25 , 29 , 38 - 40 , 55 ]. In all studies, outcome data was either obtained from medical or clinical records, medical data sets or birth registry records.
Fourteen studies included in the quantitative synthesis adjusted for one or more confounders (Table 1 ). Maternal age was the most common adjusted factor. Five studies adjusted for socioeconomic status (SES) including education [ 13 , 14 , 37 , 38 , 40 ]. Other factors that were adjusted for include parity, BMI, ethnicity/race, infant sex, perinatal care and marital status, alcohol, caffeine intake and cohabitation. Four studies [ 24 , 27 , 36 , 39 ] did not adjust for any confounders and six studies [ 26 , 31 , 32 , 35 , 54 , 55 ] had OR calculated therefore considered as not adjusted for any confounders. Factors adjusted in cohort studies varied widely. Several studies [ 12 , 17 , 28 , 34 , 37 , 41 , 56 ] considered one or more medical conditions such as pregnancy complications, diabetes mellitus and hypertension as possible confounders.
Meta-analysis of maternal smoking and risk of stillbirth
In meta-analysis of all 25 studies (Figure 2 ), smoking during pregnancy was associated with a 47% increase in the odds of stillbirth (OR 1.47, 95% CI 1.37, 1.57, p < 0.0001) with an overall moderate heterogeneity (I 2 = 79%). There was no significant difference in the size of this estimate between study designs (p = 0.11); the odds of stillbirth were increased by 34% in relation to smoking in pregnancy in case-control studies (OR 1.34, 95% CI 1.23, 1.45, p < 0.0001, 8 studies), 49% in cohort studies (OR 1.49, 95% CI 1.35, 1.64, p < 0.0001, 15 studies) and 62% in cross-sectional studies (OR 1.62, 95% CI 1.31, 2.00, p < 0.0001, 2 studies). Results were more heterogeneous for cohort studies than other study types.
Maternal smoking in pregnancy and the risk of stillbirth.
Subgroup analysis by study quality
Results from high quality score studies showed that women who smoke during pregnancy, are 41% more likely to have a stillbirth compared to women who do not smoke during pregnancy (OR 1.41, 95% CI 1.28, 1.55, p <0.0001, 14 studies). Low quality score studies showed that women who smoke during pregnancy were at 49% increased odds of stillbirth compared to women who do not smoke during pregnancy (OR 1.49, 95% CI 1.33, 1.67, p <0.0001, 11 studies). There was no significant difference between the two subgroups (p = 0.44).
Subgroup analysis by categories of cigarette consumption
Seven studies (14, 26 a-b,31,34,45,48) were included in this subgroup analysis, with consumption categorised as 1-9 cig/day and ≥10 cig/day (Figure 3 ). One study [ 29 ] presented OR values for the ≥10 cig/day category only. A study by Robson et al [ 41 ] used slightly different categories; 1-10 cig/day and >10 cig/day. This study was included in this analysis with odds ratios for the 1-10 cig/day, and > 10 cig/day categories used for the 1-9 cig/day and ≥ 10 cig/day groups respectively. Further two studies by Salihu et al (2008a) [ 23 ] and Salihu et al (2008b) [ 23 ] had a classification of 1-9 cig/day, 10-19 cig/day and ≥ 20 cig/day. There was no sufficient raw data to calculate the OR for ≥10 cig/day. Therefore these studies have been included in this analysis conservatively using the ORs from the 10-19 cig/day categories (which is a lower value than the ≥ 20 cig/day value) for the ≥ 10 cig/day subgroup. Most of the studies included in this subgroup analysis (4 out of 7) used this categorisation (1-9 cig/day and ≥10 cig/day), thus the same was applied in the subgroup analysis. Meta-analysis showed that smoking 1-9 cig/day was associated with an 9% increased odds of having a stillbirth compared to women who do not smoke in pregnancy (OR 1.09, 95% CI 1.09, 1.24, p = 0.55, 6 studies), whilst smoking ≥10 cig/day was associated with a 52% increase in odds of stillbirth (OR 1.52, 95% CI 1.30, 1.78, p < 0.0001, 7studies); the results for these two subgroups differed significantly (p = 0.001).
Stillbirth risk according to the amount of daily cigarette consumption.
Subgroup analysis by gestation of stillbirth
Twenty-two studies were included in this subgroup analysis (Figure 4 ). The meta-analysis of studies defining stillbirth at ≥ 20 weeks demonstrated a 43% increase in odds for smoking mothers compared to mothers who do not smoke, (OR 1.43, 95% CI 1.32, 1.54, p < 0.0001, 11 studies), whereas studies with stillbirth defined at ≥ 24 weeks showed 58% increase in odds (OR 1.58, 95% CI 1.21, 1.2.06, p < 0.0003, 4 studies) and in pooled estimates of studies with stillbirth defined at ≥ 28 weeks, the odds was increased to 33% (OR 1.33, 95% CI 1.18, 1.49, p < 0.02, 7 studies). There was no significant difference in the odds ratios for these different subgroups (p = 0.39).
Maternal smoking and the risk of stillbirth according to the gestation period.
Subgroup analysis by study location
Only two studies were conducted in developing countries [ 24 , 40 ]. Both studies support evidence from research conducted in developed countries concluding that women who smoke during pregnancy are at a risk of stillbirth. A study [ 40 ] demonstrated a very marginal difference showing those women who smoke during pregnancy are at least 40% at risk of experiencing a stillbirth (OR 1.40, 95% CI 1.0, 1.96), similar to the pooled odds ratio of studies conducted in North America (OR 1.39, 95% CI 1.29, 1.50) P value < 0.0001, 9 studies) and Europe (OR 1.55, 95% CI 1.36, 1.78, P value < 0.0001,11 studies).
Narrative synthesis
Seven studies [ 18 , 42 - 45 , 57 , 58 ] presented their results in other than odds ratio and there was no sufficient data to calculate the odds ratio, and three studies [ 16 , 25 , 30 ] were derived from same study population as [ 17 , 31 ] respectively. Six of the studies [ 16 , 18 , 30 , 42 , 45 , 57 ] observed an association between maternal smoking in pregnancy and the risk of stillbirth but the other four [ 25 , 43 , 44 , 58 ] observed no significant differences. Two studies [ 16 , 42 ] observed a dose related response with high risk ratio of stillbirth in mothers who smoke ≥10 cig/day compared to 1-9 cig/day and those who smoke ≥1 pack/day in contrast to <1 pack/day respectively. Three studies (51, 35 54) showed the risk of stillbirth to be higher in young mothers <15 years old (HR 3.3, 95% CI 1.4, 7.8) and also in older mothers (≥35 years, HR 3.2, 95% CI 2.2, 4.5) (≥40 years, HR 2.71, 95% CI 1.88, 3.91) respectively. A study from Sweden suggested that placental abruption is likely to be common in smokers [ 25 ].
Publication bias
The publication bias was assessed visually using a funnel plot developed by Rev Man 5.3 [ 22 ]. The plot was symmetrical indicating a low risk of publication bias (Additional file 2 ).
This study provides a comprehensive review of the current evidence and estimates of the effect of maternal smoking on the risk of stillbirth. It suggests, women who smoke during pregnancy have a 47% increased risk of stillbirth and that the risk of stillbirth is more at higher levels of cigarette consumption (Table 2 ). This effect does not vary according to the gestation at which still birth occurs, however, comparisons between studies would be easier if future work adopted the definition of stillbirth recommended by the WHO [ 20 ].
Strengths and limitations
We implemented a comprehensive search strategy with strict adherence to the protocol, and results were presented in accordance with MOOSE guidelines [ 19 ]. Search strategy, data extraction, analysis and quality assessment was performed independently by authors and findings were confirmed within the team. There was no evidence of publication bias. However, the review has some limitations. We could not obtain one study within available timelines [ 59 ], and due to limited data presentation, seven studies [ 18 , 42 - 45 , 57 , 58 ] were considered in the narrative synthesis. Secondly, only English language studies were considered mainly for practical purposes and the available timeframe.
Heterogeneity was explored using a variety of subgroup analyses; a high level of heterogeneity was found amongst cohort studies and the pooled estimate derived from these should be treated with some caution. This could be partially attributable to the different factors controlled for in the cohort studies as explained previously. The overall heterogeneity (I 2 = 77%, Figure 2 ) was higher mainly due to cohort studies, however in case of case control studies (I 2 = 20%) and cross-sectional studies (I 2 = 0%), very low levels of heterogeneity was observed. However, this estimate was consistent with synthesised estimates derived from studies with other designs and this consistency suggests it is likely to be valid. It appears that the effect could differ by age [ 24 , 30 , 38 , 45 , 57 ], but data on maternal age was not available in included studies. Study quality was found to be satisfactory.
The most recent systematic review by Flenady et al [ 8 ] on maternal smoking and the risk of stillbirth, focusing on high-income countries, reported a 36% increase in the odds of stillbirth (OR 1.36). The review considered cohort and case control studies published between 1998 and 2009 and excluded those which did not control for confounding factors, hospital based studies and took a slightly different approach to dealing with duplicate studies. Acknowledging the difference in the approach, our study is in agreement with the results from Flenady’s et al [ 8 ] review, and showed a 46% increased risk of stillbirth (OR 1.46 95% CI 1.36, 1.55). We included 25 studies in our main meta-analysis, conducted in both developing and developed countries making our findings generalisable. Our review has also analysed the available data according to the definition of stillbirth in gestational weeks, number of cigarettes and study quality, which were not reported in the previous published literature. The review also provides strong evidence that the risk is higher at higher levels of cigarette consumption, indicating a dose related response relationship, outlined in some of the included studies [ 15 , 38 , 42 ].
The consistency in the size of the effect observed in this study throughout different subgroup analyses and across different study designs, each with their own biases and strengths and limitations, as well as the consistency across studies of differing quality, suggests that this is a true estimate of the size of effect. Further support is obtained from studies where mothers change in smoking behaviour from one pregnancy to another. Hogberg and Cnattingius [ 14 ] suggested that mothers who quit smoking from first to second pregnancy reduced their risk of stillbirth to the same level as non-smokers in both pregnancies, while those who smoked in both pregnancies had a 35% increased risk of stillbirth (OR 1.35) compared to non-smokers.
Our results also provide evidence that the effect of maternal smoking in pregnancy on risk of stillbirth is not dependent on the definition of stillbirth or on the stage of pregnancy at which it occurs. Moreover we did not ascertain, at what point during pregnancy the effect of the exposure is occurring as most studies evaluated the exposure at one point in time only. Previous studies [ 26 , 38 ] suggested that the timing of exposure may influence the risk of stillbirth and in particular [ 38 ] suggested that those who quite early in pregnancy may have similar risk to non-smokers. Future studies of this association should measure exposure at different stages of gestation. There are important potential confounders for the effect of maternal smoking on risk of stillbirth, the most important ones are SES, maternal age, maternal weight and medical comorbidities, majority of studies have adjusted for these. Only two studies did not adjust for any confounders [ 36 , 43 ] with no further information.
The exact pathophysiology of fetal exposure to tobacco smoke is not entirely understood, however based on available evidence, possible mechanisms have been conceptualised [ 30 ]. Nicotine has been known to cause narrowing of the placental vessels [ 60 , 61 ], which coupled with reduced prostacycline synthesis [ 62 ], resulting in increased vascular resistance and consequently impairing blood supply to the fetus [ 63 ]. Carbon monoxide in tobacco smoke also reduces fetal oxygenation by forming carboxy-haemoglobin in turn interfering with oxygenation transfer [ 64 ]. The correlation between smoking and stillbirth is likely to be explained through increased prevalence of placental complications and fetal growth restriction [ 14 ]. The resultant physiological effect of these changes increases the risk of fetal morbidity (small for gestation age and preterm birth) and subsequently may lead to fetal death [ 30 ]. Therefore, there is strong biological plausibility for smoking in pregnancy causing stillbirth [ 46 , 65 ].
Recent Cochrane reviews have concluded that behavioural interventions can be effective in helping women to stop smoking in pregnancy [ 66 ] even though there remains no evidence for the effectiveness of pharmacotherapy such as NRT in pregnancy [ 67 ]. The result of our study suggests that such interventions are important to reduce the risk of stillbirth.
Our findings support that smoking greatly increases the risk of stillbirth. Smoking in pregnancy is an established cause of a range of pregnancy complications and poor pregnancy outcomes. Every opportunity must be utilised to record smoking status during pregnancy, to give advice and support, including necessary facilities to help women stop smoking. Although we have conducted a comprehensive review, most of the available evidence on the risk from smoking is from developed countries. Smoking is rapidly increasing in the developing world where stillbirth is a major problem; therefore along with focusing on research initiatives, it is important to invest efforts on educating women on the risks of smoking to their unborn child and providing smoking cessation support for pregnant women in these parts of world.
Abbreviations
New castle Ottawa scale
Socioeconomic status
World Health Organisation (WHO). Recommendations for the prevention and management of tobacco use and second- hand smoke exposure in pregnancy. Geneva: World Health Organization; 2013.
Google Scholar
Royal College of Physicians. Passive smoking and children: a report of the Tobacco Advisory Group of the Royal College of Physicians. London: Royal College of Physicians; 2013.
Royal College of Physicians. Smoking and the young. London: Royal college of Physicians; 1992.
Cnattingius S, Bergstrom R, Lipworth L, Kramer MS. Pre-pregnancy weight and the risk of adverse pregnancy outcomes. N Eng J Med. 1998;338:147–52.
Article CAS Google Scholar
Fretts RC. Etiology and prevention of stillbirth. Am J Obstet Gynecol. 2005;193:1923–35.
Article PubMed Google Scholar
Lawn JE, Blencowe H, Pattinson R, Cousens S, Kumar R, Ibiebele I, et al. Stillbirths: Where? When? Why? How to make the data count? Lancet. 2011;377:1448–63.
Cnattingius S. The epidemiology of smoking during pregnancy: smoking prevalence, maternal characteristics, and pregnancy outcomes. Nicotine Tob Res. 2004;6:S125–40.
Flenady V, Koopmans L, Middleton P, Froen JF, Smith GC, Gibbons K, et al. Major risk factors for stillbirth in high-income countries: a systematic review and meta-analysis. Lancet. 2011;377:1331–40.
NHS Information centre. The Infant Feeding Survey 2010. Available at http://www.hscic.gov.uk [Accessed January 2013].
Scollo MM, Winstanley MH. Tobacco in Australia: Facts and issues. 4th ed. Melbourne: Cancer Council Victoria; 2012. Available from http://www.TobaccoInAustralia.org.au .
Ebert LM, Fahy K. Why do women continue to smoke in pregnancy? Women Birth. 2007;20:161–8.
Wood SL, Chen S, Ross S, Sauve R. The risk of unexplained antepartum stillbirth in second pregnancies following caesarean section in the first pregnancy. BJOG. 2008;115:726–31.
Article CAS PubMed Google Scholar
Smith GCS, Shah I, White IR, Pell JP, Crossley JA, Dobbie R. Maternal and biochemical predictors of antepartum stillbirth among nulliparous women in relation to gestational age of fetal death. BJOG. 2007;114:705–14.
Hogberg L, Cnattingius S. The influence of maternal smoking habits on the risk of subsequent stillbirth: Is there a causal relation? BJOG. 2007;114:699–704.
Article CAS PubMed PubMed Central Google Scholar
United States Department of Health and Human Services (USDHHS). Health Consequences of Tobacco use among women. In: Women and smoking: a report of the surgeon general. Rockville MD: U.S: Department of Health and Human Services, Centre for Disease Control and Prevention, National Centre for Chronic Disease Prevention and Health Promotion, Office of Smoking and Health; 2001. p. 177–450.
Cnattingius S, Haglund B, Meirik O. Cigarette smoking as risk factor for late fetal and early neonatal death. BMJ. 1988;297:258–61.
Raymond EG, Cnattingius S, Kiely JL. Effects of maternal age, parity and smoking on the risk of stillbirth. BJOG. 1994;101:301–6.
Schramm WF. Smoking during pregnancy: Missouri longitudinal study. Paediatr Perinat Epidemiol. 1997;11:73–83.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 2000;283:1–5.
Article Google Scholar
Zegers-Hochschild F, Adamson GD, de Mouzon J, Ishihara O, Mansour R, Nygren K, et al. The International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) Revised Glossary on ART Terminology. Hum Reprod. 2009;24:2683–7.
Wells G, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. Newcastle-Ottawa scale (NOS) for assessing the quality of non-randomised studies in meta-analysis. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm (accessed March 10, 2012).
Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration 2011. Available from http://www.cochrane-handbook.org .
Salihu HM, Sharma PP, Getahun D, Hedaytzadeh M, Peters S, Kirby RS, et al. Prenatal tobacco use and risk of stillbirth: a case-control and bidirectional case-crossover study. Nicotine Tob Res. 2008;10:159–66.
Ferraz EM, Gray RH. A case-control study of stillbirths in Northeast Brazil. Int J Gynaecol Obstet. 1991;34:3–19.
Walles B, Tyden T, Herbst A, Ljungblad U, Rydhstrom H. Maternal health-care program and makers for late fetal death. Acta Obstet Gynecol Scand. 1994;73:773–8.
Dodds L, King WD, Fell DB, Armson BA, Allen A, Nimrod C. Stillbirth risk factors according to timing of exposure. Ann Epidemiol. 2006;16:607–13.
Efkarpidis S, Alexopoulos E, Kean L, Liu D, Fay T. Case-control study of factors associated with intrauterine fetal deaths. MedGenMed. 2004;6:53.
PubMed PubMed Central Google Scholar
Goy J, Dodds L, Rosenberg MW, King W. Health-risk behaviours: examining social disparities in the occurrence of stillbirth. Paediatr Perinat Epidemiol. 2008;22:314–20.
Ahlenius I, Thomassen P. The changing panorama of late fetal death in Sweden between 1984 and 1991. Acta Obstet Gynecol Scand. 1999;78:408–14.
Aliyu MH, Salihu HM, Wilson RE, Alio AP, Kirby RS. The risk of intrapartum stillbirth among smokers of advanced maternal age. Arch Gynecol Obstet. 2008;278:39–45.
Aliyu MH, Salihu HM, Wilson RE, Kirby RS. Prenatal smoking and risk of intrapartum stillbirth. Arch Environ Occup Health. 2007;62:87–92.
Gray R, Bonellie SR, Chalmers J, Greer I, Jarvis S, Kurinczuk JJ, et al. Contribution of smoking during pregnancy to inequalities in stillbirth and infant death in Scotland 1994-2003: retrospective population based study using hospital maternity records. BMJ. 2009;339:b3754.
Article PubMed PubMed Central Google Scholar
Miller EC, Cao HL, Wen SW, Yang Q, Lafleche J, Walker M. The risk of adverse pregnancy outcomes is increased in preeclamptic women who smoke compared with nonpreeclamptic women who do not smoke. Am J Obstet Gynecol. 2010;203:1–8.
Mohsin M, Bauman AE, Jalaludin B. The influence of antenatal and maternal factors on stillbirths and neonatal deaths in New South Wales. J Biosoc Sci. 2006;38:643–57.
Reddy UM, Laughon SK, Sun L, Troendle J, Willinger M, Zhang J. Prepregnancy risk factors for antepartum stillbirth in the United States. Obstet Gynecol. 2010;116:1119–26.
Sutan R, Campbell D, Prescott GJ, Smith WCS. The risk factors for unexplained antepartum stillbirths in Scotland, 1994 to 2003. J Perinatol. 2010;30:311–8.
Tuthill DP, Stewart AH, Coles EC, Andrews J, Cartidge PH. Maternal cigarette smoking and pregnancy outcome. Paediatr Perinat Epidemiol. 1999;13:245–53.
Wisborg K, Kesmodel U, Henriksen TB, Olsen SF, Secher NJ. Exposure to tobacco smoke in utero and the risk of stillbirth and death in the first year of life. Am J Epidemiol. 2001;154:322–7.
Winbo I, Serenius F, Dahlquist G, Kallen B. Maternal risk factors for cause-specific stillbirth and neonatal death. Acta Obstet Gynecol Scand. 2001;80:235–44.
Mishra V, Retherford RD, Smith KR. Cooking smoke and tobacco smoke as risk factors for stillbirth. Int J Environ Health Res. 2005;15:397–410.
Robson S, Cameron CA, Roberts CL. Birth outcomes for teenage women in New South Wales, 1998-2003. Aust N Z J Obstet Gynaecol. 2006;46:305–10.
Kleinman JC, Pierre MB, Madans Jr JH, Land GH, Schramm WF. The effects of maternal smoking on fetal and infant mortality. Am J Epidemiol. 1988;127:274–82.
CAS PubMed Google Scholar
Raatikainen K, Huurinainen P, Heinonen S. Smoking in early gestation or through pregnancy: a decision crucial to pregnancy outcome. Prev Med. 2007;44:59–63.
Huang DY, Usher RH, Kramer MS, Yang H, Morin L, Fretts RC. Determinants of unexplained antepartum fetal deaths. Obstet Gynecol. 2000;95:215–21.
Salihu HM, Shumpert MN, Aliyu MH, Alexander MR, Kirby RS, Alexander GR. Stillbirths and infant deaths associated with maternal smoking among mothers aged not greater than or equal to 40 years: a population study. Am J Perinatol. 2004;21:121–9.
Moga M, Preda GH. Smoking and pregnancy. J Environ Prot Ecol. 2008;9:566–73.
Bukowski R, Carpenter M, Conway D, Coustan D, Dudley DJ, Goldenberg RL, et al. Association between stillbirth and risk factors known at pregnancy confirmation. JAMA. 2011;306:2469–79.
Kunzel W, Misselwitz B. Unexpected fetal death during pregnancy – a problem of unrecognised fetal disorders during antenatal care? Eur J Obstet Gynecol Reprod Biol. 2003;110:S86–92.
Xu B, Jarvelin MR, Rantakallio P. Maternal smoking in pregnancy and sex differences in perinatal death between boys and girls. Soc Biol. 1998;45:273–7.
Kallen K. The impact of maternal smoking during pregnancy on delivery outcome. Eur J Public Health. 2001;11:329–33.
Verkerk PH, Buitendijk SE, Verloove-Vanhorick SP. Differential misclassification of alcohol and cigarette consumption by pregnancy outcome. Int J Epidemiol. 1994;23:1218–25.
Maleckiene L, Nadisauskiene R, Bergstrom S. Scio-economic demographic and obstetric risk factors for late death of unknown etiology in Luthuania: a case—referent study. Acta Obstet Gynecol Scand. 2001;80:321–5.
Beyerleinl A, Von Kriesl R, Hummel M, Lack N, Schiessl B, Giani G. Improvement in pregnancy-related outcomes in the offspring of diabetic mothers in Bavaria, Germany, during 1987–2007. Diabet Med. 2010;27:1379–84.
Froen JF, Arnestad M, Vege A, Irgens, Rognum T, Saugstad O. Comparative epidemiology of sudden infant death syndrome and sudden intrauterine unexplained death. Arch Dis Child. 2002;87:118–21.
Little RE, Weinberg CR. Risk-factors for antepartum and intrapartum still-birth. Am J Epidemiol. 1993;137:1177–89.
McCowan LM, George-Haddad M, Stacey T, Thompson JM. Fetal growth restriction and other risk factors for stillbirth in a New Zealand setting. Aust N Z J Obstet Gynaecol. 2007;47:450–6.
Aliyu MH, Salihu HM, Alio AP, Wilson RE, Charkrabarty S, Clayton HB. Prenatal smoking among adolescents and risk of fetal demise before and during labor. J Pediatr Adolesc Gynecol. 2010;23:129–35.
Bai J, Wong FW, Gyneshwar R, Stewart HC. Profile of maternal smokers and their pregnancy outcomes in south western Sydney. J Obstet Gynaecol Res. 2000;26:127–32.
Flenady V. Causes and risk factors of stillbirth in Australia. Perinatal Society of Austarlia and New Zealand Congress 2008. Gold Coast. Paediatr Child Health. 2008:2-10.
Marrow RJ, Ritchie JW, Bull SB. Maternal cigarette smoking: the effects on umbilical and uterine blood flow velocity. Am J Obstet Gynecol. 1988;159:1069–71.
Lambers DS, Clark KE. The maternal and fetal physiologic effects of nicotine. Semin Perinatol. 1996;20:115–26.
Ylikorkala O, Viinikka L, Lehtovirta P. Effect of nicotine on fetal prostacyclin and thromboxane in humans. Obstet Gynecol. 1985;66:102–5.
Bruner JP, Forouzan I. Smoking and Bucally administered nicotine. Acute effect on uterine and umbilical artery Doppler flow velocity wave forms. J Reprod Med. 1991;36:435–40.
Salihu HM, Aliyu HM, Kirby RS. In utero nicotine exposure and fetal growth inhibition among twins. Am J Perinatol. 2005;22:421–7.
Hammoud AO, Bujold E, Sorokin Y, Schild C, Krapp M, Baumann P. Smoking in pregnancy revisited: findings from a large population-based study. Am J Obstet Gynecol. 2005;192:1856–63.
Hajek P, Stead LF, West R, Jarvis M, Hartmann-Boyce J, Lancaster T. Relapse prevention interventions for smoking cessation. Cochrane Database Syst Rev 2013, Issue 8. Art. No.: CD003999. doi:10.1002/14651858.CD003999.pub4.
Coleman T, Chamberlain C, Davey MA, Cooper SE, Leonardi-Bee J. Pharmacological interventions for promoting smoking cessation during pregnancy. Cochrane Database Syst Rev 2012, Issue 9. Art. No.: CD010078. doi:10.1002/14651858.CD010078
Download references
Acknowledgments
We acknowledge the support of the Greenfield Medical Library staff of University of Nottingham, UK.
Author information
Authors and affiliations.
Division of Epidemiology and Public Health, Faculty of Medicine and Health Sciences, University of Nottingham, Clinical Sciences Building 2, Nottingham City Hospital, Hucknall Road, Nottingham, NG5 1 PB, UK
Takawira C Marufu, Anand Ahankari & Sarah Lewis
Halo Medical Foundation, Osmanabad, India
Anand Ahankari
Division of Primary Care, Faculty of Medicine and Health Sciences, University of Nottingham, Nottingham, UK
Tim Coleman
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Sarah Lewis .
Additional information
Competing interests.
The authors declare that they have no competing interests.
Authors’ contributions
The study was conceptualised by SL, TC, TM and AA. All authors participated in electronic search, title screening, study selection, data extraction, analysis and manuscript preparation. AA and TM contributed equally in the manuscript preparation, and thus shared the first authorship equally. All four authors provided intellectual content and approved the manuscript for publication.
Additional files
Additional file 1:.
Supplementary 1. Sample search strategy used for identification of studies.
Additional file 2:
Funnel plot.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/ .
The Creative Commons Public Domain Dedication waiver ( https://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Marufu, T.C., Ahankari, A., Coleman, T. et al. Maternal smoking and the risk of still birth: systematic review and meta-analysis. BMC Public Health 15 , 239 (2015). https://doi.org/10.1186/s12889-015-1552-5
Download citation
Received : 24 October 2014
Accepted : 16 February 2015
Published : 13 March 2015
DOI : https://doi.org/10.1186/s12889-015-1552-5
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Maternal Smoking
- Adverse Pregnancy Outcome
- Cigarette Consumption
- Newcastle Ottawa Scale
BMC Public Health
ISSN: 1471-2458
- General enquiries: [email protected]
Maternal smoking status during pregnancy and low birth weight in offspring: systematic review and meta-analysis of 55 cohort studies published from 1986 to 2020
- Meta-analysis
- Published: 28 January 2022
- Volume 18 , pages 176–185, ( 2022 )
Cite this article

- Hong-Kun Di 1 ,
- Yong Gan 1 ,
- Chao Wang 1 ,
- Xin Meng 1 ,
- Wen-Qi Xia 1 ,
- Min-Zhi Xu 1 ,
- Jing Feng 1 ,
- Qing-Feng Tian 3 ,
- Zhi-Qiang Nie 1 ,
- Jun-An Liu 1 ,
- Fu-Jian Song 4 &
- Zu-Xun Lu ORCID: orcid.org/0000-0001-8432-5109 1
2583 Accesses
35 Citations
1 Altmetric
Explore all metrics
Maternal smoking during pregnancy may be associated with low birth weight (LBW) in offspring and global risk estimates have not been summarized previously. We aimed to systematically explore evidence regarding maternal smoking and the LBW risk in offspring globally and examine possible causes of heterogeneity across relevant studies.
Comprehensive search of PubMed, Ovid Embase, Ovid Medline (R), and Web of science from inception until October 2021 was carried out. A random-effects meta-analysis was used to estimate the pooled odds ratio (OR) and corresponding 95% confidence interval (CI). Restricted cubic spline analysis with three knots was used to further examine the dose–response relationship.
Literature searches yielded 4940 articles, of which 53 met inclusion criteria (comprising 55 independent studies). Maternal smoking during pregnancy was significantly associated with the risk of LBW in offspring (OR = 1.89, 95% CI = 1.80–1.98). Furthermore, an obvious dose–response relationship between the amount of cigarettes daily smoked in pregnancy and the risk of LBW in offspring was observed. The results of subgroup analyses indicated that the risk of maternal smoking on LBW was larger in more recently conducted studies ( P = 0.020) and longer period of active smoking during pregnancy ( P = 0.002). No evidence of publication bias was found.
Conclusions
In summary, maternal smoking in pregnancy was significantly associated with a higher risk of LBW in offspring on a global scale. The risk of maternal smoking on infant LBW seems to be increasing over time, and was higher with longer smoking duration throughout pregnancy and more cigarettes smoked daily.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions
Similar content being viewed by others

Maternal smoking during pregnancy and offspring overweight: is there a dose–response relationship? An individual patient data meta-analysis
Maternal smoking and the risk of still birth: systematic review and meta-analysis.
The maternal factors associated with infant low birth weight: an umbrella review
Data availability statement.
Studies used for meta-analysis are listed in Supplementary eTable 2.
Sellers R, Warne N, Rice F, Langley K, Maughan B, Pickles A, et al. Using a cross-cohort comparison design to test the role of maternal smoking in pregnancy in child mental health and learning: evidence from two UK cohorts born four decades apart. Int J Epidemiol. 2020;49:390–9.
Article PubMed PubMed Central Google Scholar
Nakamura MU, Alexandre SM, Kuhn dos Santos JF, de Souza E, Sass N, Auritscher Beck AP, et al. Obstetric and perinatal effects of active and/or passive smoking during pregnancy. Sao Paulo Med J. 2004;122:94–8.
Article PubMed Google Scholar
Talati A, Keyes KM, Hasin DS. Changing relationships between smoking and psychiatric disorders across twentieth century birth cohorts: clinical and research implications. Mol Psychiatry. 2016;21:464–71.
Article CAS PubMed PubMed Central Google Scholar
Bednarczuk N, Milner A, Greenough A. The Role of maternal smoking in sudden fetal and infant death pathogenesis. Front Neurol. 2020;11:586068.
Hansen AR, Akomolafe TO, McGalliard Z, Belle-Isle L, Zhang J. Striving to meet healthy people 2020 objectives: trend analysis of maternal smoking. Public Health Rep. 2018;133:644–9.
Philips EM, Santos S, Trasande L, Aurrekoetxea JJ, Barros H, von Berg A, et al. Changes in parental smoking during pregnancy and risks of adverse birth outcomes and childhood overweight in Europe and North America: an individual participant data meta-analysis of 229,000 singleton births. PLoS Med. 2020;17:e1003182.
Li H, Hansen AR, McGalliard Z, Gover L, Yan F, Zhang J. Trends in Smoking and Smoking Cessation During Pregnancy from 1985 to 2014, Racial and Ethnic Disparity Observed from Multiple National Surveys. Matern Child Health J. 2018;22:685–93.
Veloso HJ, da Silva AA, Bettiol H, Goldani MZ, Filho FL, Simões VM, et al. Low birth weight in São Luís, northeastern Brazil: trends and associated factors. BMC Pregnancy Childbirth. 2014;14:155.
Mehaffey K, Higginson A, Cowan J, Osborne GM, Arbour LT. Maternal smoking at first prenatal visit as a marker of risk for adverse pregnancy outcomes in the Qikiqtaaluk (Baffin) Region. Rural Remote Health. 2010;10:1484.
PubMed Google Scholar
Okah FA, Cai J, Hoff GL. Term-gestation low birth weight and health-compromising behaviors during pregnancy. Obstet Gynecol. 2005;105:543–50.
Armstrong BG, McDonald AD, Sloan M. Cigarette, alcohol, and coffee consumption and spontaneous abortion. Am J Public Health. 1992;82:85–7.
Räisänen S, Sankilampi U, Gissler M, Kramer MR, Hakulinen-Viitanen T, Saari J, et al. Smoking cessation in the first trimester reduces most obstetric risks, but not the risks of major congenital anomalies and admission to neonatal care: a population-based cohort study of 1,164,953 singleton pregnancies in Finland. J Epidemiol Community Health. 2014;68:159–64.
Jaddoe VW, Troe EJ, Hofman A, Mackenbach JP, Moll HA, Steegers EA, et al. Active and passive maternal smoking during pregnancy and the risks of low birthweight and preterm birth: the Generation R Study. Paediatr Perinat Epidemiol. 2008;22:162–71.
Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018;392:1923–94.
Ratnasiri AWG, Gordon L, Dieckmann RA, Lee HC, Parry SS, Arief VN, et al. Smoking during pregnancy and adverse birth and maternal outcomes in California, 2007 to 2016. Am J Perinatol. 2020;37:1364–76.
Pereira PP, Da Mata FA, Figueiredo AC, de Andrade KR, Pereira MG. Maternal active smoking during pregnancy and low birth weight in the Americas: a systematic review and meta-analysis. Nicotine Tob Res. 2017;19:497–505.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000;283:2008–12.
Article CAS PubMed Google Scholar
Zhang J, Yu KF. What’s the relative risk? A method of correcting the odds ratio in cohort studies of common outcomes. JAMA. 1998;280:1690–1.
Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301–9.
Xaverius PK, O’Reilly Z, Li A, Flick LH, Arnold LD. Smoking cessation and pregnancy: timing of cessation reduces or eliminates the effect on low birth weight. Matern Child Health J. 2019;23:1434–41.
Franco Monsreal J, Tun Cobos MDR, Hernández Gómez JR, Serralta PL. Risk factors for low birth weight according to the multiple logistic regression model. A retrospective cohort study in José María Morelos municipality, Quintana Roo. Mexico Medwave. 2018;18:7139.
Google Scholar
Ju AC, Heyman MB, Garber AK, Wojcicki JM. Maternal Obesity and Risk of Preterm Birth and Low Birthweight in Hawaii PRAMS, 2000–2011. Matern Child Health J. 2018;22:893–902.
Witt WP, Mandell KC, Wisk LE, Cheng ER, Chatterjee D, Wakeel F, et al. Infant birthweight in the US: the role of preconception stressful life events and substance use. Arch Womens Ment Health. 2016;19:529–42.
Lundsberg LS, Illuzzi JL, Belanger K, Triche EW, Bracken MB. Low-to-moderate prenatal alcohol consumption and the risk of selected birth outcomes: a prospective cohort study. Ann Epidemiol. 2015;25:46-54.e43.
Nkansah-Amankra S, Luchok KJ, Hussey JR, Watkins K, Liu X. Effects of maternal stress on low birth weight and preterm birth outcomes across neighborhoods of South Carolina, 2000–2003. Matern Child Health J. 2010;14:215–26.
Pogodina C, Brunner Huber LR, Racine EF, Platonova E. Smoke-free homes for smoke-free babies: the role of residential environmental tobacco smoke on low birth weight. J Community Health. 2009;34:376–82.
Shiono PH, Klebanoff MA, Rhoads GG. Smoking and drinking during pregnancy. Their effects on preterm birth. JAMA. 1986;255:82–4.
Fox SH, Koepsell TD, Daling JR. Birth weight and smoking during pregnancy–effect modification by maternal age. Am J Epidemiol. 1994;139:1008–15.
da Silva AA, Gomes UA, Bettiol H, Dal Bo CM, Mucillo G. Barbieri MA [Correlation between maternal age, social class and smoking, and birth weight]. Rev Saude Publica. 1992;26:150–4.
Scholl TO, Salmon RW, Miller LK. Smoking and adolescent pregnancy outcome. J Adolesc Health Care. 1986;7:390–4.
Barbieri MA, Silva AA, Bettiol H, Gomes UA. Risk factors for the increasing trend in low birth weight among live births born by vaginal delivery. Brazil Rev Saude Publica. 2000;34:596–602.
Silva AA, Barbieri MA, Gomes UA, Bettiol H. Trends in low birth weight: a comparison of two birth cohorts separated by a 15-year interval in Ribeirão Preto. Brazil Bull World Health Organ. 1998;76:73–84.
CAS PubMed Google Scholar
Horta BL, Victora CG, Menezes AM, Halpern R, Barros FC. Low birthweight, preterm births and intrauterine growth retardation in relation to maternal smoking. Paediatr Perinat Epidemiol. 1997;11:140–51.
Solla JJ, Pereira RA, Medina MG, Pinto LL. Mota E [Multifactorial analysis of risk factors for low birth weight in Salvador, Bahia]. Rev Panam Salud Publica. 1997;2:1–6.
Rumrich I, Vähäkangas K, Viluksela M, Gissler M, de Ruyter H, Hänninen O. Effects of maternal smoking on body size and proportions at birth: a register-based cohort study of 1.4 million births. BMJ Open. 2020;10:e033465.
Møller SE, Ajslev TA, Andersen CS, Dalgård C, Sørensen TI. Risk of childhood overweight after exposure to tobacco smoking in prenatal and early postnatal life. PLoS ONE. 2014;9:e109184.
Balázs P, Rákóczi I, Grenczer A, Foley KL. Risk factors of preterm birth and low birth weight babies among Roma and non-Roma mothers: a population-based study. Eur J Public Health. 2013;23:480–5.
Bakker R, Kruithof C, Steegers EA, Tiemeier H, Mackenbach JP, Hofman A, et al. Assessment of maternal smoking status during pregnancy and the associations with neonatal outcomes. Nicotine Tob Res. 2011;13:1250–6.
Vardavas CI, Chatzi L, Patelarou E, Plana E, Sarri K, Kafatos A, et al. Smoking and smoking cessation during early pregnancy and its effect on adverse pregnancy outcomes and fetal growth. Eur J Pediatr. 2010;169:741–8.
Ward C, Lewis S, Coleman T. Prevalence of maternal smoking and environmental tobacco smoke exposure during pregnancy and impact on birth weight: retrospective study using Millennium Cohort. BMC Public Health. 2007;7:81.
Delpisheh A, Kelly Y, Rizwan S, Brabin BJ. Socio-economic status, smoking during pregnancy and birth outcomes: an analysis of cross-sectional community studies in Liverpool (1993–2001). J Child Health Care. 2006;10:140–8.
Chiolero A, Bovet P, Paccaud F. Association between maternal smoking and low birth weight in Switzerland: the EDEN study. Swiss Med Wkly. 2005;135:525–30.
Dewan N, Brabin B, Wood L, Dramond S, Cooper C. The effects of smoking on birthweight-for-gestational-age curves in teenage and adult primigravidae. Public Health. 2003;117:31–5.
Meis PJ, Michielutte R, Peters TJ, Wells HB, Sands RE, Coles EC, et al. Factors associated with term low birthweight in Cardiff, Wales. Paediatr Perinat Epidemiol. 1997;11:287–97.
Ahlborg G Jr, Bodin L. Tobacco smoke exposure and pregnancy outcome among working women. A prospective study at prenatal care centers in Orebro County. Sweden Am J Epidemiol. 1991;133:338–47.
Lewandowska M, Więckowska B, Sztorc L, Sajdak S. Smoking and Smoking Cessation in the Risk for Fetal Growth Restriction and Low Birth Weight and Additive Effect of Maternal Obesity. J Clin Med 2020;9.
Flower A, Shawe J, Stephenson J, Doyle P. Pregnancy planning, smoking behaviour during pregnancy, and neonatal outcome: UK Millennium Cohort Study. BMC Pregnancy Childbirth. 2013;13:238.
Chen S, Yang Y, Qu Y, Zou Y, Zhu H, Yang H, et al. Both maternal and paternal risk factors for term singleton low birthweight infants in rural Chinese population: a population-based, retrospective cohort study. Sci Rep. 2018;8:12539.
Zheng W, Suzuki K, Tanaka T, Kohama M, Yamagata Z. Association between maternal smoking during pregnancy and low birthweight: effects by maternal age. PLoS ONE. 2016;11:e0146241.
Andriani H, Kuo HW. Adverse effects of parental smoking during pregnancy in urban and rural areas. BMC Pregnancy Childbirth. 2014;14:414.
Ko TJ, Tsai LY, Chu LC, Yeh SJ, Leung C, Chen CY, et al. Parental smoking during pregnancy and its association with low birth weight, small for gestational age, and preterm birth offspring: a birth cohort study. Pediatr Neonatol. 2014;55:20–7.
Miyake Y, Tanaka K, Arakawa M. Active and passive maternal smoking during pregnancy and birth outcomes: the Kyushu Okinawa maternal and child health study. BMC Pregnancy Childbirth. 2013;13:157.
Bener A, Salameh KM, Yousafzai MT, Saleh NM. Pattern of maternal complications and low birth weight: associated risk factors among highly endogamous women. ISRN Obstet Gynecol. 2012;2012:540495.
Suzuki K, Tanaka T, Kondo N, Minai J, Sato M, Yamagata Z. Is maternal smoking during early pregnancy a risk factor for all low birth weight infants? J Epidemiol. 2008;18:89–96.
Tamim H, Yunis KA, Chemaitelly H, Alameh M, Nassar AH. Effect of narghile and cigarette smoking on newborn birthweight. BJOG. 2008;115:91–7.
Vahdaninia M, Tavafian SS, Montazeri A. Correlates of low birth weight in term pregnancies: a retrospective study from Iran. BMC Pregnancy Childbirth. 2008;8:12.
Maruoka K, Yagi M, Akazawa K, Kinukawa N, Ueda K, Nose Y. Risk factors for low birthweight in Japanese infants. Acta Paediatr. 1998;87:304–9.
Jadsri S, Jadsri C. Parental smoking habits and infant birth weight. Asia Pac J Public Health. 1995;8:134–7.
Nematollahi S, Mansournia MA, Foroushani AR, Mahmoodi M, Alavi A, Shekari M, et al. The effects of water-pipe smoking on birth weight: a population-based prospective cohort study in southern Iran. Epidemiol Health. 2018;40:e2018008.
Bird AL, Grant CC, Bandara DK, Mohal J, Atatoa-Carr PE, Wise MR, et al. Maternal health in pregnancy and associations with adverse birth outcomes: evidence from Growing Up in New Zealand. Aust N Z J Obstet Gynaecol. 2017;57:16–24.
Zhao L, McCauley K, Sheeran L. The interaction of pregnancy, substance use and mental illness on birthing outcomes in Australia. Midwifery. 2017;54:81–8.
Uncu Y, Ozcakir A, Ercan I, Bilgel N, Uncu G. Pregnant women quit smoking; what about fathers? Survey study in Bursa Region. Turkey Croat Med J. 2005;46:832–7.
Bai J, Wong FW, Gyaneshwar R, Stewart HC. Profile of maternal smokers and their pregnancy outcomes in south western Sydney. J Obstet Gynaecol Res. 2000;26:127–32.
Salihu HM, Aliyu MH, Kirby RS. In utero nicotine exposure and fetal growth inhibition among twins. Am J Perinatol. 2005;22:421–7.
Chan A, Keane RJ, Robinson JS. The contribution of maternal smoking to preterm birth, small for gestational age and low birthweight among Aboriginal and non-Aboriginal births in South Australia. Med J Aust. 2001;174:389–93.
Rice F, Langley K, Woodford C, Davey Smith G, Thapar A. Identifying the contribution of prenatal risk factors to offspring development and psychopathology: What designs to use and a critique of literature on maternal smoking and stress in pregnancy. Dev Psychopathol. 2018;30:1107–28.
Hackshaw A, Rodeck C, Boniface S. Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update. 2011;17:589–604.
Veisani Y, Jenabi E, Delpisheh A, Khazaei S. Effect of prenatal smoking cessation interventions on birth weight: meta-analysis. J Matern Fetal Neonatal Med. 2019;32:332–8.
Banderali G, Martelli A, Landi M, Moretti F, Betti F, Radaelli G, et al. Short and long term health effects of parental tobacco smoking during pregnancy and lactation: a descriptive review. J Transl Med. 2015;13:327.
England LJ, Kendrick JS, Gargiullo PM, Zahniser SC, Hannon WH. Measures of maternal tobacco exposure and infant birth weight at term. Am J Epidemiol. 2001;153:954–60.
Soneji S, Beltrán-Sánchez H. Association of maternal cigarette smoking and smoking cessation with preterm birth. JAMA Netw Open. 2019;2:e192514.
Ghosh RE, Berild JD, Sterrantino AF, Toledano MB, Hansell AL. Birth weight trends in England and Wales (1986–2012): babies are getting heavier. Arch Dis Child Fetal Neonatal Ed. 2018;103:F264-70.
Alberman E, Botting B. Trends in prevalence and survival of very low birthweight infants, England and Wales: 1983–7. Arch Dis Child. 1991;66:1304–8.
Olsen J, Frische G. Social differences in reproductive health. A study on birth weight, stillbirths and congenital malformations in Denmark. Scand J Soc Med. 1993;21:90–7.
Adams KE, Melvin CL, Raskind-Hood CL. Sociodemographic, insurance, and risk profiles of maternal smokers post the 1990s: how can we reach them? Nicotine Tob Res. 2008;10:1121–9.
Download references
Acknowledgements
We thank all the authors of the studies included in our meta-analysis.
This study was funded by the by the National Social Science Foundation of China (Grant No. 18ZDA085).
Author information
Authors and affiliations.
Department of Social Medicine and Health Management, School of Public Health, Tongji Medical College, Huazhong University of Science and Technology, No. 13 Hangkong Road, Wuhan, 430030, Hubei, China
Hong-Kun Di, Yong Gan, Chao Wang, Yi Zhu, Xin Meng, Wen-Qi Xia, Min-Zhi Xu, Jing Feng, Zhi-Qiang Nie, Jun-An Liu & Zu-Xun Lu
Tongji Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, Hubei, China
School of Public Health, Zhengzhou University, Zhengzhou, Henan, China
Qing-Feng Tian & Yan He
Norwich Medical School, Faculty of Medicine and Health Science, University of East Anglia, Norwich, NR47TJ, UK
Fu-Jian Song
You can also search for this author in PubMed Google Scholar
Contributions
HKD and YG contributed equally to this work. HKD, YG, and ZXL conceived the study. HKD and YG searched the databases and checked them according to the eligible criteria and exclusion criteria. KL, CW and YZ helped develop search strategies. HKD and KL did the data extraction and quality assessment. HKD and YG analyzed the data. YZ, XM, JAL and FJS gave advice on meta-analysis methodology. H.K.D and Y.G wrote the draft of the paper. HKD, YG, YZ, WQX, XM, MZX, JF, JAL, FJS and ZXL contributed to reviewing or revising the paper. All authors read and approved the final manuscript. ZXL is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding authors
Correspondence to Fu-Jian Song or Zu-Xun Lu .
Ethics declarations
Ethical approval.
Not needed.
Conflict of interest
We declare that we have no conflicts of interest.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file(PDF 1088 KB)
Rights and permissions.
Reprints and permissions
About this article
Di, HK., Gan, Y., Lu, K. et al. Maternal smoking status during pregnancy and low birth weight in offspring: systematic review and meta-analysis of 55 cohort studies published from 1986 to 2020. World J Pediatr 18 , 176–185 (2022). https://doi.org/10.1007/s12519-021-00501-5
Download citation
Received : 17 August 2021
Accepted : 08 December 2021
Published : 28 January 2022
Issue Date : March 2022
DOI : https://doi.org/10.1007/s12519-021-00501-5
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Low birth weight
- Maternal smoking
- Find a journal
- Publish with us
- Track your research
- Introduction
- Conclusions
- Article Information
Differences in standardized subcortical volumes in children aged 10 years who were exposed to maternal smoking compared with nonexposed children. Linear regression models were adjusted for child sex and age at brain assessment, and maternal ethnicity, age at enrollment, marital status, educational level, psychopathology score, alcohol use during pregnancy, household income, and intracranial volume.
Cortical morphology (ie, thickness, surface area, and gyrification) in children aged 10 years born to mothers who continued smoked during pregnancy (n = 364) compared with those born to mothers who never smoked during pregnancy (reference category [n = 2091], excluding children who did not have adequate vertex-wise data). Vertex-wise linear regression was used; the presented model was adjusted for child sex and age at brain assessment and maternal ethnicity, age at enrollment, marital status, educational level, psychopathology score, alcohol use during pregnancy, and household income. Red to yellow brain regions represent larger surface areas, thicker cortices, or more gyrification; dark to light blue regions represent smaller surface areas, thinner cortices, or less gyrification. The colored clusters in this figure all survived a clusterwise (Monte Carlo simulation with 5000 iterations) correction for multiple comparisons ( P < .001). Statistical details of the associated clusters are provided in eTable 2 in the Supplement . LH indicates left hemisphere; RH, right hemisphere.
eMethods. Parental Tobacco Use, Neuroimaging, and Statistical Analysis
eFigure 1. Flowchart for Study Population Selection
eTable 1. Nonresponse Analysis
eTable 2. Association of Maternal Smoking During Pregnancy With Cortical Morphology in 10-Year-Old Children
eTable 3. Demographic Information of Children With DNA Methylation (n = 784)
eFigure 2. Mediation Analysis of Methylation Risk Score
eTable 4. Association of Maternal Smoking During Pregnancy With Regional Brain Volumes in 10-Year-Old Children, Inverse Probability Weighted
eTable 5. Association of Maternal Smoking During Pregnancy With Regional Brain Volumes in 10-Year-Old Children, Prenatally Included Only
eFigure 3. Association of Maternal Smoking During Pregnancy With Cortical Morphology in 10-Year-Old Children, Prenatally Included Only
- Association Between Smoking During Pregnancy and Compromised Brain Development of Offspring JAMA Network Open Invited Commentary August 1, 2022 Mikael O. Ekblad, MD, PhD
See More About
Sign up for emails based on your interests, select your interests.
Customize your JAMA Network experience by selecting one or more topics from the list below.
- Academic Medicine
- Acid Base, Electrolytes, Fluids
- Allergy and Clinical Immunology
- American Indian or Alaska Natives
- Anesthesiology
- Anticoagulation
- Art and Images in Psychiatry
- Artificial Intelligence
- Assisted Reproduction
- Bleeding and Transfusion
- Caring for the Critically Ill Patient
- Challenges in Clinical Electrocardiography
- Climate and Health
- Climate Change
- Clinical Challenge
- Clinical Decision Support
- Clinical Implications of Basic Neuroscience
- Clinical Pharmacy and Pharmacology
- Complementary and Alternative Medicine
- Consensus Statements
- Coronavirus (COVID-19)
- Critical Care Medicine
- Cultural Competency
- Dental Medicine
- Dermatology
- Diabetes and Endocrinology
- Diagnostic Test Interpretation
- Drug Development
- Electronic Health Records
- Emergency Medicine
- End of Life, Hospice, Palliative Care
- Environmental Health
- Equity, Diversity, and Inclusion
- Facial Plastic Surgery
- Gastroenterology and Hepatology
- Genetics and Genomics
- Genomics and Precision Health
- Global Health
- Guide to Statistics and Methods
- Hair Disorders
- Health Care Delivery Models
- Health Care Economics, Insurance, Payment
- Health Care Quality
- Health Care Reform
- Health Care Safety
- Health Care Workforce
- Health Disparities
- Health Inequities
- Health Policy
- Health Systems Science
- History of Medicine
- Hypertension
- Images in Neurology
- Implementation Science
- Infectious Diseases
- Innovations in Health Care Delivery
- JAMA Infographic
- Law and Medicine
- Leading Change
- Less is More
- LGBTQIA Medicine
- Lifestyle Behaviors
- Medical Coding
- Medical Devices and Equipment
- Medical Education
- Medical Education and Training
- Medical Journals and Publishing
- Mobile Health and Telemedicine
- Narrative Medicine
- Neuroscience and Psychiatry
- Notable Notes
- Nutrition, Obesity, Exercise
- Obstetrics and Gynecology
- Occupational Health
- Ophthalmology
- Orthopedics
- Otolaryngology
- Pain Medicine
- Palliative Care
- Pathology and Laboratory Medicine
- Patient Care
- Patient Information
- Performance Improvement
- Performance Measures
- Perioperative Care and Consultation
- Pharmacoeconomics
- Pharmacoepidemiology
- Pharmacogenetics
- Pharmacy and Clinical Pharmacology
- Physical Medicine and Rehabilitation
- Physical Therapy
- Physician Leadership
- Population Health
- Primary Care
- Professional Well-being
- Professionalism
- Psychiatry and Behavioral Health
- Public Health
- Pulmonary Medicine
- Regulatory Agencies
- Reproductive Health
- Research, Methods, Statistics
- Resuscitation
- Rheumatology
- Risk Management
- Scientific Discovery and the Future of Medicine
- Shared Decision Making and Communication
- Sleep Medicine
- Sports Medicine
- Stem Cell Transplantation
- Substance Use and Addiction Medicine
- Surgical Innovation
- Surgical Pearls
- Teachable Moment
- Technology and Finance
- The Art of JAMA
- The Arts and Medicine
- The Rational Clinical Examination
- Tobacco and e-Cigarettes
- Translational Medicine
- Trauma and Injury
- Treatment Adherence
- Ultrasonography
- Users' Guide to the Medical Literature
- Vaccination
- Venous Thromboembolism
- Veterans Health
- Women's Health
- Workflow and Process
- Wound Care, Infection, Healing
Get the latest research based on your areas of interest.
Others also liked.
- Download PDF
- X Facebook More LinkedIn
Zou R , Boer OD , Felix JF, et al. Association of Maternal Tobacco Use During Pregnancy With Preadolescent Brain Morphology Among Offspring. JAMA Netw Open. 2022;5(8):e2224701. doi:10.1001/jamanetworkopen.2022.24701
Manage citations:
© 2024
- Permissions
Association of Maternal Tobacco Use During Pregnancy With Preadolescent Brain Morphology Among Offspring
- 1 Department of Child and Adolescent Psychiatry/Psychology, Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands
- 2 Julius Center for Health Sciences and Primary Care, University Medical Center Utrecht, Utrecht University, Utrecht, the Netherlands
- 3 Institute for Risk Assessment Sciences, Utrecht University, Utrecht, the Netherlands
- 4 Department of Psychology, Education and Child Studies, Erasmus School of Social and Behavioral Sciences, Erasmus University Rotterdam, Rotterdam, the Netherlands
- 5 The Generation R Study Group, Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands
- 6 Department of Pediatrics, Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands
- 7 Department of Epidemiology, Erasmus MC, University Medical Center Rotterdam, Rotterdam, the Netherlands
- 8 Molecular Epidemiology, Department of Biomedical Data Sciences, Leiden University Medical Center, Leiden, the Netherlands
- Invited Commentary Association Between Smoking During Pregnancy and Compromised Brain Development of Offspring Mikael O. Ekblad, MD, PhD JAMA Network Open
Question Is maternal tobacco use during pregnancy associated with preadolescent brain morphology among offspring?
Findings In this cohort study of 2704 children in the Netherlands, exposure to continued maternal smoking during pregnancy was associated with lower global and regional brain volumes as well as smaller surface area and less gyrification at 9 to 11 years of age.
Meaning These findings suggest that maternal smoking throughout pregnancy may be associated with suboptimal brain development among offspring in the long term.
Importance Maternal tobacco use during pregnancy has been associated with various health consequences, including suboptimal neurodevelopment in offspring. However, the effect of prenatal exposure to maternal smoking on child brain development has yet to be elucidated.
Objective To investigate the association between maternal smoking during pregnancy and offspring brain development in preadolescence as well as the mediating pathways.
Design, Setting, and Participants This prospective, population-based cohort study was embedded in the Generation R Study, Rotterdam, the Netherlands. The Generation R Study was launched in 2002, with follow-up ongoing. Child brain morphology was assessed at 9 to 11 years of age (ie, 10-12 years between exposure and outcome assessment). Data analysis was performed from March 1, 2021, to February 28, 2022, and at the time of manuscript revision. Participants included the singleton children of pregnant women residing in the study area with an expected date of delivery between April 1, 2002, and January 31, 2006; 2704 children with information on maternal smoking during pregnancy and structural neuroimaging at 9 to 11 years of age were included. A subsample of 784 children with data on DNA methylation at birth was examined in the mediation analysis.
Exposures Information on maternal smoking during pregnancy was collected via a questionnaire in each trimester. As a contrast, paternal smoking was assessed at recruitment.
Main Outcomes and Measures Brain morphology, including brain volumes and surface-based cortical measures (thickness, surface area, and gyrification), was assessed with magnetic resonance imaging. For mediation analysis, DNA methylation at birth was quantified by a weighted methylation risk score.
Results The 2704 participating children (1370 [50.7%] girls and 1334 [49.3%] boys) underwent brain imaging assessment at a mean (SD) age of 10.1 (0.6) years. Compared with nonexposed children (n = 2102), exposure to continued maternal smoking during pregnancy (n = 364) was associated with smaller total brain volume (volumetric difference [ b ] = −14.5 [95% CI, −25.1 to −4.0] cm 3 ), cerebral gray matter volume ( b = −7.8 [95% CI, −13.4 to −2.3] cm 3 ), cerebral white matter volume ( b = −5.9 [95% CI, −10.7 to −1.0] cm 3 ), and surface area and less gyrification. These associations were not explained by paternal smoking nor mediated by smoking-associated DNA methylation patterns at birth. Children exposed to maternal smoking only in the first trimester (n = 238) showed no differences in brain morphology compared with nonexposed children.
Conclusions and Relevance The findings of this cohort study suggest that continued maternal tobacco use during pregnancy was associated with lower brain volumes and suboptimal cortical traits of offspring in preadolescence, which seemed to be independent of shared family factors. Tobacco cessation before pregnancy, or as soon as pregnancy is known, should be recommended to women for optimal brain development of their offspring.
Maternal tobacco use during pregnancy affects the health of not only the mother but also her offspring. Despite the well-documented child growth consequences, 1 accumulating evidence suggests that maternal tobacco use during pregnancy is also associated with suboptimal neurodevelopment of offspring, including impaired cognitive abilities, bipolar disorder, and schizophrenia spectrum disorders, but shared genetic and family factors hamper causal inference. 2 - 4
Differences in structural brain development may underlie the observed neurocognitive outcomes. A prospective study including 232 preterm infants 5 found associations of prenatal maternal smoking with smaller frontal lobe and cerebellum at corrected term age. Using a retrospective design, 6 a smaller amygdala was observed in 180 adolescents prenatally exposed to maternal smoking compared with 198 without exposure, although no difference was found in total brain volume (TBV). In a subgroup of our own cohort in an earlier phase, 7 maternal smoking throughout pregnancy was associated with less TBV and cortical gray matter volume, as well as thinner cortices (primarily in the frontal and parietal lobes) in 97 exposed children and 113 nonexposed controls aged 6 to 8 years. However, the small sample sizes of the aforementioned research made it difficult to capture subtle effect sizes, and investigations in specific groups (eg, preterm children) limit the generalizability of the findings. In addition, more efforts should be made to investigate the causal nature of observed relations, given the confounding issues in research on maternal smoking during pregnancy and child outcomes. 8 Using paternal smoking as a negative control is a well-known approach to examine whether there is a direct intrauterine effect of a maternal exposure such as smoking. Similar maternal and paternal effects suggest that there may be familial or genetic confounding factors, whereas a larger maternal vs paternal effect supports a direct intrauterine effect. 9
Moreover, biological mechanisms underlying potential effects of tobacco exposure at the molecular level remain unclear. Interestingly, tobacco exposure is arguably one of the most significant environmental factors associated with DNA methylation alterations in both neonates (prenatal exposure) and adults (direct smoking). 10 For example, a genome-wide consortium meta-analysis including 13 cohorts 11 found that persistent maternal smoking during pregnancy was associated with more than 6000 differentially methylated 5′-C-phosphate-G-3′ (CpG) sites in neonates, many of which were mapped to genes involved in neural development. However, whether DNA methylation explains the observed associations between maternal smoking during pregnancy and offspring brain development remains unknown.
The present study aimed to investigate the prospective association between maternal tobacco use during pregnancy and brain morphology, including volumetric measures and cortical metrics in preadolescents. Paternal smoking during pregnancy was used as a negative control to test for shared genetic and family confounding. We also explored whether previously identified smoking-associated DNA methylation patterns at birth mediate any observed associations.
This cohort study was embedded in the Generation R Study, a prospective population-based study in Rotterdam, the Netherlands. Pregnant women with an expected delivery date between April 1, 2002, and January 31, 2006, residing in the study area were eligible for recruitment. 12 The Generation R Study is approved by the Medical Ethical Committee of Erasmus MC, University Medical Center Rotterdam. Written informed consent was obtained from all participants. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology ( STROBE ) reporting guideline.
The Generation R Study recruited a total of 9778 women who gave birth to 9506 live singletons. Among these children, 2704 with information on maternal tobacco use during pregnancy and usable brain magnetic resonance imaging (MRI) data at 9 to 11 years of age were included in the study population. eFigure 1 in the Supplement depicts the study population selection.
Postal questionnaires were used to collect information on maternal tobacco use in each trimester. 13 Maternal tobacco use during pregnancy was categorized based on all 3 questionnaires as (1) never during pregnancy, (2) until pregnancy was known, and (3) continued during pregnancy. For mothers enrolled during the postnatal period (n = 201), information on tobacco use during pregnancy was retrospectively obtained (with the same categorization). We also collected information on the frequency of tobacco use for the smoking mothers.
Information on paternal tobacco use preceding the pregnancy was obtained from both the mother and the partner using questionnaires at enrollment. Maternal- and self-reported paternal smoking status showed high agreement: 1606 of 1721 pairs (93.3%) provided the same answer.
Before the MRI scan, children underwent a mock scanning procedure to become familiar with the neuroimaging assessment. Brain images were acquired using the same sequence on the same 3.0-T MRI scanner (Discovery MR750; GE Healthcare) using an 8-channel head coil. Following a 3-plane localizer scan, T1-weighted structural images were acquired with an inversion recovery–prepared fast spoiled gradient recalled sequence. Further information on the sequence and neuroimaging procedure is described elsewhere. 14
Volumetric segmentation and cortical reconstruction were performed using the FreeSurfer software suite. 15 The standard reconstruction stream was applied, and surface-based models of white matter and gray matter were generated. The quality of surface reconstruction was visually inspected, after which data with insufficient quality were eliminated. 16
Based on previous studies, 6 , 7 , 17 we included the following covariates: child sex and age at neuroimaging, maternal age at enrollment, ethnicity (categorized as Dutch, non-Dutch western, and non-Dutch nonwestern according to the classification of Statistics Netherlands), educational level (categorized into primary, secondary, and higher education), marital status, parity, prepregnancy body mass index, psychopathology score, alcohol use, and household net monthly income. The psychopathology score was based on the Global Severity Index derived from the Brief Symptom Inventory (range, 0-4, with higher scores indicating more clinically relevant psychological symptoms). 18 Child sex and date of birth were obtained from medical records. Maternal prepregnancy body mass index was calculated by self-reported prepregnancy weight and height measured at enrollment. Information on all other covariates was collected with questionnaires during pregnancy.
Variance in DNA methylation was indexed using a methylation risk score (MRS). In brief, DNA was extracted from cord blood, and samples were processed with the Infinium HumanMethylation450 BeadChip array (Illumina Inc) followed by standardized laboratory quality control. 19 We quantified DNA methylation at each CpG site. Among the 6073 CpG sites that were associated with continued maternal smoking during pregnancy with false discovery rate significance, 11 5643 were available in 784 participants of European ancestry in our sample and were included to construct the weighted MRS. We also constructed an MRS using 551 CpG sites that survived a more stringent Bonferroni correction to compare results. In addition, we constructed a second MRS consisting specifically of CpG sites (405 with false discovery rate significance and 62 with Bonferroni significance) that in addition to showing associations with maternal smoking during pregnancy were also annotated to genes listed as part of the brain development pathway (GO:0007420). Information on plate number to indicate batch, cell types, and 4 genetic principal components of ancestry was collected as additional covariates in analyses of MRS. 20
Data were analyzed from March 1, 2021, to February 28, 2022. For descriptive purposes, continuous variables are presented as mean (SD) and categorical variables as No. (%). In the nonresponse analysis, we compared maternal and child variables between respondents (n = 2704) and nonrespondents (n = 5318) using an unpaired t test or Wilcoxon test for continuous variables and a χ 2 test for categorical variables.
In primary analyses, we used linear regression to examine maternal smoking in early pregnancy only or continued maternal smoking during pregnancy in association with offspring brain morphology at 9 to 11 years of age, with nonexposed children as the reference group. For volumetric measures, we used a hierarchical approach by first examining TBV as a global measure, followed by regional brain volumes including cerebral gray matter and white matter and the cerebellum. Volumetric differences were reported as b values. In secondary analyses, we examined subcortical brain volumes including the thalamus, amygdala, hippocampus, putamen, pallidum, caudate, and nucleus accumbens. These subcortical volumes were standardized before analysis owing to the variance in absolute volumes. For surface-based cortical brain measures, we used vertex-wise linear regression with a custom in-house QDECR R package at each cortical vertex to examine cortical thickness, surface area, and gyrification in children. 16 , 21 To investigate possible dose-response associations, we further evaluated maternal smoking frequency as a continuum to both volumetric and cortical measures in children born to continued smokers, because use of categorical variables can be subject to unbalanced sample sizes and is less sensitive to detect small effects. 22 , 23
In addition, we evaluated paternal smoking preceding pregnancy with child brain outcomes to explore whether any associations were explained by shared genetic or environmental factors. We also conducted a mediation analysis to examine whether the smoking-associated DNA MRS at birth mediates the association between prenatal exposure to maternal smoking and brain morphology at 9 to 11 years of age.
Two sensitivity analyses were performed to assess our findings. First, we used inverse probability weighting to count for potential attrition. Second, we excluded participants included after delivery to rule out recall bias on maternal smoking information.
We used multistage covariates adjustment in the above analyses. Child sex and age at neuroimaging were adjusted for in the minimally adjusted model, and maternal age at enrollment, ethnicity, marital status, educational level, psychopathology score, alcohol use during pregnancy, and household income were additionally adjusted for in the fully adjusted model based on the 5% change-in-estimate criterion. 24 For subcortical structures, intracranial volume (ICV) was additionally adjusted for in a third model to examine whether any volumetric differences were independent of the global brain size. For mediation analyses using the MRS, we adjusted for child sex and age at neuroimaging, maternal age at enrollment, maternal educational level, cell types, plate number, and genetic principal components.
Missing data on covariates were estimated using multivariate imputation by chained equations, 25 and we only report pooled results (results from the first imputed data set are reported for the mediation analyses). Statistical significance was set as α < .05 (2-sided), and a false discovery rate correction was applied in the primary analyses to minimize false-positive findings. 26 For surface-based analysis, correction for multiple testing was performed using built-in Gaussian Monte Carlo simulations. 27 All statistical analyses were performed using R, version 3.6.2 (R Project for Statistical Computing). In addition to standard packages for general analyses, we used the mediation R package to perform mediation analysis, 28 which estimates direct and indirect effects and their CIs based on bootstrapped simulations. More information on tobacco use assessment, neuroimaging, and statistical analysis can be found in eMethods in the Supplement .
Table 1 presents the demographic information of the participants. The 2704 participating children underwent brain MRI assessment at a mean (SD) age of 10.1 (0.6) years and included slightly more girls (1370 [50.7%]) than boys (1334 [49.3%]). Most of the mothers (2102 [77.7%]) never smoked during pregnancy, followed by 364 (13.5%) who continued smoking throughout pregnancy. In addition, 238 women (8.8%) stopped smoking when they were aware of the pregnancy, mostly in the first trimester (ie, before gestational age of 13 weeks). Results of the nonresponse analysis are summarized in eTable 1 in the Supplement .
Focusing on the fully adjusted models, continued maternal smoking during pregnancy was associated with lower TBV ( b = −14.5 [95% CI −25.1 to −4.0] cm 3 ) in children at 10 years of age, whereas exposure to smoking in early pregnancy only was not associated with TBV ( b = 0.2 [95% CI, −12.0 to 12.5] cm 3 ). Table 2 demonstrates the associations between maternal smoking during pregnancy and regional brain volumes of the child. Children born to mothers who continued smoking during pregnancy also showed smaller cerebral gray matter volume ( b = −7.8 [95% CI, −13.4 to −2.3] cm 3 ) and white matter volume ( b = −5.9 [95% CI, −10.7 to −1.0] cm 3 ) compared with nonexposed children, although no differences were found in cerebellar volume ( b = −0.8 [95% CI, −2.2 to 0.6] cm 3 ). Compared with nonexposed children, those born to women who quit smoking in early pregnancy showed no volumetric differences in these brain regions.
Next, we examined the volumes of subcortical structures with and without adjustment for ICV in the regression model. Figure 1 presents the results with adjustment for ICV, showing that maternal smoking throughout pregnancy was associated with larger amygdala and putamen among the children. Interestingly, such differences were not observed without adjustment for child ICV in the amygdala (β = 0.01 [95% CI, −0.1 to 0.1]) or putamen (β = 0.1 [95% CI, −0.03 to 0.2]), suggesting that these 2 structures were relatively larger in children exposed to continued maternal smoking during pregnancy. On the contrary, a smaller caudate (β = −0.2 [95% CI, −0.3 to −0.04]) and nucleus accumbens (β = −0.1 [95% CI, −0.2 to −0.02]) were found among children exposed to continued maternal smoking during pregnancy when ICV was not adjusted for, suggesting differences dependent on global brain size. Among the 364 children exposed to continued maternal smoking during pregnancy, we found no association between maternal smoking frequency during pregnancy and offspring global or regional brain volumes nor volumes of the subcortical structures, suggesting no clear dose-response association.
The surface-based analysis showed that, compared with nonexposed children, those exposed to continued maternal smoking during pregnancy had thicker cortices in the inferior parietal region of the left hemisphere ( b = 0.05; 301.6 mm 2 ; clusterwise P < .001) but smaller surface area in the temporal ( b = −0.02; 987.1 mm 2 ; clusterwise P < .001) and occipital ( b = −0.03; 313.8 mm 2 ; clusterwise P = .02) lobes of the left hemisphere and the pericalcarine cortex ( b = −0.04; 1633.5 mm 2 ; clusterwise P < .001) of the right hemisphere, inferior parietal regions of the left ( b = −0.03; 1081.9 mm 2 ; clusterwise P < .001) and right ( b = −0.04; 1674.3 mm 2 ; clusterwise P < .001) hemispheres, and less gyrification in the postcentral region ( b = −0.08; 1017.2 mm 2 ; clusterwise P < .001) of the left hemisphere ( Figure 2 and eTable 2 in the Supplement ). Again, we observed no association of maternal smoking in early pregnancy only with cortical thickness, surface area, and gyrification of the child, nor was smoking frequency associated with these cortical metrics.
As shown in Table 3 , both maternal-reported and self-reported tobacco use of the partner during the periconceptional period were associated with a larger putamen of the child at 10 years of age (also when adjusted for ICV) ( b = 0.1 [95% CI, 0.04-0.2] cm 3 and b = 0.1 [95% CI, 0.03-0.2] cm 3 , respectively). In addition, maternal-reported paternal smoking was associated with child pallidum ( b = 0.1 [95% CI, 0.01-0.1] cm 3 ), whereas this association was not observed when using paternal self-reported data. Paternal smoking was not associated with other global, regional, or subcortical volumes. Surface-based analyses showed no association of paternal smoking with child cortical thickness, surface area, or gyrification.
The demographic information of children included in the mediation analysis on DNA methylation is displayed in eTable 3 in the Supplement . Although continued maternal smoking during pregnancy was associated with neonatal MRS, we found no evidence suggesting that neonatal MRS mediated the association between continued maternal smoking during pregnancy and child brain morphology at 10 years of age, using MRS calculated from all CpG sites or CpG sites annotated to genes of the brain development pathway (see eFigure 2 in the Supplement ). Consistent results were obtained when using MRS calculated from CpG sites with Bonferroni significance.
Analyses using inverse probability weighting yielded consistent results: compared with children without exposure, those exposed to continued maternal smoking during pregnancy showed a smaller TBV ( b = −15.7 [95% CI, −26.2 to −5.1] cm 3 ) and lower volumes of cerebral gray and white matter after full adjustment for covariates (eTable 4 in the Supplement ). Similarly, analyses in children born to women included during pregnancy only (n = 2503) showed smaller TBV ( b = −13.8 [95% CI, −24.6 to −3.1] cm 3 ), smaller volumes of cerebral gray matter ( b = −7.4 [95% CI, −13.1 to −1.7] cm 3 ) and white matter ( b = −5.5 [95% CI, −10.5 to −0.6] cm 3 ) (eTable 5 in the Supplement ), and smaller surface area and less gyrification (eTable 2 and eFigure 3 in the Supplement ) in children born to women who continued smoking during pregnancy than those born to women who never smoked during pregnancy, suggesting that our findings were not subject to recall bias.
In this study, continued maternal tobacco use during pregnancy was associated with smaller global and regional brain volumes as well as smaller cortical surface area and less gyrification in 10-year-old children. Importantly, we found no evidence suggesting that DNA methylation at birth, when indexed by a prenatal smoking–associated MRS, mediated these associations. No associations of exposure to early-pregnancy smoking with offspring brain morphology were observed. Furthermore, no evident association between paternal tobacco use and child brain morphology was observed. These findings suggest that persistent tobacco exposure in utero could compromise brain development 10 years later, which is unlikely to be explained by shared genetics or family factors.
Previous studies have shown that exposure to continued maternal tobacco use during pregnancy is associated with reduced growth of the head and smaller cerebellar size in fetal life 13 as well as smaller TBV, smaller cortical gray matter volume, and thinner cortices in early childhood (ie, age 6-8 years). 7 In the present study, we showed that the reduction in global and regional brain volumes in children exposed to continued maternal smoking during pregnancy remained at 9 to 11 years of age. This is in line with the findings from Rivkin et al 17 suggesting lower total parenchymal volume and cortical gray matter volume in children aged 10 to 14 years exposed to maternal cigarette use in utero. Interestingly, in contrast to previously cited studies 7 , 13 and a study by Toro et al 29 reporting thinner cortices in children aged 6 to 8 years and 15-year-old adolescents, the most notable cortical findings were smaller surface areas and less gyrification in the present study sample. The nonlinear pattern of cortical development, characterized by initial thickening in childhood and accelerated thinning in adolescence, may account for the heterogeneous findings in cortical thickness and hamper straightforward interpretations. 30 , 31 Importantly, in addition to adjusting for various sociodemographic and lifestyle factors, we used a negative control (ie, paternal smoking) in a separate analysis to address confounding bias, which is uncommon in neuroimaging studies.
Consistent with previous studies 7 , 13 showing no differences in fetal head growth or brain morphology at 6 to 8 years of age, we did not observe any brain morphological differences in children exposed to smoking in early pregnancy only. Therefore, we speculate that exposure to tobacco throughout pregnancy is more relevant for brain development than transient exposure in early gestation. This can be explained by the fact that human brain development in the embryonic period (ie, until 8 weeks of gestation) is primarily characterized by the differentiation of the neural progenitors and formation of the neural tube, thus exposure to maternal smoking at this stage exerts less impact on brain expansion than later in gestation, when substantial neurogenesis takes place. 32 In practice, this finding suggests that smoking cessation as soon as pregnancy is known, if not before pregnancy, is not too late for offspring brain development, which is an important public health message for pregnant smokers and their health care practitioners, such as midwives and obstetricians.
Several mechanisms could underlie the associations of intrauterine exposure to smoking with brain development at 10 years of age. First, nicotine is the major psychoactive compound and is known to interfere with neurodevelopment as a neuroteratogen. 33 Animal models have shown that prenatal nicotine exposure induces apoptotic cell death and decreases cell size in various brain regions, 34 , 35 which may lead to a smaller brain of the fetus that persists in the long term. Second, a recent study 36 suggested an adverse effect of tobacco exposure on bone growth in early life, which may cause a reduced skull capacity that originates in fetal life and persists in childhood and thus restricts brain development in the long term. Last, the prenatal period is a time of considerable epigenomic plasticity that is relevant for human brain development both in utero and after birth. 37 , 38 In the present study, neonatal DNA methylation patterns known to associate with prenatal maternal smoking, here indexed by the MRS, did not mediate the association between prenatal exposure to maternal smoking and brain morphology at 10 years of age. However, this potential mechanism cannot yet be excluded, because we did not systematically examine smoking-related CpG sites at an individual level, some of which may play a more profound role in neurodevelopment. 39 In addition, research has found maternal smoking–related CpG sites that are differentially methylated with the same direction and of a similar magnitude from birth to childhood. 11 , 40 Investigations of these specific CpG sites can be interesting, because these CpG sites may exert cumulative direct or indirect effects on the continuous brain development after birth. Furthermore, the substantially reduced sample size for the mediation analysis makes it difficult to capture subtle indirect effects.
Our study has several limitations. First, information on tobacco use was only collected with a questionnaire without using biomarkers such as plasma cotinine. 41 In addition, using paternal smoking proceeding pregnancy to index paternal smoking during pregnancy might lead to misclassification, although evidence shows consistent smoking habits of the partner. 42 Second, all of the children included in the DNA methylation sample in the Generation R Study were White; therefore, the results cannot be generalized to other racial or ethnic groups. Third, because this was an observational study, residual confounding may not be fully ruled out; thus, caution should be used when inferring causality. Future studies using complementary strategies (eg, sibling designs, mendelian randomization) to the paternal negative controls are warranted to strengthen causal inference. Additionally, the clinical implications of our findings are not explicit. Follow-up data collection on neurocognitive outcomes and the inclusion of functional brain measures such as electroencephalography or functional MRI are needed to unravel the clinical relevance of the observed differences in brain morphology.
The findings of this study suggest that continued maternal tobacco use during pregnancy was associated with offspring brain development in preadolescence. Therefore, interventions targeting maternal smoking cessation before pregnancy or in early pregnancy may favor normal brain development among children in the long term.
Accepted for Publication: June 2, 2022.
Published: August 1, 2022. doi:10.1001/jamanetworkopen.2022.24701
Open Access: This is an open access article distributed under the terms of the CC-BY License . © 2022 Zou R et al. JAMA Network Open .
Corresponding Author: Hanan El Marroun, PhD, Department of Child and Adolescent Psychiatry/Psychology, Erasmus MC, University Medical Center Rotterdam, PO Box 2060, 3000 CB Rotterdam, the Netherlands ( [email protected] ).
Author Contributions: Drs Zou and El Marroun had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Concept and design: Zou, Cecil, El Marroun.
Acquisition, analysis, or interpretation of data: Zou, Boer, Felix, Muetzel, Franken, El Marroun.
Drafting of the manuscript: Zou, Boer, El Marroun.
Critical revision of the manuscript for important intellectual content: All authors.
Statistical analysis: Zou, El Marroun.
Obtained funding: Muetzel, El Marroun.
Administrative, technical, or material support: Muetzel, El Marroun.
Supervision: Felix, Franken, Cecil, El Marroun.
Conflict of Interest Disclosures: None reported.
Funding/Support: This study was supported by Stichting Volksbond Rotterdam (Ms Boer and Dr El Marroun); NARSAD Young Investigator Grant 27853 from the Brain & Behavior Research Foundation (Dr El Marroun); Aspasia grant 015.016.056 from the Netherlands Organization for Scientific Research (NWO) (Dr El Marroun); and grant agreement 848158 (EarlyCause Project) (Drs Felix and Cecil) and 733206 LifeCycle from the European Union’s Horizon 2020 Research and Innovation Program. Neuroimaging, image analysis, and informatics were supported by grant S18-20 from the Sophia Foundation (Dr Muetzel). Supercomputing resources were supported by the Netherlands Organization for Scientific Research (Exacte Wetenschappen) and SURFsara (Cartesius Computer Cluster). The general design of Generation R Study is made possible by financial support from Erasmus MC, Erasmus University Rotterdam, the Netherlands Organization for Health Research and Development, the Netherlands Organization for Scientific Research, the Ministry of Health, Welfare and Sport, and the Ministry of Youth and Families. The epigenome-wide association study data were funded by project 050-060-810 from the Netherlands Genomics Initiative/Netherlands Organization for Scientific Research, Netherlands Consortium for Healthy Aging, by funds from the Genetic Laboratory of the Department of Internal Medicine, Erasmus MC, and by grant R01HD068437 from the National Institute of Child and Human Development.
Role of the Funder/Sponsor: The sponsors had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
- Register for email alerts with links to free full-text articles
- Access PDFs of free articles
- Manage your interests
- Save searches and receive search alerts
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- HHS Author Manuscripts

Maternal smoking during pregnancy and child outcomes: Real or spurious effect?
Valerie s. knopik.
1 Center for Alcohol and Addiction Studies, Department of Community Health, Brown University, Providence, RI
Maternal smoking during pregnancy (MSDP) is a major public health concern with clearly established consequences to both mother and newborn (e.g., low birth weight, altered cardiorespiratory responses). MSDP has also been associated with higher rates of a variety of poor cognitive and behavioral outcomes in children, including ADHD, conduct disorder, impaired learning and memory, and cognitive dysfunction. However, the evidence suggesting causal effects of MSDP for these outcomes is muddied in the existing literature due to the frequent inability to separate prenatal exposure effects from other confounding environmental and genetic factors. Carefully designed studies using genetically sensitive strategies can build upon current evidence and begin to elucidate the likely complex factors contributing to associations between MSDP and child outcomes.
Introduction
Maternal smoking during pregnancy (MSDP) is a major public health concern with nearly half of all women who smoke continuing to do so throughout their pregnancies ( Centers for Disease Control (CDC), 2002 , 2004 ; Ebrahim, Floyd, Merrit, Decoufle, & Holtzman, 2000 ). As a result, more than half a million infants per year are prenatally exposed to maternal smoking ( CDC, 2004 ; Smith, Martin, & Ventura, 1999 ). Offspring of women who smoke during pregnancy show low birth weight (e.g., Ricketts, Murray, & Schwalberg, 2005 ), increased risk of stillbirth (e.g., Salihu et al., 2008 ), altered cardiorespiratory responses (e.g., Huang et al., 2006 ; Neff, Simmens, Evans & Mendelowicz, 2004 ), and increased asthma and wheezing (e.g., Gilliland, Li, & Peters, 2001 ; Janson, 2004 ; Stocks & Dezateux, 2003 ) as well as behavioral abnormalities, including increased evidence of attentional deficits, impaired learning and memory, lowered IQ, and cognitive dysfunction ( DiFranza & Lew, 1995 ; Levin & Slotkin, 1998 ; Naeye & Peters, 1984 ; Rantakallio & Koiranen, 1987 ; Roy, Seidler & Slotkin, 2002 ; Wakschlag, Lahey, Loeber, Green, Gordon & Leventhal, 1997 ). Despite this large literature suggesting undesirable outcomes in children exposed to MSDP, the underlying biological processes in humans are not well understood. Moreover, the evidence suggesting causal effects of MSDP for these childhood outcomes is muddied in the existing literature due to the frequent inability to separate prenatal tobacco exposure effects from other confounding environmental and genetic factors. Specifically, the vast majority of existing studies provide only limited control for the fact that prenatal exposures may be correlated with parental behaviors that could act as more proximal risk factors that are in turn transmitted to their offspring. Failure to control for such (possibly heritable) confounding factors may account for a large part of the suggested associations between MSDP and offspring outcomes. For example, if mothers with ADHD more commonly smoke during pregnancy, and also confer increased child risk of ADHD via genetic transmission, the observed correlation between MSDP and childhood ADHD would be largely spurious, with limited etiological relevance. Genetically-sensitive study designs can begin to elucidate the likely complex factors contributing to the association between MSDP and child outcomes.
The outcomes associated with MSDP cover broad cognitive and behavioral domains such that a comprehensive review is beyond the scope of this report. Thus, a brief overview of the pertinent literature is provided below and will draw on two bodies of work: (i) animal models of prenatal nicotine, and (ii) human studies of maternal smoking during pregnancy (see Tables 1 and and2). 2 ). The final section will focus on a review of the few genetically informative studies of MSDP. These will be presented within the context of descriptions of a number of different behavior genetic designs that can be used to study the influence of genetic and environmental factors associated with specific measures of the environment.
Selected review of animal models of prenatal nicotine exposure
| Authors and Publication date | Prospective or Retrospective | Populations species and size | Measures | Brief results |
|---|---|---|---|---|
| Daily 0.1 ml subcutaneous injection of nicotine during pregnancy | Swiss-Webster strain mice | Post-natal body weight, latency to eye-opening, latency to the appearance of body hair, and sensory motor reflexes | Exposure resulted in reduced weight gain and delays in development. Exposure also resulted in increased motor activity into early adulthood. | |
| 175 ug/0.9 ul/hr for 7 days using minipumps | Sprague Dawley rats | H nicotine binding | Exposure resulted in increased Bmax values for H nicotine binding | |
| Nicotine 3mg/kg twice daily throughout pregnancy | Duncan –Hartley “English short hair” guinea pigs n = 10 nicotine treated and 10 saline treated | Spontaneous alternation, response to novel alley, black-white discrimination, and reversal beginning | Nicotine exposure resulted in impaired alternating, novel alley entry, and discrimination, and reversal. | |
| Twice daily doses of 0.5, 1.5, or 2.5 mg/kg nicotine throughout gestation | Duncan –Hartley “English short hair” guinea pigs n = 15 in each group | Spontaneous alternation, response to novel alley | Prenatal nicotine treatment resulted in deficits in learned and innate behaviors | |
| 3.4 mg/day of nicotine for 3 weeks and then one week of withdrawal before assessment | Young adult male Sprague Dawley rats | Radial arm maze | Evidence of nicotine-induced cognitive facilitation at least 4 weeks after withdrawal that is not dependent on state dependent learning, nicotinic or muscarinic responsivity | |
| 0.7 mg/kg nicotine | Sprague-Dawley rat pups | Adult cortical function and auditory learning | Nicotine exposure impairs nicotine regulation of cortical function and auditory learning | |
| 3.0 mg/kg twice daily or hypoxic episodes | Female rats derived from HOT: Holtzman stock | Appetitive schedules | Nicotine exposed and hypoxic offspring performed more poorly on fixed ratio, variable interval discrimination, and discrimination reversal schedules | |
| Free choice of nicotine/saccharin or pure water (controls had free choice of saccharin or pure water) | Offspring of C57BL/6J dams | Spontaneous locomotion, fear associated learning, addictive and depression related behaviors | Nicotine exposure associated with increased spontaneous locomotion, preference for cocaine-associated place, and latency to escape in a learned helplessness paradigm | |
| 1.5 and 3 mg/kg/day of nicotine | Fischer rats | Maternal weight gain, birth weight, righting reflex, temperature regulation, adherence to screen, organ/body weight ratios, maze testing, brain protein content | Nicotine treatment led to deficits in maternal weight gain, birth weight, righting reflex, temperature regulation, adherence to screen, organ/body weight ratios, maze testing, brain protein content | |
| 3 mg/kg twice daily | Sprague Dawley rats | H nicotine binding | Exposure resulted in elevated binding on gestational day 18 | |
| 6 mg/kg/day throughout gestation by minipump | Sprague Dawley rats | Adenylate cyclase activity in brain | Nicotine exposure alters adenylate cyclase activity differently by brain region and neurotransmitter systems | |
| Exposure to 6.0 mg/kg/day of nicotine in drinking water | Sprague Dawley rats n = 12 mothers | Radial arm maze performance | Nicotine exposure resulted in poorer performance on radial arm maze | |
| Exposure to saline or 2.0 mg/kg/h nicotine during last half of pregnancy | Offspring of C3H/21bg crosses and C57BL crosses | H nicotine binding | Increased maternal nicotine binding sites but not alpha-I bungarotoxin binding sites | |
| Exposure to 1.5mg/kg nicotine twice daily on gestation days 9–18 (PreN) or daily SC injections on postnatal days 2–21 (NeoN) | HS/Ibg mice PreN = 12 NeoN = 8 Controls = 18 | 8-Arm maze, Morris water maze, Muscarinic receptor binding | Early or neonatal nicotine exposure decreased performance on the behavioral tasks and increased the number of muscarinic receptors in the hippocampus | |
Selected human models of maternal smoking during pregnancy (MSDP).
| Authors and Publication date | Prospective or Retrospective | Populations species and size | Measures | Brief results |
|---|---|---|---|---|
| | ||||
| Prospective – maternal smoking data collected “during pregnancy” | Dyads recruited from the NICHD Neonatal Research Network n = 9637 mother-infant dyads | Birth weight, intrauterine growth restriction | Maternal smoking associated with lower birth weight and intrauterine growth restriction | |
| Retrospective – maternal smoking data collected the 3 or 4 day after delivery | Nationwide multicenter obstetric/pediatric survey in Italy n = 12,987 babies | Birth weight | Maternal smoking associated with lower birth weight in dose response relationship | |
| Prospective – maternal smoking data collected at 36 weeks | Mother giving birth in one of two South Wales towns n = 1159 mothers | Maternal weight gain, fetal growth | Maternal smoking associated in a dose-response fashion with lower maternal weight gain, and lower birth weights, length, and head circumference | |
| Unclear – presumably retrospective | Children of twins from two twin samples | Birth weight | Smoking during pregnancy covaries with offspring birth weight through a direct environmental pathway rather than genetic or shared environmental factors. | |
| Retrospective – maternal smoking data collected in mothers of twins aged 11–19 years | Adolescent female twins pairs n = 1936 twin pairs | DSM-IV ADHD, low birth weight | No evidence for maternal smoking effects on ADHD status when covariates included. Maternal smoking associated with low birth weight. | |
| Prospective – maternal smoking data collected at first antenatal visit (usually before week 15) | All singleton births in two Swedish hospitals were examined for very preterm birth n = 295 | Very preterm births (between 22 and 32 weeks) | Dose dependent association between maternal smoking and very preterm birth. | |
| Prospective – maternal smoking data collected at first antenatal visit (typically weeks 8–12) | All cases in Swedish Medical Birth Registry between 1983 and 1997 with cleft palate or cleft lip excluding multiple births, immigrants, those with missing smoking data and recurrent cleft births and controls n = 872 with cleft palate, 678 with isolated cleft palate, 1456 with cleft lip and 1175 with isolated cleft lip 10% of 128,688 noncleft births used as controls | Cleft lip and cleft palate | Cleft palate associated with MSDP using multiple designs, cleft lip associated with MSDP only with case-control design | |
| Prospective – Maternal smoking data collected at 11–25 weeks of gestation | Mothers and offspring from Danish National Cohort n = 76,768 births | Congenital malformations using EUROCAT criteria | No association of congenital malformations with maternal smoking, children born to nonsmokers using nicotine substitutes had increased congenital malformations (especially musculoskeletal malformations) | |
| Prospective-“during pregnancy” | Participants in the Collaborative Perinatal Project n = 53,518 pregnancies at 12 hospitals in the United States | Birth weight, placental health, and length of pregnancy | Maternal smoking associated with lower birth weight, poorer placental health, and shorter pregnancies | |
| Prospective-100ug/kg body weight over 20 minutes during pregnancy | 8 pregnant rhesus monkeys near term | Uterine arterial blood flow | Nicotine decreases uterine arterial blood flow | |
| | ||||
| Prospective –maternal smoking data collected at first antenatal visit, 32 week of gestation, and after birth | Offspring of women enrolled in the Port Pirie Cohort Study n = 548 children followed from birth to 4 years | Bayley Scales of Infant Development at 2 years and McCarthy Scales of Children’s Abilities at 4 years | No significant association between maternal smoking and neuropsychological development | |
| Retrospective – maternal smoking data collected to mother’s of 2 year olds | Community sample of 99 toddlers and mothers. 52 smoked during pregnancy, 47 did not smoke during pregnancy | Negativity as measured by impulsivity, risk taking, and rebelliousness | Maternal smoking during pregnancy was associated with negativity | |
| Prospective – maternal smoking data collected in each trimester | Children aged 2–8 days that had participated in the Ottawa Prenatal Prospective Study n = 250 babies | Brazelton Neonatal Behavioral Assessment Scale | Maternal smoking associated with increased tremors and poorer auditory habituation | |
| Prospective – maternal smoking data collected in each trimester | Children aged 13 months days that had participated in the Ottawa Prenatal Prospective Study n = 84 offspring | Bayley Mental and Motor Scales | Maternal smoking associated with decreased motor scores, poorer verbal comprehension, and poorer fine motor skills | |
| Unclear- maternal smoking data collected within 48 hours of birth | Smoking exposed neonates and controls taken from a larger study on early language development n = 8 exposed and 8 unexposed neonates | Newborn speech discrimination ability measured by event-related potentials | Infants of smoking mothers discriminated fewer syllables and began the discrimination process 150 ms later than matched controls. | |
| Prospective –self report and biomarkers used during pregnancy | 27 nicotine exposed and 29 unexposed infants n = 56 mother offspring pairs | NICU Network Neurobehavioral Scale within 48 hours of birth | Offspring of mothers who smoked were excitable, showed greater asymmetrical reflexes and more hypertonia than unexposed infants. Exposed infants also had higher scores on the stress abstinence scale in a dose response fashion. | |
| Prospective – Maternal smoking behavior reported at 16 weeks gestation | Singleton infants without disability n = 1871 | Babbling abilities at home visit lasting 1–1.5 hours | Trend of dose response relationship between maternal smoking during pregnancy and the likelihood of being a nonbabbling infant was found. | |
| Unclear when assessed | 15 smokers and 17 nonsmokers | Brazelton Neonatal Behavioural Assessment Scale | Prenatally exposed children exhibited decrements to bell, and deficits in inanimate orientation (auditory), animate orientation (auditory), and consolability | |
| Prospective – maternal smoking data collected during pregnancy | Subsamples of the Groningen Perinatal Project were identified and re-examined at age 5.5 – 11 years n = 1186 singleton births | Reading spelling and arithmetic and parent and teacher ratings of attention and level of internalizing/externalizing | Children of mothers who smoked showed increased attention problems, externalizing behavior, and did worse on arithmetic and spelling tasks | |
| Prospective – maternal smoking data collected “during pregnancy” | 9–11-year old children who participated in the Child Health and Development Studies n = varies between 1745 and 3260 by longitudinal timepoint | Peabody Picture Vocabulary Test, Raven Coloured Progressive Matrices Test, Goodenough-Jarrios Drawing Test, and Quick test | Offspring of mothers who smoked during pregnancy but quit afterwards did not differ on language and matrices tasks from offspring not exposed to smoking during pregnancy. However, children exposed to MSDP and smoking after pregnancy scored lower than either group. | |
| Retrospective – maternal smoking data collected to mother’s of 2 year olds | Community sample of 99 toddlers and mothers. 52 smoked during pregnancy, 47 did not smoke during pregnancy | Negativity as measured by impulsivity, risk taking, and rebelliousness | Maternal smoking during pregnancy was associated with negativity | |
| Prospective – maternal smoking data collected from fourth prenatal month | 10 year old children n = 593 mother/offspring pairs | Wide Range Assessment of Memory and Learning Screening, Wisconsin Card Sorting Task, Stroop and Trail making Tasks, Pediatric Assessment of Cognitive Efficiency, Grooved Pegboard. | Maternal smoking associated with deficits in verbal learning, design memory, eye hand coordination, flexible problem solving, and increases in impulsivity | |
| Mixed- mothers were initially assessed on smoking during pregnancy 4 years after entry into the study; mothers who had a child within these four years would be retrospective whereas those giving birth later would be prospective | Females from the National Longitudinal Survey on Youth and their children n = 11, 192 children form 4886 mothers | Behavioral Problem Index | Although smoking during pregnancy was associated in a dose response fashion with offspring conduct problems, oppositional defiant problems or attention deficit hyperactivity problems (especially in males), the relationship between conduct problems and oppositional defiant problems were not found when examined in siblings that differed in exposure to prenatal nicotine suggesting the possibility of environmental effects that vary between families confound this relationship. | |
| Eskanazi & Trupin, (1995) | Prospective – maternal smoking data collected “during pregnancy” | 5-year old children who participated in the Child Health and Development Studies n = 2,124 | Peabody Picture Vocabulary Test, Raven Coloured Progressive Matrices Test and a behavioral rating scale | Maternal smoking during pregnancy was not significantly associated with differences on the neurobehavioral assessment |
| Prospective – maternal smoking data collected in each trimester | Children aged 6 years that had been followed in the Ottawa Prenatal Prospective Study n = 135 60-month old children and 137 72-month old children | McCarthy Scales of Children’s Abilities and Home Observation for Measurement of the Environment (only a subset) | Maternal smoking associated with impaired cognitive and receptive language scores at both 60 and 72 months | |
| Prospective – maternal smoking data collected in each trimester | Children aged 6 years that had been followed in the Ottawa Prenatal Prospective Study n = 126 children | The Gordon Diagnostic System, sustained attention, The Sentence Memory Test, McCarthy Scales of Children’s Abilities, Target Test and Conners Parent Rating Scale | Maternal smoking was associated with poorer performance on memory tasks (in particular those with verbal recall) | |
| Retrospective – maternal smoking data collected when offspring were 13–21 years old. | Australian female twin pairs where at least one twin had a history of alcohol abuse or dependence (DSM-IV) and at least one twin had children between ages of 13–21 n = 536 twin mothers (268 pairs) and 922 children | Child ADHD assessed with items from Diagnostic Interview for Children and Adolescents and Semi-Structured Assessment of the Genetics of Alcoholism | MSDP associated with offspring ADHD but children-of-twin design suggests genetic transmission of risk for ADHD is not fully explained by MSDP | |
| Prospective – maternal smoking data collection begun at first antenatal visit | 15 year old males and females n = 400,000 | Educational achievement: grade point summary score | Maternal smoking associated with increased risk of poor scholastic achievement OR = 1.59 for 1–9 cigarettes daily, OR =1.92 for 10+ cigarettes daily. However, within mother comparisons suggested that siblings exposed to MSDP also at increased risk for poor school performance. | |
| Prospective – maternal smoking data collected in each trimester | Children between the ages of 6 and 9 years that had been followed in the Ottawa Prenatal Prospective Study N = 91 children | Test battery including Sound blending, Pegboard test, Conners parent Questionnaire, Developmental Drawings test. Peabody Picture Vocabulary Test, Wide Range, Achievement Test, and Hand Dominance | Maternal smoking associated with poorer performance on tests of speech and language skills, intelligence, visual/spatial abilities, and maternal rating of behavior | |
| Retrospective-maternal smoking data collected one year after twins’ birth | High risk twin pairs at age 5 with 18-month follow-up n = 1116 twin pairs | Conduct problems on Achenbach instruments | Prenatal smoking associated with children conduct problems at age 5 and 7 years with dose response relationship for light, moderate and heavy smokers. However, once antisocial behavior in both parents, maternal depression, familial disadvantage, and genetic influences were controlled for, the effects of MSDP were substantially reduced. | |
| Prospective – assessed in each trimester | Children aged 6–11 years that had participated in the Ottawa Prenatal Prospective Study n = 110 children | Central auditory processing task (SCAN) | Prenatal exposure associated with poorer performance on SCAN | |
| Retrospective-maternal smoking data collected shortly after birth | 2–3 year old twin pairs from the Netherlands Twin Register n = 377 twin pairs | Child Behavior Checklist | Association of MSDP with externalizing (especially aggression) but not internalizing behaviors | |
| Prospective-maternal smoking data collected to gain entry into smoking cessation study | 3 year old offspring of mothers who smoked during pregnancy n = 366 offspring | McCarthy Scales of Children’s Abilities and Minnesota Child Development Inventory (MCDI) | Offspring of mothers who quit smoking showed higher scores on the General Cognitive Index of the McCarthy Scales and MCDI scores compared with offspring of mothers who did not quit smoking | |
| Retrospective – maternal smoking data collected when offspring were aged 8–16 years | Twins from the Virginia Twin Study of Adolescent Behavioral Development n = 1413 families | Conduct disturbance and smoking behavior form the Child and Adolescent Psychiatric assessment | Conduct disorder related to some other variable than smoking during pregnancy | |
| | ||||
| Prospective – mother reported smoking behavior in third trimester | Birth cohort of 4169 males at age 34 years | Adult criminal outcomes | Dose response relationship between maternal smoking behavior and arrests for nonviolent and violent crimes (especially persistent criminal behavior. | |
| Prospective – maternal smoking behavior assessed at each prenatal visit | Offspring from the National Collaborative Perinatal Project n = 1248 | DSM III nicotine and marijuana dependence | Offspring of mothers who smoked during pregnancy were more likely to develop nicotine but not marijuana dependence compared with unexposed offspring | |
| Prospective – maternal smoking data collected at fourth and seventh prenatal visit and at delivery | Low-SES birth cohort randomly sampled from an urban prenatal clinic n = 589 10-year olds | Questions about use of tobacco, alcohol and marijuana | Association of MSDP and early tobacco experimentation in preadolescence | |
| Retrospective – maternal smoking data collected at birth | 18 year old offspring followed in a longitudinal study n = 1022 children | Mental health problems measured with Composite International Diagnostic Interview at age 18 | Offspring of mothers who smoked were more likely to have higher psychiatric symptom rates for conduct disorder, alcohol and substance abuse and depression | |
| Retrospective – maternal smoking data collected at 4-year follow-up | Siblings of ADHD and non-ADHD probands n = 266 | DSM III- R | Maternal smoking associated with ADHD, | |
| Retrospective – maternal smoking data collected at 4-year follow-up | Siblings of ADHD and non-ADHD probands n = 266 | DSM III- R | Maternal smoking associated with ADHD, CD, Major Depression and drug abuse | |
| Retrospective- Maternal smoking data collected from mothers of ADHD probands and non-ADHD comparison probands (mean age of 13) | High risk-siblings ascertained through probands of ADHD and non-ADHD controls n = 174 siblings of ADHD probands and 129 siblings of non-ADHD probands | DSM III-R diagnosis of ADHD | Association of MSDP and ADHD. Association also found after controlling for SES, parental IQ, and parental ADHD status | |
| Mixed - maternal smoking data collected during and in first few days after pregnancy | Assessment at mean age of 18.7 n = 3044 singleton males | Intelligence assessed by Børge Priens Prøve. | Negative association of maternal smoking with intelligence in a dose-response relationship for five levels of maternal smoking. | |
| Unclear – appears to be prospective | Men from a general population cohort n = 5636 | Criminal records | Maternal smoking associated with violent and persistent (but not nonviolent) offenses. | |
| Retrospective – maternal smoking data collected when offspring were aged 7–12 years | A longitudinal study of boys referred to one of two university outpatient clinics n = 177 boys | DSM-III-R using the Diagnostic Interview Schedule | Maternal smoking associated with conduct disorder | |
| Retrospective-maternal smoking data collected when offspring were 2.5–5.5 years old | Preschoolers referred to Preschool Behavior Problems Clinic and controls n = 79 referred to clinic and 52 controls recruited from a general pediatric clinic | DSM-IV symptoms of ODD and CD assessed using K-SADS | Disruptive behavior disorder symptoms were associated with prenatal exposure to cigarettes | |
| Retrospective – maternal smoking data collected when offspring were between 6 and 23 years old | Mother offspring pairs selected for presence or absence of lifetime history of major depression in parents N = 147 offspring whose mothers reported either never smoking during pregnancy (97) or 10+ cigarettes daily during pregnancy (50) | Schedule for Affective Disorders and Schizophrenia-Lifetime version (SADS-LA) and Peabody Picture Vocabulary Test (PPVT) | Male offspring of mothers who smoked during pregnancy had more than a 3-fold increased lifetime risk of conduct disorder, female offspring of mothers who smoked during pregnancy had a more than 5-fold increased risk of drug abuse/dependence | |

Animal models: the role and mode of action of prenatal nicotine
Animal models tend to show the most consistent support of the effects, as well as the mode of action, of prenatal nicotine, which is just one toxic component of cigarettes. Importantly, animal studies do pinpoint nicotine, which partially mimics the actions of acetylcholine, as a neuroteratogen ( Slikker, Xu, Levin & Slotkin, 2005 ). The major outcome variables examined in prenatally exposed animals include birth weight, locomotor activity, and cognitive performance.
Birth weight
Similar to results in humans (e.g., Eskenazi, Prehn, & Christianson, 1995 ; Ricketts et al., 2005 ), findings in rats consistently show lower birth weight in offspring exposed to prenatal nicotine when compared with nonexposed offspring (see Ernst, Moolchan & Robinson, 2001 for review). Although prenatally exposed mice do not exhibit significantly lower birth weight, pups born to nicotine-administered dams show a significantly slower rate in postnatal weight gain ( Ajarem & Ahmad, 1998 ). These findings are of importance since, in humans, low birth weight has been shown to be associated with long-term cognitive deficits and ADHD (e.g., Botting, Powls, Cooke & Marlow, 1997 ; Bresleau & Chilcoat, 2000).
Locomotor activity and cognitive function
In general, animal studies tend to show increased locomotor activity in offspring who have been exposed to nicotine prenatally (see Ernst et al., 2001 for review). Studies in rats and mice have reported cognitive impairment, such as attention and memory deficits in various maze tasks, associated with prenatal nicotine exposure ( Levin, Briggs, Christopher & Rose, 1993 ; Liang, Poytress, Chen, Leslie, Weinberger & Metherate, 2006 ; Martin & Becker, 1971 ; Paz, Barsness, Martenson, Tanner & Allen., 2006 ; Peters & Ngan, 1982 ; Sorenson, Raskin & Suh, 1991 ; Yanai, Pick, Rogel-Fuchs, Zahalka, 1992 ). Mild deficits in learning have also been reported in rats (e.g., Liang et al., 2006 ; Martin & Becker, 1971 ), mice (e.g., Paz et al., 2006 ) and guinea pigs (e.g., Johns, Louis, Becker & Means, 1982 ; Johns, Walters & Zimmerman, 1993 ). These impairments in attention, memory, and learning are consistent with the cognitive deficits found in children diagnosed with, for example, ADHD ( Ernst et al., 2001 ). It has also been hypothesized that the observed deficits in operant learning found in animals, might translate to, and be associated with, dysfunction in reward or motivational processes, which could also predispose to substance abuse ( Ernst et al., 2001 ).
Hypothesized mode of action (for more detail see Ernst et al., 2001 ; Slikker et al., 2005 ; Shea & Steiner, 2008 )
Prenatal exposure to nicotine evokes a spectrum of effects by discoordinating the timing of trophic events linked to a subset of cholinergic receptors, specifically nicotinic cholinergic receptors (nAChRs), present very early in the developing brain of rodents (embryonic day 10) and humans (4–5 weeks of gestation) ( Hellstrom-Lindahl, Seiger, Kjaeldgaard & Nordberg, 2001 ; Levin & Slotkin, 1998 ; Slikker et al., 2005 ; Slotkin, 1998 ; Slotkin, 1999 ; Slotkin, McCook, Lappi & Seidler, 1992 ; Slotkin, Orband-Miller & Queen, 1987 ). Once nicotine enters the fetal bloodstream it binds to nAChRs, which are found in the central and peripheral nervous system and can be found both postsynaptically (e.g., acetylcholine neurotransmission) and presynaptically influencing the release of other neurotransmitters ( Dani, 2001 ).
nAChRs are ligand-gated channels including five subunits, usually made of two alpha (a) and three beta (B) subunits. Several nAChR subtypes (or combinations of subunits) exist, each of which has a specific pharmacology, physiology, and anatomical distribution ( Pakkanen, Jokitalo & Tuominen, 2005 ). The two most abundant subtypes in vertebrate brain are: (i) α4, β2 combination, and (ii) α7. The different subtypes have important functional implications, particularly during development, as their relative distribution in the brain varies with developmental stage and age ( Ernst et al., 2001 ). nAChRs are significantly involved in brain development via promotion of cell division during gastrulation and subsequent promotion of the switch from cell replication to cell differentiation in terminal neuronal differentiation ( Shea & Steiner, 2008 ). The presence of these receptors in early embryogenesis ( Hagino & Lee, 1985 ) suggests that nicotinic signaling may be an important part of neural development. Reported changes in receptor density during normal development (e.g., high levels found at early gestation) might also imply windows of vulnerability to exogenous nicotine. In humans, periods of high density have been found in the frontal cortex, hippocampus, cerebellum, and brainstem during mid-gestation and neonatal periods ( Hellstrom-Lindahl, Gorbounova, Seiger, Mousavi & Nordberg., 1998 ; Hellstrom-Lindahl et al, 2001 ; Huizink & Mulder, 2006 ).
In the rat (e.g., Slotkin et al., 1987 ), and to a lesser extent in the mouse ( Van de Kamp & Collins, 1994 ), binding to the nAChR during development, whether during prenatal or early postnatal stages, is a necessary and key step leading to the adverse effects of nicotine. Several studies indicate that chronic prenatal nicotine exposure in rats and mice results in increased receptor density of fetal and neonatal cerebral nAChRs (for example, Slotkin, 1998 ; Van de Kamp & Collins, 1994 ). Upregulation of the nAChRs during development is conclusive evidence that the cell has experienced chronic nicotinic stimulation. The long-term effects of this up-regulation remain unclear ( Ernst et al., 2001 ); although the proposed mode of action suggests that this stimulation results in premature onset of cell differentiation, at the expense of replication, leading to (i) brain cell death, (ii) structural changes in regional brain areas, and (iii) altered neurotransmitter systems (i.e., acetylcholine, norephinephrine, epinephrine, dopamine, serotonin, as well as glutamate and gamma-aminobutyric acid; Shea & Steiner, 2008 ; Slikker et al., 2005 ). Such alterations could translate to physical deficits, such as impaired cardiac function associated with hypoxia, as well as deficits in later learning, memory, behavior, and development. Differences in developmental profiles of receptor binding between species and strains suggest that genetic factors regulate the maturation of the nicotinic receptor ( Van de Kamp & Collins, 1994 ). These genetic factors may explain interindividual differences in sensitivity to the effects of in utero exposure to nicotine ( Ernst et al., 2001 ).
There is no question that animal work is vital to the study of human problems; however the rat brain, for example, is obviously different from the human brain. Effects of MSDP in humans, for example, often show up in higher-level cognitive (executive) function, which are controlled by the prefrontal cortex. Functional and structural differences in the region of rat brain traditionally considered homologous to the dorsolateral prefrontal cortex in primates suggest that the rat may not have an equivalent region ( Preuss, 1995 ). Moreover, in humans, MSDP results in fetal exposure not only to nicotine, but to a large amount of other toxic components, such as carbon monoxide, ammonia, nitrogen oxide, lead, and other metals ( Huizink & Mulder, 2006 ). Thus, one should not limit the effects of MSDP in humans to nicotine alone. Importantly, while we can use the evidence of negative effects of prenatal nicotine exposure that we garner from animal work as a guide to narrow our focus on potential effects in humans, we cannot directly extrapolate from animal findings to the complex human condition.
Maternal smoking during pregnancy: A more complicated story
As suggested earlier, the evidence for deleterious effects of MSDP on behavior and cognition later in life in human studies is muddied in the existing literature due to the inability to separate these effects from other confounding environmental and genetic factors. In a methodological review of the literature on effects of MSDP, Ramsay and Reynolds (2000) suggest that women who smoke during pregnancy may possess a constellation of personality traits that distinguishes them from other women. They focus on traits such as (i) increased depression and thus decreased motivation to quit smoking during pregnancy (Depression-Compulsivity model), (ii) elevated antisocial traits and thus reduced awareness of their consequences of MSDP as well as reduced concern for others (Antisocial model), and (iii) reduced attention to her own and, by extension, her infant’s nutrition and general well-being (Self-Care model). Thus, the personality of pregnant smokers may reflect a familial vulnerability for later disorders. Ernst and colleagues (2001) go on to outline numerous potential confounds, which include those suggested by Ramsay and Reynolds (2000) , as well as others: (1) parental characteristics: including IQ, psychiatric history (e.g., ADHD, antisocial personality disorder, substance abuse) and parenting; (2) maternal characteristics (e.g. health, height and weight (affecting metabolism of tobacco by-products)); and (3) smoking characteristics: intensity, gestational age at consumption ( Ernst et al., 2001 ). Importantly, a number of these confounds can be controlled for via alternative genetically sensitive designs. However, there is a surprising lack of comprehensive examination of the effects of MSDP within a genetically-informative framework. Specifically, the joint roles of environmental factors (e.g., MSDP) and genetic transmission in the risk for deficits, such as behavioral, learning, and cognitive dysfunction, are downplayed and there is a lack of control for differences between women who smoke during pregnancy and those who do not.
Neurobehavioral and cognitive effects of MSDP in humans
The offspring outcomes associated with MSDP cover broad cognitive and behavioral domains and are outlined thoroughly in several well laid-out and comprehensive reviews of the effects of MSDP (see Cnattingius, 2004 ; Ernst et al., 2001 ; Huizink & Mulder, 2006 ; Linnet et al., 2003 ; Shea & Steiner, 2008 ). These reviews are presented primarily from the phenotypic association point of view and say very little about how genetic factors may influence the reported associations between MSDP and offspring outcome. The main points of these reviews are presented briefly in this section, along with results from a few recent studies. The scope of results concerning the negative impact of MSDP, both suggestive and inconclusive, are presented. What is clear from these reviews is the need for more comprehensive study design as well as the lack of genetically informed studies on MSDP. The few studies that have considered genetic effects are reviewed in the final section of this report.
Pregnancy and birth outcomes
Epidemiological evidence from prospective and case-control studies show relatively high consistency for the association of adverse pregnancy outcomes (i.e., fetal growth restriction, hypoxia and placental effects, stillbirth, sudden infant death syndrome, etc) with MSDP (see Cnattingius, 2004 for detailed review ; Ernst et al., 2001 ); however, neurobehavioral outcomes have shown less consistency, indicating the potential need for more sensitive sampling designs and strategies.
MSDP is reported to increase rates of spontaneous abortion, stillbirth, sudden infant death syndrome, cleft palate, and most relevant to long-term neurobehavioral effects, preterm birth and low birth weight ( Bada et al., 2005 ; Conter, Cortinovis, Rogari & Riva, 1995 ; DiFranza & Lew, 1985; D’Onofrio et al, 2003 ; Ernst et al., 2001 ; Knopik et al., 2005 ; Kyrklund-Blomberg, Granath & Cnattinguis, 2005 ; Levin & Slotkin, 1998 ; Meyer, Williams, Hernandez-Diaz & Cnattinguis, 2004 ; Salihu, Aliyu & Kirby, 2006 ; Salihu et al., 2008 ; Sastry, 1991 ). Recent evidence also suggests that offspring of nonsmokers who used nicotine substitutes during pregnancy are at increased risk for congenital malformations ( Morales-Suarez-Varela, Bille, Christiansen & Olson, 2006 ).
These outcomes reported to be associated with prenatal exposure may be indirect or direct toxic consequences of MSDP. Nicotine produces anorexigenic, hypoxic, vascular, and placental effects that can adversely affect fetal development ( Cnattingius, 2004 ; Ernst et al., 2001 ). Existing theories focus on (i) maternal and fetal undernutrition due to the acute anorexigen effects of tobacco smoking ( Davies & Abernethy, 1976 ; Perkins, Sexton, DiMarco & Fonte, 1994 ); (ii) intrauterine hypoxia secondary to increased carbon monoxide and dioxide, reduced blood flow, and inhibition of respiratory enzymes ( Abel, 1980 , 1984 ; Byrd & Howard, 1995 ); (iii) disruption of the function of the placenta ( Huizink & Mulder, 2006 ; Naeye, 1978 ; Sastry, 1991 ; Suzuki, Minei & Johnson, 1980 ) via nicotinic activation of placental cholinergic systems which depresses transplacental amino acid transport, which may contribute to intrauterine growth retardation ( Cnattingius, 2004 ; Ernst et al., 2001 ). Thus, prenatal exposure may have direct teratogenic effects on the fetus leading to more readily observed adverse phenotypes; however, these effects most likely depend on the specific outcome measure of interest ( D’Onofrio et al., 2003 ).
Infant and Toddler outcomes
The evidence for effects of MSDP on infant and toddler outcomes has been overall, inconsistent, perhaps due to the possibility that a certain level of brain maturation needs to be achieved before deficits become detectable ( Ernst et al., 2001 ; Huizink & Mulder, 2006 ). The inconsistency may also be due to less sensitive assessment tools for this age group. Data showing negative effects of MSDP suggest deficits in speech processing ability ( Key, Ferguson, Molfese, Peach, Lehman & Molfese, 2006 ), decreased scores in motor ability and verbal comprehension ( Gusella & Fried, 1984 ), reduced auditory acuity ( Saxton, 1978 ), increased hypotonicity, heightened tremors and startles ( Fried & Makin, 1987 ), and negative affect ( Brook, Brook & Whiteman, 2000 ) among infants who were prenatally exposed to nicotine. Since it has been shown that adverse birth outcome, such as preterm birth, is related to neurologic and developmental disabilities during the first two years of life ( Marlow, Wolke, Bracewell, Samara & EPI Cure Study Group, 2005 ), a recent study ( Law, Stroud, LaGasse, Niaura, Liu & Lester, 2003 ) adjusted their findings for factors relating to birth outcome and still found that newborns exposed to MSDP were more excitable and hypotonic and showed more stress/abstinence signs on a standard neurobehavioral assessment. Not all studies have found significantly negative relationships however. For instance, Obel, Henriksen, Hedegaard, Secher, and Ostergaard (1998) found mixed results when comparing babbling abilities in prenatally exposed 8-month olds to controls. When comparing nonbabblers to di- and polysyllable babblers, a trend toward a dose-response effect of MSDP was found, with those children exposed to more cigarettes per day showing less babbling ability. However, this trend was nonsignificant when comparing nonpolysyllable babblers to polysyllable babblers. Baghurst, Tong, Woodward, and McMichael (1992) also found no evidence for differences in verbal, perceptual, and motor scores due to prenatal exposure once adjusting for social class, home environment, and mother’s intelligence. Together, these findings suggest the possibility that MSDP is associated with motor, sensory, and cognitive deficits in infants and toddlers, which may indicate a pervasive toxic effect on early neurodevelopment.
Childhood outcomes
Findings in children also seem to support a negative influence of in utero exposure to smoking on behavior and cognitive function; however, there are again some inconsistencies. MSDP has been associated with a significant increase in externalizing (e.g., oppositional, aggressive, overactive) scores but not internalizing behavior ( Brook, Zhang, Rosenberg & Brook, 2006 ; Day, Richardson, Goldschmidt & Cornelius, 2000 ; Orlebeke, Knol, & Verhulst 1999 ). Cognitive function has also been shown to be negatively affected by MSDP, with deficits in sustained attention ( Fried, O’Connell & Watkinson., 1992a ), response inhibition, memory, and impulsivity, overall cognitive function, receptive language ( Fried, Watkinson & Gray, 1992b ), verbal learning and design memory ( Cornelius, Ryan, Day, Goldschmidt & Willford, 2001 ), problem solving ( Cornelius et al., 2001 ), speech and language ( Makin, Fried, & Watkinson, 1991 ), school performance ( Lambe, Hultman, Torrang, MacCabe & Cnattinguis, 2006 ), and auditory processing ( McCartney, Fried & Watkinson, 1994 ). Dose-response relationships, in which the smoking-related relative risk increases with amount smoked, have also been reported for general cognitive ability ( Sexton, Fox & Hebel, 1990 ), arithmetic, and spelling ( Batstra, Hadders-Algra & Neeleman, 2003 ), suggesting the presence of vulnerable periods during fetal development ( Ernst et al., 2001 ).
As with infant and toddler outcomes however, some negative findings are also reported. For example, Bauman, Flewelling and LaPrelle (1991) reported that scores on receptive language and matrices tasks of more than 3000 9–11 yr olds exposed to MSDP but whose mothers quit afterwards, were similar to those of children not exposed to MSDP; however, both of these groups performed better than children exposed to both MSDP and smoking after pregnancy, suggesting the importance of also considering postnatal environment. No clear relationship was observed for MSDP and receptive language scores at 5 yrs or at 15–17 yrs. Eskanazi and Trupin (1995) also found no dose-response relationship of MSDP during the third trimester and cognitive performance in 5 yr olds. Moreover, despite findings of adverse effects of MSDP on school performance using a between family analysis ( Lambe et al., 2006 ), a within-sibling comparison of siblings exposed to differential amounts of MSDP (an example of a case-crossover design which is detailed below) indicated that if a mother had smoked during either pregnancy, both siblings were at increased risk of poor school performance ( Lambe et al., 2006 ); results suggesting that observed associations between MSDP and poor cognitive performance might not be causal.
In one of the most comprehensive analyses to date, D’Onofrio and colleagues (2008) analyzed data from the National Longitudinal Survey of Youth (NLSY), with particular attention to controlling for differences between women who do and do not smoke during pregnancy. They focused their efforts on the association between MSDP and offspring externalizing behavior [conduct (CP), oppositional defiant (ODP), attention deficit hyperactivity (ADHP) problems]. Their comparisons of unrelated children were consistent with the results of previous studies ( Wakschlag, Pickett, Cook, Benowitz & Leventhal, 2002 ) in several respects: (a) CP, ODP, and ADHP were significantly associated with MSDP; (b) each association followed a dose-response relationship; (c) the number of CP demonstrated by children exposed to MSDP was higher for males; and (d) each association remained significant after statistically controlling for associated maternal characteristics. In addition to the use of statistical covariates used in previous studies, D’Onofrio et al. (2008) utilized the clustered nature of NLSY data to account for unmeasured confounds. The hypothesis was that if MSDP caused higher externalizing, the relation would have been evident both when comparing related (e.g. within mothers) and unrelated children (e.g., Rodgers, Cleveland, van den Oord & Rowe, 2000 ). However, similar to Lambe et al. (2006) , when siblings who differed in exposure to MSDP (i.e., none/some vs. more exposure, a broad definition of discordance for MSDP ) were compared, the offspring did not differ significantly with respect to CP or ODP. These results suggest that previous studies found a relationship between MSDP and offspring CP not because MSDP causes increased risk for CP or ODP, but because environmental influences that vary between families confound associations between MSDP and offspring externalizing ( D’Onofrio et al., 2008 ). This finding is consistent with studies that have included more precise measurement of adult characteristics that may confound the relation, such as maternal and paternal antisocial characteristics ( Maughan, Taylor, Caspi, & Moffitt, 2004 ) and maternal delinquency during adolescence ( Silberg et al., 2003 ). It is also generally supportive of a recent children-of-twins study of maternal alcohol use disorder, MSDP and ADHD ( Knopik et al., 2006 ; detailed below).
Adolescent and adult outcomes
Overall, it seems that behavioral and cognitive deficits associated with MSDP continue into late childhood and early adolescence and lead to increased risk for ADHD and Conduct Disorder (CD). MSDP has been associated with ADHD, CD, criminality and substance use (particularly smoking) in adolescence ( Ernst et al., 2001 ). Milberger and colleagues (1996 , 1997 , 1998) investigated MSDP as a risk factor for ADHD and found that 22% of children with ADHD had a history of MSDP, compared with 8% of controls. Significantly lower IQ scores were also found in children exposed to MSDP versus those who were not exposed ( Milberger, Biederman, Faraone & Jones 1998 ). Wakschlag and colleagues (1997 , 2001 , 2002) have consistently shown that MSDP is a robust, independent risk-factor for CD in males. Weissman, Warner, Wickramaratne and Kandel (1999) report similar findings reporting 4-fold increases in CD rates and 5-fold increases in adolescent drug abuse in children exposed to MSDP. Cornelius, Leech, Goldschmidt and Day (2000) and Buka, Shenassa and Niaura (2003) found increased risk for early tobacco experimentation and nicotine dependence, respectively, in children exposed to MSDP. Fergusson, Woodward and Horwood (1998) also suggested that MSDP contributes to children’s risk of later externalizing problems. Children exposed to MSDP had higher psychiatric symptom rate for CD, alcohol abuse, substance abuse, and depression compared with unexposed children. These childhood associations also appear to carry into adulthood. For example, Brennan, Grekin and Mednick (1999) and Rasanen et al. (1999) found relationships between MSDP and later criminality in male offspring up to age 28 and Mortensen, Michaelsen, Sanders and Reinisch (2005) reported a dose-response relationship between MSDP and adult intelligence.
Summary of MSDP in humans
MSDP is associated with offspring behavioral abnormalities, including increased evidence of attentional deficits, impaired learning and memory, lowered IQ, cognitive dysfunction, later childhood conduct problems, substance use, and early adult criminality; however, not all studies have reported a significantly negative relationship between MSDP and offspring outcomes.
What is clear from these reviews, however, is the need for more comprehensive study design in the study of MSDP. In short, there are a paucity of studies investigating gene-environment interplay in the proposed associations between MSDP and subsequent child outcomes. A key approach is to use a combination of strategies, such as twin, children-of-twin, and sibling-control designs, emphasizing both behavioral and molecular genetic methods, to elucidate the likely complex factors contributing to the association between MSDP and child outcomes. Preliminary findings from this work in the area of child externalizing problems ( Maughan et al., 2004 ; Knopik et al., 2006 ; D’Onofrio et al, 2008 ) indicate that, once genetic and environmental effects are accounted for, MSDP accounts for a much smaller effect than proposed by existing literature; however, while the effects were smaller, MSDP continued to be significantly linked to childhood behavior. Such results suggest that MSDP is unlikely to be a unique cause of early childhood behavior problems and illustrate the need for comprehensive study design.
Comprehensive study design – things to consider
The idea of joint roles of genetic and environmental factors can be referred to as gene-environment interplay. This is a broad term that encompasses several different concepts with different meanings and interpretations (see Rutter, Moffitt & Caspi, 2006 for detailed review). While a thorough and comprehensive review of gene-environment interplay is beyond the scope of this report, we will focus briefly on gene-environment interaction (G×E) and gene by environment correlation (rGE). G×E occurs when the effect of environmental exposure is conditional on a person’s genotype ( Moffitt, Caspi & Rutter, 2005 ). An example of G×E is phenylketonuria (PKU), a genetic disorder characterized by deficiency of the enzyme phenylalanine hydroxylase. Children who are homozygous (carry two copies) for a certain form of the phenylalanine hydrolylase gene are deficient in phenylalanine hydroxylase and cannot metabolize phenylalanine in food. Thus, phenylalanine accumulates and damages the developing brain. Phenylalanine has no harmful effects on other children who do not carry this particular genotype. However, PKU is one of the few genetic diseases that can be controlled by diet (an example of an environmental influence). A diet low in phenylalanine can be very effective treatment, yet this low phenylalanine diet has no harmful or beneficial effect on other children. Perhaps the most well-known example of G×E in the development of psychiatric disorders was reported by Caspi et al. (2002) who found that a functional polymorphism in the gene encoding the neurotransmitter-metabolizing enzyme monoamine oxidase A (MAOA) was found to moderate the effect of maltreatment, such that maltreated children with a genotype conferring high levels of MAOA expression were less likely to develop antisocial problems. These findings provided the basis for a growing literature suggesting that genotypes can moderate children’s sensitivity to environmental insults.
rGE can be thought of as genetic control of exposure to the environment or, in other words, an individuals genotype influences the probability of exposure to certain environments ( Caspi & Moffit, 2006 ; D’Onofrio et al., 2003 ; Jaffee & Price, 2007; Kendler & Eaves, 1986 ). rGE has been described as passive, active or evocative (see Jaffee & Price, 2007, for a full review). (i) Passive gene-environment correlation refers to the association between the genotype a child inherits from her parents and the environment in which the child is raised. Parents create a home environment that is influenced by their own heritable characteristics. (ii) Evocative (or reactive) gene-environment correlation happens when individuals are reacted to based on their genetic propensities or, in other words, an individual's (heritable) behavior evokes an environmental response (see Burt, 2008 ). (iii) Active gene-environment correlation occurs when an individual seeks out or creates certain environments based on their genetic propensity. rGE results in “the contamination of measures of environmental exposure with genetic variation and thus clouds interpretation of results” ( Caspi & Moffitt, 2006 , p.587).
One of the main limitations of studying familial and environmental influence and child development is that the parents are providing both the environment and the genes to their offspring ( D’Onofrio et al., 2003 ). In addition to prenatal environment, separate consideration should also be given to environmental exposure to second-hand smoke (see Eskenazi & Castorina, 1999 for review) since children born to smoking mothers are more likely to be exposed to environmental tobacco smoke ( Key et al., 2006 ), which could increase risk of developmental deficits ( Yolton, Dietrich, Auinger, Lanphear & Hornung, 2005 ). Most studies that have considered prenatal nicotine exposure have considered latent genetic variables or have examined the presence of measured G×E by focusing on the dopaminergic system and genes involved in the metabolism of tobacco by-products. These few studies are included in the review below.
Adoption studies
At the time of this report, there have been no adoption studies that have specifically considered maternal smoking during pregnancy; however, two studies outlined in this section have considered prenatal drug exposure more generally ( Crea, Barth, Guo & Brooks, 2008 ; Neiderhiser et al., 2007 ). The lack of adoption studies in this arena does not preclude the potential importance of this design for MSDP. Adoption designs provide a direct way to disentangle genetic and environmental sources of variation. Adoption creates pairs of genetically related individuals who do not share a common family environment (and/or prenatal environment; i.e., biological siblings adopted apart and raised in different homes) and also creates family members who share family environment but who are not genetically related (i.e., non-biologically related children adopted into the same adoptive home). In both situations, any resemblance estimates the contributions of the family environment. A strong suit of the adoption design is the ability to study gene by environment interaction and additional processes through which gene-environment correlation creates the covariance between parents and children ( D’Onofrio et al., 2003 ). However, the adoption design does suffer from certain limitations. First, due to highly selective placement ensuring that the adoptive environment is excellent, there is an inherent difficulty in obtaining samples of children who are exposed to high-risk environments. Moreover, an assumption of this design is that there are no negative consequences of being adopted and that environmental processes operate similarly in adoptive and nonadoptive families ( D’Onofrio et al., 2003 ). Such an assumption is not needed in other genetically sensitive designs.
Crea et al (2008) did not focus on disentangling genetic and environmental influences on behavior per se, but rather examined behavioral trajectories for substance exposed adopted children, fourteen years after adoption. They found that prenatal exposure predicted elevated behavior problems but only slightly higher than those of nonexposed adopted counterparts. The overall rate of change in behavioral problems did not differ between exposed and nonexposed groups. This finding contradicts the argument that substance exposure alone is responsible for triggering a cascade of negative sequelae and encourages the investigation of protective familial environmental factors (e.g., positive rearing environment) that buffer the impact of this exposure ( Crea et al., 2008 ).
In a recent analysis of a sample from the Early Growth and Development Study ( Leve et al., 2007 ), Neiderhiser et al. (2007) examined 350 ‘yoked’ birth mothers, adopted children and adopted parents and 104 birth fathers. The focus was on toddler temperament and behavior problems at 18 months. The authors reported preliminary results suggesting that high levels of prenatal drug use significantly contributed to suppressed toddler affect and effects of genetic risk operated only via prenatal drug exposure ( Neiderhiser et al., 2007 ). Future planned work to extend these analyses in order to facilitate the disaggregation of prenatal exposure, genes (via DNA collection), as well as postnatal rearing environment will lend considerable and potentially important information to the effort to elucidate these complex relationships ( Leve et al., 2007 ).
Twin studies and their extensions
The twin method compares the similarity between identical (monozygotic or MZ) twins and fraternal (dizygotic or DZ) twins (see Plomin, DeFries, McClearn & McGuffin, 2008 for details). If a trait is genetically influenced, MZ twins will be more similar than DZ twins; however, it is also possible that this greater similarity is due to environmental rather than genetic factors. This design can offer considerable knowledge in the genetic etiology of, not only outcomes of interest (e.g., ADHD or cognitive ability), but also risk factors (e.g., MSDP; see Agrawal et al., 2008 for genetic etiology of MSDP; D’Onofrio et al, 2003 , 2008 ;). It can also determine whether genetic effects differ in two environments; however, the models may only partially control for genetic factors since they assume that the specified environments represent ‘true’ or ‘pure’ environmental risk factors which are free from genetic influences (i.e., that there is no gene-environment correlation; Caspi, Taylor, Moffitt & Plomin, 2000 ; D’Onofrio et al., 2003 ; Purcell & Koenen, 2005 ). Classical twin studies, even those that add explicit measures of the environment, are also not able to delineate the processes involved in intergenerational processes ( D’Onofrio et al., 2003 ).
Four recent studies have tested the association between MSDP and ADHD or conduct problems/antisocial behavior within a twin design ( Button, Thapar & McGuffin, 2005 ; Knopik et al., 2005 ; Maughan et al., 2004 ; Thapar et al., 2003 ). As discussed in this section, using a twin design allows the genetic effects that contribute to the outcomes in children to be estimated (see Purcell & Koenen, 2005 for details on limitations involving environmental mediation in the classical twin study). In an examination of conduct problems in 5–7 year old twins, Maughan et al. (2004) report that, once genetic and environmental risks were controlled for, the effects of MSDP were substantially reduced. Thapar et al (2003) found that, in addition to substantial genetic influences on ADHD symptoms, MSDP explains additional variance above and beyond genetic effects. Button et al. (2005) report similar results when considering the covariation between antisocial behavior and ADHD stating that MSDP contributes small but significant amounts to the variance of both phenotypes. Knopik et al. (2005) suggest that prenatal and parental risk factors (e.g., maternal and paternal psychopathology) combine additively with the important genetic risk of developing ADHD, rather than interactively (i.e., no significant findings for G×E interaction). Thus, in summary it appears that, while genetic influences on these ADHD phenotypes are important, MSDP also has an independent effect on ADHD.
An extension of the classical twin study is the bivariate twin study that investigates the relationship between an environmental risk factor (considered as a phenotype) and an outcome of interest. A limitation of this extension is that the bivariate design cannot study all of the possible environmental risk factors that are involved in developmental psychology because the model can only include environments for which twins can differ (i.e., individual-specific environment; Purcell & Koenen, 2005 ). Thus, in the case of exposure to smoking during pregnancy (i.e., an obligatory shared environment in twin offspring exposed prenatally; Purcell & Koenen, 2005 ), this is a design that cannot be used. However, if one is considering the etiology of the behavior of smoking during pregnancy (i.e., twin mothers who can differ in their smoking behaviors), this design can be used to determine the covariation of MSDP and another outcome of interest. For example, Agrawal et al (2008) considered the genetic covariation of maternal smoking during pregnancy and nicotine dependence. Results indicated that women who smoked during an entire pregnancy reported heavier dependence and more unsuccessful quit attempts, compared with a community sample of mothers and with women who smoked during only part of a pregnancy. Educational attainment, weekly church attendance, spousal current smoking, and nicotine dependence also were associated with MSDP. The authors also found that heritable influences, even after adjustment for the above-stated significant psychiatric and sociodemographic covariates, explain nearly half of the variation in MSDP, with the remainder of the variance being due to environmental factors not shared by members of a twin pair. A large proportion of the genetic influences on MSDP were shared with nicotine dependence. These results, though not focused on childhood outcomes of MSDP, do have strong implications for treatment and intervention, in that a lifetime history of difficulty with smoking cessation, in conjunction with social background and psychiatric comorbidity, especially during pregnancy, needs to be considered by treatment providers when counseling expectant mothers about the potential risks of MSDP.
Another expansion of the classical twin study incorporates assessment of the twins’ parents. This design has the ability to estimate environmental effects while controlling for genetic effects on both parents and children ( D’Onofrio et al., 2003 ; Rutter et al., 1997 ). Limitations exist, as outlined in Rutter, Pickles, Murray, and Eaves (2001) . Specifically, the twin-family design requires identical measures for parents and children and also assumes that the same genetic and environmental structure influences both generations ( D’Onofrio et al., 2003 ).
Children-of-twins
The Children-of-Twins (COT) design can begin to elucidate the role that specific environments (such as prenatal exposure) play in the etiology of psychological and behavioral phenomena ( D’Onofrio et al., 2003 ), while studying intergenerational associations with fewer assumptions than the twin-family design. In the case of prenatal exposure, it allows one to begin to disentangle genetic, prenatal exposure, and other environmental effects on offspring outcomes. It also offers the additional advantage of including offspring sibling pairs that may differ in their amounts and/or timing of prenatal exposure (an obligatory shared environment in classical twin studies).
There are several approaches within this design: (i) children of discordant twins, which essentially involves (a) a comparison between the children of affected and unaffected MZ twins, and (b) a subsequent comparison of the rates of the disorder in children of the unaffected MZ and DZ cotwins; (ii) the MZ half-sib design ( Nance, 1976 ; Nance & Corey, 1976 ; Nance, Corey, & Boughman, 1978 ) which is a nested analysis of variance approach to the study of offspring of MZ twin pairs; (iii) a structural equation model fitting approach as outlined in D’Onofrio et al. (2003) which is a variation on the twin-family study and examines (a) within-generation, (b) cross-generation, same-family, and (c) cross-generation, cross-family correlations; and (iv) inferring genetic and environmental risk on offspring outcome from the co-twin’s (parental) history of the phenotype of interest ( Jacob et al., 2003 ; Knopik et al., 2006 ).
The COT design (see Jacob et al., 2003 for general discussion of the method) has been used less often in behavioral genetic studies, and has just recently been expanded to not only assess the potentially complex relationship between parental psychopathology (such as substance dependence) and child behavior, but to also consider the role of prenatal exposure in intergenerational associations ( D’Onofrio et al., 2003 ; Knopik et al., 2006 ). For example, in an attempt to understand the underlying processes associated with MSDP, D’Onofrio et al (2003) used the structural equation model approach within a COT sample to move beyond the straight phenotypic association between MSDP and birth weight. Their results suggested that MSDP appears to have a specific environmental association with offspring birth weight with no apparent confounding by genetic factors, common environment, or other measured covariates ( D’Onofrio et al., 2003 ).
Given evidence that mothers who abuse alcohol, who are alcohol dependent, or who have an alcohol dependent partner are more likely to smoke or drink during pregnancy (e.g., Knopik et al., 2005 ), Knopik et al (2006) used the COT design to examine the relationship between maternal psychopathology (specifically alcohol use disorder, AUD), MSDP, and child ADHD. This approach provides a powerful pseudo-adoption design in which genetic and environmental risk status is inferred from the co-twin’s history of, in this case, AUD. Importantly, children raised by an AUD monozygotic (MZ) or dizygotic (DZ) twin parent are at high risk for psychiatric disorders (e.g., ADHD) and other health problems because of high genetic and high environmental risk. In contrast, children raised by a non-AUD twin of an AUD MZ co-twin are at reduced environmental risk because they have not grown up with a mother with AUD, but these children are at the same (high) genetic risk as children raised by an AUD twin because the mothers have identical genotypes. In turn, children raised by the non-AUD twin of an AUD DZ co-twin are also at reduced (low) environmental risk but at only intermediate genetic risk because DZ twin pairs share on average 50% of their genes.
Thus, in the absence of any environmental effect of maternal AUD, after controlling statistically for psychopathology in the biological parents, the child of an AUD mother should be no more likely to develop ADHD than the child of a non-AUD parent who is the MZ co-twin of an AUD individual. Excess rates of ADHD in children of AUD mothers, after controlling for comorbid psychiatric disorders and pertinent variables, would imply an environmental impact of maternal AUD. Therefore, the COT design is a powerful design to disentangle the genetic and environmental effects on the association between maternal (or paternal) psychopathology and offspring outcome, while also estimating direct effects of measured environmental variables, such as prenatal exposure.
These data ( Knopik et al., 2006 ) yielded a pattern of results consistent with a genetic contribution to the association between maternal AUD and increased offspring risk of ADHD, but also reaffirmed the potential importance of MSDP. Compared to controls, rates of offspring ADHD were significantly elevated not only in families where the mother had a history of AUD, but also in families where the mother had no history of AUD, but had a monozygotic twin sister with AUD. In addition, rates of maternal regular smoking, and maternal regular smoking during pregnancy, were significantly elevated in those mothers who had a history of AUD, and in mothers who were unaffected, but had an affected monozygotic cotwin. This is consistent with a strong genetic correlation between alcoholism and smoking that has been found in other research, and implies a potential confounding of MSDP and genetic risk of alcoholism. Thus, genetic transmission and effects of MSDP are partially confounded. Models predicting ADHD outcome from family risk (of AUD) status, as well as other maternal and paternal psychopathology, indicated that even when maternal genetic risk of AUD and maternal regular smoking were controlled for, heavy MSDP remained a significant and strong predictor of offspring ADHD risk. Thus, while MSDP is likely contributing to the association between maternal AUD and offspring ADHD, the evidence for a significant genetic correlation suggests: (i) pleiotropic genetic effects, with some genes that influence risk of AUD also influencing vulnerability to ADHD; or (ii) ADHD is a direct risk-factor for AUD ( Knopik et al., 2006 ). Thus, these results from the COT design ( D’Onofrio et al., 2003 ; Knopik et al., 2006 ) yielded a pattern of results consistent MSDP having an independent effect on offspring outcomes even after controlling for potential confounders (e.g., genetic transmission, other environmental factors, and other covariates). The ability to begin to disentangle genetic and environmental intergenerational transmission in the domain of MSDP is critical for understanding the magnitude of risk that MSDP carries as this can have real implications for future research, intervention, and prevention efforts.
Cotwin-control
The cotwin-control design is a modification of the traditional case-control design where data is considered from twin pairs that are discordant for (i) the outcome of interest (e.g., ADHD), (ii) a variable related to the outcome of interest (e.g., schizophrenia in a model examining cognitive ability, see Kremen et al., 2006 ; early cannabis use in a model examining drug use as in Lynskey et al., 2003 ), or (iii) a environmental measure. The design controls for effects of age, gestational influences, and genetic factors ( D’Onofrio et al., 2003 ). It can also control for many environmental factors; however, similar to twin studies and as pointed out in D’Onofrio et al. (2003) , it is limited by methodological problems that prohibit the examination of many environmental risk factors that are commonly examined in epidemiological studies such as divorce, parenting practices, parental psychopathology, and MSDP (see D’Onofrio et al., 2003 for detail). The difficulties also lie in finding large enough samples of twins that are discordant for salient environmental factors that are under consideration. Thus, there is typically not enough power to draw definitive and meaningful conclusions ( D’Onofrio et al., 2003 ; Kendler & Gardner, 2001 ).
Case-crossover design
A variation on the cotwin-control study is the case-crossover design (or within-mother between-pregnancy design) which examines siblings discordant for prenatal exposure to MSDP. A form of this design was used in two studies discussed earlier in this report which compared siblings exposed to a broad definition of differential amounts of prenatal smoking (more vs less; D’Onofrio et al., 2008 ; Lambe et al., 2006 ). Meyer et al (2004) also used a case-crossover approach to examine the effects of MSDP on risk of oral cleft; however, their cases were those with cleft lip with or without cleft palate rather than defined by exposure to MSDP. More recently, Salihu et al. (2008) examined MSDP and risk of stillbirth using case-control and case-crossover designs. Similar to Meyer et al (2004) , case status was not defined by MSDP but rather as a stillbirth with controls being defined as live births ( Salihu et al., 2008 ).
In general, this method provides statistical control for confounding factors (e.g., heritable and sociodemographic characteristics of the mother that predict increased probability of MSDP) that might otherwise artifactually create, or alternatively mask, an association between MSDP and child outcomes. Moreover, this design, in combination with molecular genetic information (see examples below), could offer substantial information to the delineation of genetic and environmental factors in the relationship between MSDP and child outcomes. There are potential limitations of this case-crossover design, e.g., (i) mothers who are able to quit in one pregnancy but not all, may be, on average, less nicotine dependent and therefore smoke less than mothers who are unable to quit; (ii) smoking during pregnancy may be secondary to other life stressors that were present during pregnancy and these life events may not be readily captured during assessment (particularly if retrospective reporting is used); (iii) there may be a selection bias if more women give up rather than initiate smoking during the reproductive years ( Meyer et al., 2004 ); (iv) MSDP tends to be highly correlated in sequential pregnancies introducing possible bias due to autocorrelation ( Levy, Lumley, Sheppard, Kaufman, & Checkoway, 2001 ; Mittleman, Maclure, & Robins, 1995 ); and (v) the prevalence of smoking during pregnancy has, in general, declined over time ( CDC, 2004 ) which could affect results. Some of these limitations can be overcome with the use of bi-directional case-crossover designs, where controls (nonexposed siblings) are chosen from both sides of the exposed pregnancy (e.g., Lumley & Levy, 2000 ; Meyer et al., 2004 ). To control for exposure trends, a case-time-control design can also be used in conjunction with the case-crossover design (see Meyer et al., 2004 ). The case-time-control design estimates an exposure trend by explicitly matching cases with controls. This exposure trend is then used to adjust the case-crossover estimates by the trend estimate. There is also the issue of identifying such samples and acquiring large enough samples to make meaningful conclusions. Despite these limitations, this case-crossover design in combination with molecular genetic information holds promise in the study of adverse effects of MSDP.
Molecular genetic studies
Earlier it was suggested that prenatal exposure may have direct teratogenic effects on the fetus leading to more readily observed adverse phenotypes; however, these effects most likely depend on the specific outcome measure of interest. In fact, the effect of MSDP on the fetus may also interact with other factors, such as genetic factors. In an investigation of gene-environment interaction (G×E), Wang and colleagues (2002) investigated the modifying role of two maternal xenobiotic [i.e., corresponding to a chemical compound (such as a drug, pesticide, or carcinogen) that is foreign to a living organism] metabolism genes (CYP1A1 and GSTT1) in the association between MSDP and infant birth weight. Their research was prompted by the fact that tobacco smoke contains approximately 4000 compounds ( Brunnemann & Hoffmann, 1991 ); the most important carcinogens in tobacco smoke are polycyclic aromatic hydrocarbons (PAHs), arylmines, and N-nitrosamines ( Bartsch et al, 2000 ). The ability of an individual to convert toxic metabolites of cigarette smoke to less harmful ones is important for minimizing other adverse health effects. As outlined in Wang et al. (2002) , the metabolic processing of PAH (for example) in humans occurs in two phases. The phase 1 metabolism is an activation process, in which the inhaled, hydrophobic PAHs are converted mainly through aryl hydrocarbon hydroxylase activity into hydrophilic, reactive, electrophilic intermediates that can bind covalently to macromolecules, especially DNA ( National Research Council, 1983 ). These intermediates may be more toxic than the original form. Aryl hydrocarbon hydroxylase, encoded by the CYP1A1 gene, is a phase 1 enzyme and is particularly relevant to the metabolism of cigarette smoke. The phase 2 metabolism is a detoxification process, in which these metabolic intermediates are detoxified by enzymes such as glutathione S-transferases (GSTs) or uridine diphosphate (UDP)-glucuronosyltransferase through transformation into conjugated forms that are sufficiently polar to be excreted from the body ( Timbrell, 1991 ). GSTT1, encoded by the GSTT1 gene, is a major phase 2 enzyme. Both CYP1A1 and GSTT1 are highly polymorphic ( Ishibe et al., 1997 ; Nelson et al., 1995 ; Xu, Kelsey, Wiencke, Wain & Christiani, 1996 ) and their polymorphisms have been associated with their encoded enzyme activities (Kawaijiri et al., 1990; Wiencke, Pemble, Ketterer & Kelsey, 1995 ). Wang et al. (2002) found that, when considering the CYP1A1 genotype (i.e., the combination of alleles for the CYP1A1 gene), increased reduction in infant birth weight was seen in children born to mothers with the Aa/aa genotype (OR=3.2, 95% CI=1.6–6.4). When the GSTT1 genotype was considered, there was increased reduction in birth weight (OR=3.5, 95% CI=1.5–8.3) in children born to mothers with the absent genotype group. When both CYP1A1 and GSTT1 genotypes were considered, the greatest reduction in birth weight was found among smoking mothers with the CYP1A1 Aa/aa and GSTT1 absent genotypes (−1285g). These results suggest an interaction between maternal metabolic genes and MSDP with regard to infant birth weight.
More recently, Tsai et al. (2008) observed a significant joint association of maternal smoking, CYP1A1 (Aa/aa) and GSTT1 (absent) genotypes with gestational age and with preterm delivery. Such joint association was particularly strong in certain preterm subgroups, including spontaneous preterm delivery, preterm delivery < 32 weeks, and preterm delivery accompanied by intrauterine infection/inflammation. Taken together, maternal smoking significantly increased the risk of preterm delivery among women with high-risk CYP1A1 and GSTT1 genotypes. Findings were strongest among preterm delivery accompanied by intrauterine infection/inflammation suggesting that intrauterine infection/inflammation may be a potential pathogenic pathway by which MSDP affects preterm delivery. Specifically, the gene-MSDP interactions may exert their effects synergistically on preterm delivery through maternal and fetal inflammatory responses and raise the possibility of identifying women at high risk for certain pregnancy outcomes by accounting for environmental exposures and genetic polymorphisms ( Tsai et al., 2008 ).
Infante-Rivard, Weinberg, and Guiguet (2006) studied CYP1A1, GSTT1, as well as a set of ‘repair’ genes (XRCC1, XRCC3, and XPD), due to the fact that cigarette smoke can generate reactive oxygen species, which are capable of inducing double-strand breaks in DNA. These ‘repair’ genes can maintain the integrity of the genetic code. The authors investigated these genetic polymorphisms and their interaction with MSDP in the role of small-for-gestational-age births (birth weight below the 10 th percentile according to gestational age and gender). Results indicated that certain genetic variants (maternal CYP1A1, maternal XRCC3, and newborn GSTT1) increased the risk of small-for-gestational-age birth and modified the effects of MSDP by increasing or decreasing its risk ( Infante-Rivard et al., 2006 ). Of particular interest here is the fact that not only are maternal genotypes involved, but also newborn genotypes which emphasizes the importance of obtaining DNA from mother, child, and father if available and conducting family-based studies to further examine the roles of these genes.
There are also a few studies that have focused on, and claim evidence for gene-environment interactions (dopaminergic pathway genes and prenatal smoking) on externalizing behavior in children ( Kahn, Khoury, Nichols & Lanphear, 2003 ; Neuman et al., 2007 ). However, these causal relationships need to be considered carefully. These studies, to the best of my knowledge, do not control for the fact that prenatal smoking may be correlated with parental behaviors that could act as more proximal risk factors that are in turn transmitted to their offspring. In brief, Kahn et al. (2003) found that children with the DAT1 480/480 homozygous genotype who were exposed to prenatal smoking had significantly elevated hyperactive-impulsive and oppositional scores on the Conners' Parent Rating Scale Revised-Long Version. The most striking association was with oppositional defiant behavior. Consistent with Kahn et al. (2003) , Becker, El-Faddagh, Schmidt, Esser and Laught (2008) also reported evidence of an environmentally moderated risk for ADHD behaviors, suggesting that effects of MSDP were dependent on genetic susceptibility (as reflected by individuals’ DAT1 genotypes) and thus operating via G×E interaction. Specifically, males who were exposed to MSDP and who were homozygous for the DAT1 480 allele had higher hyperactivity-impulsivity than males in other groups. This G×E effect was not evident in females. Recently, Neuman et al (2007) also reported that the risk of diagnosis for any DSM-IV ADHD was greatest for children exposed to MSDP and whose genotype contained either the DAT1 440 allele [in contrast to Kahn et al (2003) and Becker et al (2008) ] or the DRD4 exon 3 7-repeat allele. In summary, these results suggest an interaction between dopaminergic genes (in offspring) and MSDP with regard to child externalizing behavior; however, the conflicting nature of reported findings also stress the need for highly refined phenotypes, the measurement of other potential confounding factors (such as the fact that MSDP might only be a marker for maternal ADHD or other important genes transmitted to the child), and the measurement of other gene variants that might be in linkage disequilibrium (non-randomly associated) with the dopaminergic genes investigated ( Becker et al., 2008 ). Futher, the multifactorial nature of many child outcomes underscores the importance of studying both genetic and environmental factors and their interaction ( Becker et al., 2008 ).
Summary of genetically-informative studies
The few genetically-informed studies that have considered MSDP suggest that, for certain outcomes, MSDP does have a specific environmental effect that is not confounded with genetic factors, common environmental factors, and other covariates. GxE (measured gene) studies also indicate that there is suggestive evidence that certain genetic polymorphisms (both maternal and offspring) do moderate the teratogenic effects of prenatal smoking exposure on infant birth weight, preterm delivery, and externalizing behavior. Taken as a group, these results highlight the importance of including genetic and environmental variables in the study of the association between MSDP and offspring outcomes.
A note on prospective vs retrospective studies
Ideally, studies assessing effects of MSDP would recruit participants while pregnant, with continued follow-up of offspring to investigate outcomes associated with MSDP and its correlates (e.g., maternal/paternal psychopathology, home environment, exposure to second-hand smoke, etc). This, however, is not always possible. Many studies must rely on retrospective report of smoking during pregnancy. There has been some question of the reliability of retrospective reporting, in that such reporting could result in underreporting due to social desirability or greater measurement error which could cause the importance of prenatal exposure to be underestimated. Petitti, Friedman, and Kahn (1981) state that the reliability of retrospective reports is similar to the recall of other substance use. More recent reports also indicate high reliability and stability of maternal reporting about their pregnancies, including smoking ( Heath et al., 2003 ; Patrick et al., 1994 ; Reich, Todd, Joyner, Neuman & Heath, 2003 ; Tomeo et al., 1999 ;). Moreover, there is a high correlation between self-reported smoking and serum cotinine measures ( Klebanoff, Levine, Clemens, DerSimonian & Wilkins, 1998 ; McDonald, Perkins, & Walker, 2005 ).
Despite advances in interview assessment and procedures, the use of retrospective reports of the prenatal and postnatal environment should be used with caution. Retrospective recall of environmental exposures are likely to give rise to artefactual gene-environment associations arising from behavioral ‘contamination’ of the reported events (Jaffee & Price, 2007). Specifically, such reports may be influenced by individual differences in personality, mood, or mental health, or may reflect the degree to which past environments were elicited by an individuals behavior ( Kendler, 1996 ; Jaffee & Price, 2007).
It is unlikely, given methodological limitations and the risk factor under consideration (MSDP which, in twin offspring, will not differ), that a single design will provide the answers to the complicated nature of the association between MSDP and subsequent outcomes. Over the past three decades, behavioral geneticists have begun to use designs that combine many of the methods outlined in this report in order to bring more power to bear on analyses. For example, a necessary first step in mapping complex traits to genetic loci is to establish the amount of genetic variation that underlies the phenotypic variation of the trait (i.e., heritability). This is accomplished via twin studies. If phenotypic variation in a trait is found to be caused in part by genetic sources (i.e., the trait is heritable), linkage and/or association studies can be conducted in order to characterize the effects of specific genes on phenotypic variation ( Posthuma & Boomsma, 2000 ). But, if the trait of interest is not found to be heritable, the search for the measured genetic effects (i.e., direct main effects or interactive effects of, for example, dopaminergic genes) will most likely not be initiated. Researchers need to not only (i) use the knowledge that we can gain from the designs presented here as well as the information that animal models of MSDP provide (i.e., the teratogenic effect of nicotine on the fetus), but also (ii) to consider pooling resources in order to conduct studies that are powerful enough to make meaningful conclusions. Only then will we gain insight into the underlying processes involved in MSDP.
The ultimate goal of future research in prenatal tobacco exposure is to attempt to derive a relatively unbiased estimate of the magnitude of the association between exposure and outcome – to determine a real vs. statistically spurious effect. Indeed, the fully unbiased estimate is an elusive concept that is never achieved but hopefully more closely realized through increasingly rigorous and comprehensive methods. Future research in this domain should attempt to achieve as accurate as possible an assessment of the magnitude of the association between MSDP and neuropsychological as well as other more physical outcomes. There is strong reason to believe that the established estimates of MSDP-risk on outcome in the literature are upwardly biased due to lack of control for heritable and other confounding factors. A comprehensive approach incorporating genetically-informed samples is of critical importance to obtain a more refined estimate of these associations. Indeed, the more refined effect size may be smaller than what is currently accepted. This, in and of itself, is of great public health significance, not because it will identify a new putative causal agent, but because it will more accurately assess the upper limit of the potential causal association between MSDP and outcomes important for public health, such as low birth weight, cardiorespiratory illness, and ADHD. This should not diminish concern regarding MSDP, but rather could help clarify what are and are not potential causes of ADHD, other neuropsychological, and physical deficits seen in children across the developmental spectrum. Thus, not only is there the potential that findings could provide yet one more incentive for pregnant women to overcome tobacco dependence and quit, but findings can also guide treatment providers to think more comprehensively about smoking during pregnancy and the potential correlates of said behavior. In other words, treatment providers may not only treat, or be concerned with, MSDP, but also correlated behaviors (e.g., maternal psychopathology, detrimental rearing environment, secondhand exposure to smoking) that might also increase risk of certain offspring outcomes. This more informed approach to treatment or general cessation efforts could, in theory, have significant effects on the major public health concern that is smoking during pregnancy and thus result in something that is of substantial value to the field of public health.
Admittedly getting a pregnant woman to stop smoking is perhaps the most straightforward intervention; however, we have ignored other potential confounding factors for far too long. The reality is that, in humans, we do not understand how much of the association between MSDP and offspring outcomes can be attributed to either nicotine or other smoking by-products. By putting more realistic boundaries on the impact of MSDP and not continuing to ignore confounding factors, we open the door for other avenues of treatment, intervention, and prevention – opportunities that heretofore have been missed. The first step around this hurdle – and elucidating real vs. statistically spurious effects of MSDP -- are genetically informed designs.
Acknowledgement
This work was supported by grant DA17671 from the National Institute of Drug Abuse.
- Abel EL. Smoking during pregnancy: a review of effects on growth and development of offspring. Human Biology. 1980; 52 :593–625. [ PubMed ] [ Google Scholar ]
- Abel EL. Smoking and pregnancy. J Psychoactive Drugs. 1984; 16 :327–338. [ PubMed ] [ Google Scholar ]
- Ajarem JS, Ahmad M. Prenatal nicotine exposure modifies behavior of mice through early development. Pharmacology Biochemistry and Behavior. 1998; 59 :313–318. [ PubMed ] [ Google Scholar ]
- Agrawal A, Knopik VS, Pergadia ML, Waldron M, Bucholz KK, Martin NG, et al. Correlates of cigarette smoking during pregnancy and its genetic and environmental overlap with nicotine dependence. Nicotine and Tobacco Research. 2008; 10 :567–578. [ PubMed ] [ Google Scholar ]
- Bada HA, Das A, Bauer CR, Shankaran S, Lester BM, Gard CC, et al. Low birth weight and preterm births: etiologic function attributable to prenatal drug exposure. Journal of Perinatology. 2005; 25 :631–637. [ PubMed ] [ Google Scholar ]
- Baghurst PA, Tong SL, Woodward A, McMichael AJ. Effects of maternal smoking upon neuropsychological development in early childhood: importance of taking account of social and environmental factors. Paediatr Perinat Epidemiol. 1992; 6 :403–415. [ PubMed ] [ Google Scholar ]
- Bartsch H, Nair U, Risch A, Rojas M, Wikman H, Alexandrov K. Genetic polymorphism of CYP genes, alone or in combination, as a risk Modifier of tobacco-related cancers. Cancer Epidemiol Biomarkers Prev. 2000; 9 :3–28. [ PubMed ] [ Google Scholar ]
- Batstra L, Hadders-Algra M, Neeleman J. Effect of antenatal exposure to maternal smoking on behavioral problems and academic achievement in childhood; prospective evidence from a Dutch birth cohort. Early Human Development. 2003; 75 :21–33. [ PubMed ] [ Google Scholar ]
- Bauman KE, Flewelling RL, LaPrelle J. Parental cigarette smoking and cognitive performance of children. Health Psychol. 1991; 8 :97–105. [ PubMed ] [ Google Scholar ]
- Becker K, El-Faddagh M, Schmidt MH, Esser G, Laught M. Interaction of dopamine transporter genotype with prenatal smoke exposure on ADHD symptoms. Journal of Pediatrics. 2008; 152 :263–269. [ PubMed ] [ Google Scholar ]
- Botting N, Powls A, Cooke RW, Marlow N. Attention deficit hyperactivity disorders and other psychiatric outcomes in very low birthweight children at 12 years. J Child Psychol Psychiatry. 1997; 38 :931–941. [ PubMed ] [ Google Scholar ]
- Brennan PA, Grekin ER, Mednick SA. Maternal smoking during pregnancy and adult male criminal outcomes. Arch Gen Psychiatry. 1999; 56 :215–219. [ PubMed ] [ Google Scholar ]
- Breslau N, Chilcoat HD. Psychiatric sequelae of low birth weight at 11 years of age. Biol Psychiatry. 2000; 47 :1005–1011. [ PubMed ] [ Google Scholar ]
- Brook JS, Brook DW, Whiteman M. The influences of maternal Smoking during pregnancy on the toddler’s negativity. Arch Pediatrics Adol Med. 2000; 154 :381–385. [ PubMed ] [ Google Scholar ]
- Brook DW, Zhang C, Rosenberg G, Brook JS. Maternal cigarette smoking during pregnancy and child aggressive behavior. Am J Addictions. 2006; 15 :450–456. [ PubMed ] [ Google Scholar ]
- Brunnemann K, Hoffmann D. Analytical studies on tobacco-specific N nitrosamines in tobacco and tobacco smoke. Crit Rev Toxicol. 1991; 21 :235–240. [ PubMed ] [ Google Scholar ]
- Buka SL, Shenassa ED, Niaura R. Elevated risk of tobacco dependence among offspring of mothers who smoked during pregnancy: A 30-year prospective study. Am J Psychiatry. 2003; 160 :1978–1984. [ PubMed ] [ Google Scholar ]
- Burt SA. Genes and popularity: evidence of an evocative gene-environment correlation. Psychological Science. 2008; 19 :112–113. [ PubMed ] [ Google Scholar ]
- Button TMM, Thapar A, McGuffin P. Relationship between antisocial behaviour, attention-deficit hyperactivity disorder and maternal prenatal smoking. Br J Psychiatry. 2005; 187 :155–160. [ PubMed ] [ Google Scholar ]
- Byrd RS, Howard CR. Children's passive and prenatal exposure to cigarette smoke. Pediatr Ann. 1995; 24 :640–642. 644–645. [ PubMed ] [ Google Scholar ]
- Caspi A, McClay J, Moffitt TE, Mill J, Martin J, Craig IW, et al. Role of genotype in the cycle of violence in maltreated children. Science. 2002; 297 :851–854. [ PubMed ] [ Google Scholar ]
- Caspi A, Moffitt TE. Gene-environment interactions in psychiatry: joining forces with neuroscience. Nature Reviews Neuroscience. 2006; 7 :583–590. [ PubMed ] [ Google Scholar ]
- Caspi A, Taylor A, Moffitt TE, Plomin R. Neighborhood deprivation affects children’s mental health: Environmental risks identified in a genetic design. Psychological Science. 2000; 11 :338–342. [ PubMed ] [ Google Scholar ]
- Centers for Disease Control (CDC) Annual smoking-attributable mortality, years of potential life lost, and economic costs-United States. MMWR. 2002; 51 :300–303. [ PubMed ] [ Google Scholar ]
- Centers for Disease Control (CDC) Smoking During Pregnancy - United States, 1990–2002. MMWR. 2004; 53 :911–915. [ PubMed ] [ Google Scholar ]
- Cnattingius S. The epidemiology of smoking during pregnancy: Smoking prevalence, maternal characteristics, and pregnancy outcomes. Nicotine and Tobacco Research. 2004; 6 :S125–S140. [ PubMed ] [ Google Scholar ]
- Conter V, Cortinovis I, Rogari P, Riva L. Weight growth in infants born to mothers who smoked during pregnancy. Br J Medicine. 1995; 310 :768–771. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Cornelius MD, Leech SL, Goldschmidt L, Day NL. Prenatal tobacco exposure: Is it risk factor for early tobacco experimentation? Nicotine and Tobacco Research. 2000; 2 :45–52. [ PubMed ] [ Google Scholar ]
- Cornelius MD, Ryan CM, Day NL, Goldschmidt L, Willford JA. Prenatal tobacco effects on neyropsycholgical outcomes among preadolescents. J Developmental and Behavioral Pediatrics. 2001; 22 :217–225. [ PubMed ] [ Google Scholar ]
- Crea TM, Barth RP, Guo S, Brooks D. Behavioral outcomes for substance exposed adopted children: Fourteen years postadoption. American Journal of Orthopsychiatry. 2008; 78 :11–19. [ PubMed ] [ Google Scholar ]
- Dani JA. Overview of nicotinic receptors and their roles in the central nervous system. Biological Psychiatry. 2001; 49 :166–174. [ PubMed ] [ Google Scholar ]
- Davies DP, Abernethy M. Cigarette smoking in pregnancy: associations with maternal weight gain and fetal growth. Lancet. 1976; 1 :385–387. [ PubMed ] [ Google Scholar ]
- Day NL, Richardson GA, Goldschmidt L, Cornelius MD. Effects of prenatal tobacco exposure on preschoolers’ behavior. Journal of Developmental and Behavioral Pediatrics. 2000; 21 :180–188. [ PubMed ] [ Google Scholar ]
- DiFranza JR, Lew RA. Effect of maternal cigarette smoking on pregnancy complications and Sudden Infant Death Syndrome. J Fam Pract. 1995; 40 :385–394. [ PubMed ] [ Google Scholar ]
- D’Onofrio BM, Turkheimer EN, Eaves LJ, Corey LA, Berg K, Solaas MH, et al. The role of the Children of Twins design in elucidating causal relations between parent characteristics and child outcomes. J Child Psychol. 2003; 44 :1130–1144. [ PubMed ] [ Google Scholar ]
- D’Onofrio BM, Van Hulle CA, Waldman ID, Rodgers JL, Harden KP, Rathouz PJ, et al. Smoking during pregnancy and offspring externalizing problems: An exploration of genetic and environmental confounds. Developmenalt Psychopathology. 2008; 20 :139–164. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Ebrahim S, Floyd L, Merrit R, Decoufle P, Holtzman D. Trends in pregnancy-related smoking rates in the United States, 1987–1996. JAMA. 2000; 283 :361–366. [ PubMed ] [ Google Scholar ]
- Ernst M, Moolchan ET, Robinson ML. Behavioral and neural consequences of prenatal exposure to nicotine. J Am Acad Child Adolesc Psychiatry. 2001; 40 :630–641. [ PubMed ] [ Google Scholar ]
- Eskenazi B, Castorina R. Association of prenatal maternal or postnatal child environmental tobacco smoke exposure and neurodevelopmental and behavioral problems in children. Environ Health Perspect. 1999; 107 :991–1000. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Eskenazi B, Trupin LS. Passive and active maternal smoking during pregnancy as measured by serum cotinine, and postnatal smoke exposure II: Effects on neurodevelopment at age 5 years. Am J of Epidemiology. 1995; 142 :S19–S29. [ PubMed ] [ Google Scholar ]
- Eskenazi B, Prehn AW, Christianson RE. Passive and active maternal smoking as measured by serum cotinine: The effect on birthweight. Am J Public Health. 1995; 85 :395–398. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Fergusson DM, Woodward LJ, Horwood LJ. Maternal smoking during pregnancy and psychiatric adjustment in late adolescence. Arch Gen Psychiatry. 1998; 55 :721–727. [ PubMed ] [ Google Scholar ]
- Fried PA, Makin JE. Neonatal behavioral correlates of prenatal exposure to marijuana, cigarettes, and alcohol in a low-risk population. Neurotoxicol Teratol. 1987; 91 :1–7. [ PubMed ] [ Google Scholar ]
- Fried PA, O’Connell CM, Watkinson B. 60- and 72-month follow-up of children prenatally exposed to marijuana, cigarette, and alcohol: cognitive and language assessment. J Developmental and Behavioral Pediatrics. 1992a; 13 :383–391. [ PubMed ] [ Google Scholar ]
- Fried PA, Watkinson B, Gray R. A follow-up study of attentional behavior in 6-year-old children exposed prenatally to marijuana, cigarettes, and alcohol. Neurotoxicity and Teratology. 1992b; 14 :299–311. [ PubMed ] [ Google Scholar ]
- Gilliland FD, Li Y, Peters JM. Effects of maternal smoking during pregnancy and environmental tobacco smoke on asthma and wheezing in children. Am J Repir Crit Care Med. 2001; 163 :429–436. [ PubMed ] [ Google Scholar ]
- Gusella JL, Fried PA. Effects of maternal social drinking and smoking on offspring at 13 months. Neurobeh Toxicol Teratol. 1984; 6 :13–17. [ PubMed ] [ Google Scholar ]
- Hagino N, Lee JW. Effect of maternal nicotine on the development of sites for [ 3 H]nicotine binding in the fetal brain. Int J Dev Neurosci. 1985; 3 :567–571. [ PubMed ] [ Google Scholar ]
- Heath AC, Knopik VS, Madden PAF, Neuman RJ, Lynskey MJ, Slutske WS, et al. Accuracy of mothers’ retrospective reports of smoking during pregnancy: Comparison with twin sister informant ratings. Twin Research. 2003; 6 :297–301. [ PubMed ] [ Google Scholar ]
- Hellstrom-Lindahl E, Gorbounova O, Sieger A, Mousavi M, Nordberg A. Regional distribution of nicotinic receptors during prenatal development of human brain and spinal cord. Dev Brain Res. 1998; 108 :147–160. [ PubMed ] [ Google Scholar ]
- Hellstrom-Lindahl E, Seiger A, Kjaeldgaard A, Nordberg A. Nicotine induced alterations in the expression of nicotinic receptors in primary cultures from human prenatal brain. Neurocscience. 2001; 105 :527–534. [ PubMed ] [ Google Scholar ]
- Huang ZG, Griffioen KJ, Wang X, Dergacheva O, Kamendi H, Gorini C, et al. Differential control of central cardiorespiratory interactions by hypercapnia and the effect of prenatal nicotine. Journal of Neuroscience. 2006; 26 :21–29. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Huizink AC, Mulder EJ. Maternal smoking, drinking or cannabis use during pregnancy and neurobehavioral and cognitive functioning in human offspring. Neuroscience and Biobehavior Reviews. 2006; 30 :24–41. [ PubMed ] [ Google Scholar ]
- Infante-Rivard C, Weinberg CR, Guiguet M. Xenobiotic-metabolizing genes and small-for-gestational age births: Interaction with maternal smoking. Epidemiology. 2006; 17 :38–46. [ PubMed ] [ Google Scholar ]
- Ishibe N, Wiencke JK, Zuo ZF, McMillan A, Spitz M, Kelsey KT. Susceptibility to lung cancer in light smokers associated with CYP1A1 polymorphisms in Mexican- and African-Americans. Cancer Epidemiol Biomarkers Prev. 1997; 6 :1075–1080. [ PubMed ] [ Google Scholar ]
- Jacob T, Waterman B, Heath AC, True W, Bucholz KK, Haber R, et al. Genetic and environmental effects on offspring alcoholism. Arch Gen Psychiatry. 2003; 60 :1265–1272. [ PubMed ] [ Google Scholar ]
- Jaffee SR, Price TS. Gene-environment correlations: a review of the evidence and implications for prevention of mental illness. Molecular Psychiatry. 12 :432–442. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Janson C. The effect of passive smoking on respiratory health in children and adults. International Journal of Tuberculosis and Lung Disorders. 2004; 8 :510–516. [ PubMed ] [ Google Scholar ]
- Johns JM, Louis TM, Becker RF, Means LW. Behavioral effects of prenatal exposure to nicotine in guinea pigs. Neurobehav Toxicol Teratol. 1982; 4 :365–369. [ PubMed ] [ Google Scholar ]
- Johns JM, Walters PA, Zimmerman LI. The effects of chronic prenatal exposure to nicotine on the behavior of guinea pigs (Cavia porcellus) J Gen Psychol. 1993; 120 :49–63. [ PubMed ] [ Google Scholar ]
- Kahn RS, Khoury J, Nichols WC, Lanphear BP. Role of dopamine transporter genotype and maternal prenatal smoking in childhood hyperactive impulsive, inattentive, and oppositional behaviors. J Pediatr. 2003; 143 :104–110. [ PubMed ] [ Google Scholar ]
- Kawajiri K, Nakachi K, Imai K, Yoshii A, Shinoda N, Watanabe J. Identification of genetically high risk individuals to lung cancer by DNA polymorphisms of the cytochrome P4501A1 gene. FEBS Lett. 1990; 263 :131–133. [ PubMed ] [ Google Scholar ]
- Kendler KS. Parenting: a genetic-epidemiological perspective. American Journal of Psychiatry. 1996; 153 :11–20. [ PubMed ] [ Google Scholar ]
- Kendler KS, Eaves LJ. Models for the joint effects of genotype and environment on liability to psychiatric illness. Am J Psychiatry. 1986; 143 :279–289. [ PubMed ] [ Google Scholar ]
- Kendler KS, Gardner CO. Monozygotic twins discordant for major depression: A preliminary exploration of the role of environmental experiences in the aetiology and course of illness. Psychol Med. 2001; 31 :411–423. [ PubMed ] [ Google Scholar ]
- Key APF, Ferguson M, Molfese DL, Peach K, Lehman C, Molfese VJ. Smoking during pregnancy affects speech processing ability in newborn infants. Environmental Health Perspectives. 2006 E-pub ahead of print. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Klebanoff MA, Levine RJ, Clemens JD, DerSimonian R, Wilkins DG. Serum cotinine concentration and self-reported smoking during pregnancy. Am J Epidemiol. 1998; 148 :259–262. [ PubMed ] [ Google Scholar ]
- Knopik VS, Heath AC, Jacob T, Slutske WS, Bucholz KK, Madden PAF, et al. Maternal alcohol use disorder and offspring ADHD: Disentangling genetic and environmental effects using a children-of-twins design. Psychological Medicine. 2006; 36 :1461–1472. [ PubMed ] [ Google Scholar ]
- Knopik VS, Sparrow EP, Madden PAF, Bucholz KK, Hudziak JJ, Reich W, et al. Contributions of parental alcoholism, prenatal substance exposure, and genetic transmission to child ADHD risk: A female twin study. Psychological Medicine. 2005; 35 :625–635. [ PubMed ] [ Google Scholar ]
- Kremen WS, Lyons MJ, Boake C, Xian H, Jacobson KC, Waterman B, et al. A discordant twin study of premorbid cognitive ability in schizophrenia. J Clinical Experimental Neuropsychology. 2006; 28 :208–224. [ PubMed ] [ Google Scholar ]
- Kyrklund-Blomberg NB, Granath F, Cnattingius S. Maternal smoking and causes of very preterm birth. Acta Obstet Gynecol Scand. 2005; 84 :572–577. [ PubMed ] [ Google Scholar ]
- Lambe M, Hultman C, Torrang A, MacCabe J, Cnattingius S. Maternal smoking during pregnancy and school performance at age 15. Epidemiology. 2006; 17 :524–530. [ PubMed ] [ Google Scholar ]
- Law KL, Stroud LR, LaGasse LL, Niaura R, Liu J, Lester BM. Smoking during pregnancy and newborn neurobehavior. Pediatrics. 2003; 111 :1318–1323. [ PubMed ] [ Google Scholar ]
- Leve LD, Neiderhiser JM, Ge X, Scaramella LV, Conger RD, Reid JB, et al. The Early Growth and Development Study: A prospective adoption design. Twin Research and Human Genetics. 2007; 10 :84–95. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Levin ED, Briggs SJ, Christopher NS, Rose JE. Prenatal nicotine exposure and cognitive performance in rats. Neurotoxicol Teratol. 1993; 15 :251–260. [ PubMed ] [ Google Scholar ]
- Levin ED, Slotkin TA. Developmental neurotoxicity of nicotine. In: Slikker W, Chang LW, editors. Handbook of Developmental Neurotoxicology. San Diego: Academic Press; 1998. pp. 587–615. [ Google Scholar ]
- Levy D, Lumley T, Sheppard L, Kaufman J, Checkoway H. Referent selection in case-crossover analyses of acute health effects of air pollution. Epidemiology. 2001; 12 :186–192. [ PubMed ] [ Google Scholar ]
- Liang K, Poytress BS, Chen Y, Leslie FM, Weinberger NM, Metherate R. Neonatal nicotine exposure impairs nicotinic enhancement of central auditory processing and auditory learning in adult rats. European J Neuroscience. 2006; 24 :857–866. [ PubMed ] [ Google Scholar ]
- Linnet KM, Dalsgaard S, Obel C, Wisborg K, Henriksen TB, Rodriguez A, et al. Maternal lifestyle factors in pregnancy risk of attention deficit hyperactivity disorder and associated behaviors: review of the current evidence. Am J Psychiatry. 2003; 160 :1028–1040. [ PubMed ] [ Google Scholar ]
- Lumley T, Levy D. Bias in the case-crossover design: Implications for studies of air pollution. Environmetrics. 2000; 11 :689–704. [ Google Scholar ]
- Lynskey MT, Heath AC, Bucholz KK, Slutske WS, Madden PAF, Nelson EC, et al. Escalation of drug use in early-onset cannabis users vs co-twin controls. JAMA. 2003; 289 :427–483. [ PubMed ] [ Google Scholar ]
- Makin J, Fried PA, Watkinson B. A comparison of active and passive smoking during pregnancy: Long term effects. Neurotoxicol Teratol. 1991; 13 :5–12. [ PubMed ] [ Google Scholar ]
- Marlow N, Wolke D, Bracewell MA, Samara M EPI Cure Study Group. Neurologic and developmental disability at six years of age after extremely preterm birth. NE Journal of Medicine. 2005; 352 :9–19. [ PubMed ] [ Google Scholar ]
- Martin JC, Becker RF. The effects of maternal nicotine absorption of hypoxic upon appetitive behavior of rat offspring. Dev Psychobiol. 1971; 4 :133–147. [ PubMed ] [ Google Scholar ]
- Maughan B, Taylor A, Caspi A, Moffitt TE. Prenatal smoking and early childhood conduct problems. Archives of General Psychiatry. 2004; 61 :836–843. [ PubMed ] [ Google Scholar ]
- McCartney JS, Fried PA, Watkinson B. Central auditory processing in school age children prenatally exposed to cigarette smoke. Neurotoxicol Teratol. 1994; 16 :269–276. [ PubMed ] [ Google Scholar ]
- McDonald SD, Perkins SL, Walker MC. Correlation between self-reported smoking status and serum cotinine during pregnancy. Addictive Behaviors. 2005; 30 :853–857. [ PubMed ] [ Google Scholar ]
- Meyer KA, Williams P, Hernandez-Diaz S, Cnattingius S. Smoking and the risk of oral clefts: Exploring the impact of study designs. Epidemiology. 2004; 15 :671–678. [ PubMed ] [ Google Scholar ]
- Milberger S, Biederman J, Faraone SV, Chen L, Jones J. Is maternal smoking during pregnancy a risk factor for attention-deficit hyperactivity disorder in children? Am J Psychiatry. 1996; 153 :1138–1142. [ PubMed ] [ Google Scholar ]
- Milberger S, Biederman J, Faraone SV, Chen L, Jones J. Further evidence of an association between attention-deficit hyperactivity disorder and cigarette smoking: Findings from a high-risk sample of siblings. Am J Addict. 1997; 6 :205–217. [ PubMed ] [ Google Scholar ]
- Milberger S, Biederman J, Faraone SV, Jones J. Further evidence of and association between maternal smoking during pregnancy and attention-deficit hyperactivity disorder: Findings from a high-risk sample of siblings. J Clin Child Psychol. 1998; 27 :352–358. [ PubMed ] [ Google Scholar ]
- Mittleman MA, Maclure M, Robins JM. Control sampling strategies for case crossover studies: an assessment of relative efficiency. Am J Epidemiol. 1995; 142 :91–98. [ PubMed ] [ Google Scholar ]
- Moffitt TE, Caspi A, Rutter M. Strategy for investigating interactions between measured genes and measured environments. Arch Gen Psychiatry. 2005; 62 :473–481. [ PubMed ] [ Google Scholar ]
- Morales-Suarez-Varela MM, Bille C, Christensen K, Olson J. Smoking habits, nicotine use, and congenital malformations. Obstetrics and Gynecology. 2006; 107 :51–57. [ PubMed ] [ Google Scholar ]
- Mortensen EL, Michaelsen KF, Sanders SA, Reinisch JM. A dose response relationship between maternal smoking during late pregnancy and adult intelligence in male offspring. Paediatric and Perinatal Epidemiology. 2005; 19 :4–11. [ PubMed ] [ Google Scholar ]
- Naeye RL. Effects of maternal cigarette smoking on the fetus and the placenta. British Journal Obstet Gynaecol. 1978; 85 :732–737. [ PubMed ] [ Google Scholar ]
- Naeye RL, Peters EC. Mental development of children whose mothers smoked during pregnancy. Obstet Gynecol. 1984; 64 :601–607. [ PubMed ] [ Google Scholar ]
- Nance WE. Genetic studies of offspring of identical twins: A model for the analysis of quantitative inheritance in man. Acta Geneticae Medicae et Gemellologiae. 1976; 24 :103–113. [ PubMed ] [ Google Scholar ]
- Nance WE, Corey LA. Genetic models for the analyses of data from families of identical twins. Genetics. 1976; 83 :811–826. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Nance WE, Corey LA, Boughman JA. Monozygotic twin kinships: A new design for genetic and epidemiological research. In: Morton NE, Chung CS, editors. Genetic Epidemiology. New York: Academic Press; 1978. pp. 87–132. [ Google Scholar ]
- National Research Council. Polycyclic Aromatic Hydrocarbons: Evaluation Of Sources and Effects. Washington, DC: National Academy Press; 1983. pp. 197–262. [ PubMed ] [ Google Scholar ]
- Neff RA, Simmens SJ, Evans C, Mendelowitz D. Prenatal nicotine exposure alters central cardiorespiratory responses to hypoxia in rats: Implementation for sudden infant death syndrome. Journal of Neuroscience. 2004; 24 :9261–9268. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Neiderhiser JM, Leve LD, Ge X, Scaramella LV, Conger RD, Reid JB, et al. The impact of prenatal drug exposure on toddler behavior: Distinguishing genetic effects from exposure using an adoption design. Behavior Genetics. 2007; 37 :780. [ Google Scholar ]
- Nelson HH, Wiencke JK, Christiani DC, Cheng TJ, Zuo ZF, Schwartz BS, et al. Ethnic differences in the prevalence of homozygous deleted genotype of glutathione S-transferase theta. Carcinogenesis. 1995; 16 :1243–1245. [ PubMed ] [ Google Scholar ]
- Neuman RJ, Lobos E, Reich W, Henderson CA, Sun L-W, Todd RD. Prenatal smoking and exposure and dopaminergic genotypes interact to cause a severe ADHD subtype. Biological Psychiatry. 2007; 61 :1320–1328. [ PubMed ] [ Google Scholar ]
- Obel C, Henriksen TB, Hedegaard M, Secher NJ, Ostergaard J. Smoking during pregnancy and babbling abilities in the 8-month-old infant. Paediatr Perinat Epidemiol. 1998; 12 :37–48. [ PubMed ] [ Google Scholar ]
- Orlebeke JF, Knol DL, Verhulst FC. Child behavior problems increase by maternal smoking during pregnancy. Archives of Env Health. 1999; 54 :15–19. [ PubMed ] [ Google Scholar ]
- Pakkanen JS, Jokitalo E, Tuominen RK. Upregulation of beta2 and alpha7 subunit containing nicotinic acetylcholine receptors in mouse striatum at cellular level. European Journal of Neuroscience. 2005; 21 :2681–2691. [ PubMed ] [ Google Scholar ]
- Patrick DL, Cheadle A, Thompson DC, Diehr P, Koepsell T, Kinne S. The validity of self-reported smoking: A review and meta-analysis. Am J Public Health. 1994; 84 :1086–1093. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Paz R, Barsness B, Martenson T, Tanner D, Allan AM. Behavioral teratogenicity induced by nonforced maternal nicotine consumption. Neuropsychopharmacology. 2006 E-pub ahead of print. [ PubMed ] [ Google Scholar ]
- Perkins KA, Sexton JE, DiMarco A, Fonte C. Acute effects of tobacco smoking on hunger and eating in male and female smokers. Appetite. 1994; 22 :149–158. [ PubMed ] [ Google Scholar ]
- Peters MA, Ngan LL. The effects of totigestational exposure to nicotine on pre- and postnatal development in the rat. Arch Int Pharmacodyn Ther. 1982; 257 :155–167. [ PubMed ] [ Google Scholar ]
- Petitti DB, Friedman GD, Kahn W. Accuracy of information on smoking habits provided on self-administered research questionnaires. Am J Public Health. 2005; 71 :308–311. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Plomin R, DeFries JC, McClearn G, McGuffin P. Behavioral Genetics. Fifth Edition. Worth Publishers; 2008. [ Google Scholar ]
- Posthuma D, Boomsma DI. A note on statistical power in extended twin designs. Behavioral Genetics. 2000; 30 :147–158. [ PubMed ] [ Google Scholar ]
- Preuss TM. Do rats have prefrontal cortex? The Rose-Woolsey-Akert program reconsidered. J Cognitive Neuroscience. 1995; 7 :1–24. [ PubMed ] [ Google Scholar ]
- Purcell S, Koenen KC. Environmental mediation and the twin design. Behavior Genetics. 2005; 35 :491–498. [ PubMed ] [ Google Scholar ]
- Ramsay MC, Reynolds CR. Does smoking by pregnant women influence IQ, birth weight, and developmental disabilities in their infants? A Methodological review and multivariate analysis. Neuropsychology Review. 2000; 10 :1–40. [ Google Scholar ]
- Rantakallio P, Koiranen M. Neurological handicaps among children whose mothers smoked during pregnancy. Prev Med. 1987; 16 :597–606. [ PubMed ] [ Google Scholar ]
- Rasanen P, Haako H, Isohanni M, Hodgins S, Jarvelin MR, Tiihonen J. Maternal smoking during pregnancy and risk of criminal behavior among adult male offspring in the Northern Finland Birth Cohort. Am J Psychiatry. 1999; 156 :857–862. [ PubMed ] [ Google Scholar ]
- Reich W, Todd RD, Joyner CA, Neuman RJ, Heath AC. Reliability And stability of mothers’ reports about their pregnancies with twins. Twin Research. 2003; 6 :85–88. [ PubMed ] [ Google Scholar ]
- Ricketts SA, Murray EK, Schwalberg R. Reducing low birthweight by resolving risks: Results from the Colorado’s Prenatal Plus Program. Am J Public Health. 2005; 95 :1952–1957. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Rodgers J, Cleveland H, van den Oord E, Rowe D. Resolving the debate over birth order, family size, and intelligence. American Psychologist. 2000; 55 :599–612. [ PubMed ] [ Google Scholar ]
- Roy TS, Seidler FJ, Slotkin TA. Prenatal nicotine exposure evokes alterations of cell structure in hippocampus and somatosensory cortex. Journal of Pharmacology and Experimental Therapeutics. 2002; 300 :124–133. [ PubMed ] [ Google Scholar ]
- Rutter M, Dunn J, Plomin R, Simonoff E, Pickles A, Maughan B, et al. Integrating nature and nurture: Implications of person environment correlations and interactions for developmental psychopathology. Development and Psychopathology. 1997; 9 :335–364. [ PubMed ] [ Google Scholar ]
- Rutter M, Moffitt TE, Caspi A. Gene-environment interplay and psychopathology: Multiple varieties but real effects. J Child Psychol Psychiatry. 2006; 47 :226–261. [ PubMed ] [ Google Scholar ]
- Rutter M, Pickles A, Murray R, Eaves LJ. Testing hypotheses on specific environmental causal effects on behavior. Psychological Bulletin. 2001; 127 :291–324. [ PubMed ] [ Google Scholar ]
- Salihu HM, Aliyu MH, Kirby RS. In utero nicotine exposure and fetal growth inhibition among twins. Am J Perinatol. 2006; 22 :421–427. [ PubMed ] [ Google Scholar ]
- Salihu HM, Sharma PP, Getahun D, Hedayatzadeh M, Peters S, Kirby RS, et al. Prenatal tobacco use and risk of stillbirth: A case-control and bidirectional case-crossover study. Nicotine and Tobacco Research. 2008; 10 :159–166. [ PubMed ] [ Google Scholar ]
- Sastry BV. Placental toxicology: tobacco smoke, abused drugs, multiple chemical interactions, and placental function. Reprod Fertil Dev. 1991; 3 :355–372. [ PubMed ] [ Google Scholar ]
- Saxton DW. The behavior of infants whose mothers smoke in pregnancy. Early Hum Dev. 1978; 2 :363–369. [ PubMed ] [ Google Scholar ]
- Sexton M, Fox NL, Hebel JR. Prenatal exposure to tobacco II: Effects on cognitive functioning at age three. International J of Epidemiology. 1990; 19 :72–77. [ PubMed ] [ Google Scholar ]
- Shea AK, Steiner M. Cigarette smoking during pregnancy. Nicotine and Tobacco Research. 2008; 10 :267–278. [ PubMed ] [ Google Scholar ]
- Silberg JL, Parr T, Neale MC, Rutter M, Angold A, Eaves LJ. Maternal smoking during pregnancy and risk to boys' conduct disturbance: An examination of the causal hypothesis. Biological Psychiatry. 2003; 53 :130–135. [ PubMed ] [ Google Scholar ]
- Slikker W, Jr, Xu ZA, Levin ED, Slotkin TA. Mode of action: Disruption of brain cell replication, second messenger, and neurotransmitter systems during development leading to cognitive dysfunction – Developmental toxicity of nicotine. Critical Reviews of Toxicology. 2005; 35 :703–711. [ PubMed ] [ Google Scholar ]
- Slotkin TA. Fetal nicotine or cocaine exposure: which one is worse? J Pharmacol Exp Ther. 1998; 285 :931–945. [ PubMed ] [ Google Scholar ]
- Slotkin TA. Development cholinotoxicants: Nicotine and chlorpyrifos. Envrionm Health Perspect. 1999; 107 (suppl 1):71–80. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Slotkin TA, McCook EC, Lappi SE, Seidler FJ. Altered development of basal and forskolin-stimulated adenylate cyclase activity in brain regions of rats exposed to nicotine prenatally. Dev Brain Res. 1992; 68 :233–239. [ PubMed ] [ Google Scholar ]
- Slotkin TA, Orband-Miller L, Queen KL. Development of [3H]nicotine binding sites in brain regions of rats exposed to nicotine prenatally via maternal injections or infusions. J Pharmacol Exp Ther. 1987; 242 :232–237. [ PubMed ] [ Google Scholar ]
- Smith B, Martin J, Ventura S. Births and Deaths: Preliminary Data for July 1997-June 1998. Hyattsville, MD: National Center for Health Services; 1999. [ PubMed ] [ Google Scholar ]
- Sorenson CA, Raskin LA, Suh Y. The effects of prenatal nicotine on radial arm maze performance in rats. Pharmacol Biochem Behav. 1991; 40 :991–993. [ PubMed ] [ Google Scholar ]
- Stocks J, Dezateux C. The effect of parental smoking on lung function and development during infancy. Repirology. 2003; 8 :266–285. [ PubMed ] [ Google Scholar ]
- Suzuki K, Minei LJ, Johnson EE. Effects of nicotine upon uterine blood flow in the pregnant rhesus monkey. Am J Obstetrics and Gynecology. 1980; 136 :1009–1013. [ PubMed ] [ Google Scholar ]
- Thapar A, Fowler T, Rice F, Scourfield J, van den Bree M, Thomas H, et al. Maternal smoking during pregnancy and attention deficit hyperactivity disorder symptoms in offspring. Am J Psychiatry. 2003; 160 :1985–1989. [ PubMed ] [ Google Scholar ]
- Timbrell J. Principles of Biochemical Toxicology. 2nd Edition. Washington, DC: Taylor & Francis; 1991. [ Google Scholar ]
- Tomeo CA, Rich-Edward JW, Michels KB, Berkey CS, Hunter DJ, Frazier AL, et al. Reproducibility and validity of maternal recall of pregnancy-related events. Epidemiology. 1999; 10 :774–777. [ PubMed ] [ Google Scholar ]
- Tsai HJ, Liu X, Mestan K, Yu Y, Zhang S, Fang Y, et al. Maternal cigarette smoking, metabolic gene polymorphisms, and preterm delivery: new insights on GxE interactions and pathogenic pathways. Human Genetics. 2008; 123 :356–369. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Van de Kamp JL, Collins AC. Prenatal nicotine alters nicotinic receptor development in the mouse brain. Pharmacol Biochem Behav. 1994; 47 :889–900. [ PubMed ] [ Google Scholar ]
- Wakschlag L, Keenan K. Clinical significance and correlates of disruptive behavior symptoms in environmentally at-risk preschoolers. J Clin Child Psychol. 2001; 30 :262–275. [ PubMed ] [ Google Scholar ]
- Wakschlag LS, Lahey BB, Loeber R, Green SM, Gordon RA, Leventhal BL. Maternal smoking during pregnancy and the risk of conduct disorder in boys. Arch Gen Psychiatry. 1997; 54 :670–676. [ PubMed ] [ Google Scholar ]
- Wakschlag LS, Pickett KE, Cook E, Benowitz NL, Leventhal BL. Maternal smoking during pregnancy and severe antisocial behavior in offspring: A review. American Journal of Public Health. 2002; 92 :966–974. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Wang X, Zuckerman B, Pearson C, Kaufman G, Chen C, Wang G, et al. Maternal cigarette smoking, metabolic gene polymorphism, and infant birth weight. JAMA. 2002; 287 :195–202. [ PubMed ] [ Google Scholar ]
- Weissman MM, Warner V, Wickramaratne PJ, Kandel DB. Maternal smoking during pregnancy and psychopathology in offspring followed to adulthood. J Am Acad Child Adolesc Psychiatry. 1999; 38 :892–899. [ PubMed ] [ Google Scholar ]
- Wiencke JK, Pemble S, Ketterer B, Kelsey KT. Gene deletion of glutathione S-transferase theta. Cancer Epidemiol Biomarkers Prev. 1995; 4 :253–259. [ PubMed ] [ Google Scholar ]
- Xu X, Kelsey KT, Wiencke JK, Wain JC, Christiani DC. Cytochrome P450 CYP1A1 MspI polymorphism and lung cancer susceptibility. Cancer Epidemiol Biomarkers Prev. 1996; 5 :687–692. [ PubMed ] [ Google Scholar ]
- Yanai J, Pick CG, Rogel-Fuchs Y, Zahalka EA. Alterations in hippocampal cholinergic and hippocampal behaviors after early exposure to nicotine. Brain Res Bull. 1992; 29 :363–368. [ PubMed ] [ Google Scholar ]
- Yolton K, Dietrich K, Auinger P, Lanphear B, Hornung R. Exposure to environmental tobacco smoke and cognitive abilities among U.S. children and adolescents. Environ Health Perspect. 2005; 113 :98–103. [ PMC free article ] [ PubMed ] [ Google Scholar ]
- Open access
- Published: 05 January 2006
Maternal smoking during pregnancy increases the risk of recurrent wheezing during the first years of life (BAMSE)
- Eva Lannerö 1 , 2 , 3 ,
- Magnus Wickman 1 , 3 , 4 ,
- Goran Pershagen 1 , 3 &
- Lennart Nordvall 5
Respiratory Research volume 7 , Article number: 3 ( 2006 ) Cite this article
17k Accesses
150 Citations
6 Altmetric
Metrics details
Exposure to cigarette smoking during foetal and early postnatal life may have implications for lung health. The aim of this study was to assess the possible effects of such exposure in utero on lower respiratory disease in children up to two years of age.
A birth cohort of 4089 newborn infants was followed for two years using parental questionnaires. When the infant was two months old the parents completed a questionnaire on various lifestyle factors, including maternal smoking during pregnancy and after birth. At one and two years of age information was obtained by questionnaire on symptoms of allergic and respiratory diseases as well as on environmental exposures, particularly exposure to environmental tobacco smoke (ETS). Adjustments were made for potential confounders.
When the mother had smoked during pregnancy but not after that, there was an increased risk of recurrent wheezing up to two years' age, OR adj = 2.2, (95% CI 1.3 – 3.6). The corresponding OR was 1.6, (95% CI 1.2 – 2.3) for reported exposure to ETS with or without maternal smoking in utero. Maternal smoking during pregnancy but no exposure to ETS also increased the risk of doctor's diagnosed asthma up to two years of age, OR adj = 2.1, (95% CI 1.2 – 3.7).
Exposure to maternal cigarette smoking in utero is a risk factor for recurrent wheezing, as well as doctor's diagnosed asthma in children up to two yearsof age.
Many children are exposed to tobacco smoking, both before and after they are born. Maternal smoking during pregnancy is believed to affect the utero-placental flow, leading to an impaired foetal nutrition and consequent intrauterine growth retardation [ 1 ]. The foetus of smoking women is exposed from the time of conception to the same levels of nicotine as active smokers [ 2 ]. Smoking during pregnancy affects foetal lung development, reflected in spirometric flow in the neonate, especially when there is a family history of asthma and hypertension during pregnancy [ 3 , 4 ] and causes abnormal airway function [ 5 , 6 ]. Effects of ETS due to parental smoking on respiratory health in early childhood have been described in epidemiological studies [ 7 – 10 ] but few have made an effort to discriminate between effects of prenatal and postnatal exposure. Recent studies, however, suggest that smoke exposure in utero may be at least as detrimental to respiratory health in early life as postnatal exposure to ETS [ 11 , 12 ].
This prospective birth cohort study focuses on maternal smoking during pregnancy as a risk factor for recurrent wheezing during the first two years of life.
Study subjects
From February 1994 until November 1996, 4089 newborn infants (2,024 girls and 2,065 boys) were included in a population based prospective study, BAMSE (Children, Allergy, Milieu, Stockholm, Epidemiological survey). The children were born in predefined areas in Stockholm and recruited at their first visit to the Child Health Centre. During the recruitment period 7,221 infants were born in the study area and of these 1,256 were excluded because the families planned to move within a year, had insufficient knowledge of Swedish or an already enrolled older sibling. Another reason for exclusion was a serious disease in the neonate. For 477 infants correct addresses were not available. Thirteen hundred and ninety-nine declined participation. The final study cohort thus constituted 75 % of the eligible children. Details of the study design, inclusion criteria, enrolment and data collection are described in detail elsewhere [ 13 – 15 ].
Questionnaire
The first questionnaire was filled in by the parents at the time of enrolment (Q0) at a median age of the children of 2 months (10 th percentile 0 months, 90 th percentile 5 months of age). The questionnaire aimed to assess the home environment as well as various indoor environmental exposures such as maternal smoking during pregnancy and smoking habits of both parents after birth of the child. A second part of the questionnaire covered the health of both parents with focus on allergic diseases i.e. asthma, allergic rhino-conjunctivitis and eczema. Socioeconomic status was classified according to the Nordic standard occupational classification (NYK) and Swedish socio-economic classification (SEI) [ 16 ]. The children were categorised on the basis of their parents' occupation into blue-collar workers, white-collar workers and others (students, unemployed). Identical questionnaires (Q1 and Q2) dealt with disease symptoms in the children and were distributed by mail to the parents when the children were one and two years of age. Combinations of reported symptoms were used to define criteria for different diagnoses (see below). Information on important exposure factors, such as parental smoking and breast-feeding, were also obtained from the questionnaires. The questions on symptoms and tobacco smoke exposure have been used in earlier studies [ 17 – 19 ]. Reminders for all three questionnaires were sent three times. The response rates to Q1 and Q2 were 96% and 94%, respectively. The median age for answering Q1 was 12 months and for Q2 24 months. Those who had responded to all three questionnaires (N = 3,791, 93%) before one, two and three years of age of the child, respectively, constituted the basis for this study.
Assessment of pre- and postnatal tobacco smoke exposure
Foetal exposure to maternal smoking was reported in Q0 and was defined as maternal daily smoking of one cigarette or more during any trimester of pregnancy. The degree of such exposure was quantified for each trimester separately. Information on paternal smoking during the period in utero was not collected.
ETS was defined from exposure to maternal smoking of one cigarette or more daily during the first months of life and/or maternal smoking at one year of age of the child. Quantitative information i.e. the number of cigarettes smoked both of mothers and fathers, was obtained in Q0 for the first two months, Q1 and Q2 for the first and second year of life, respectively. In Q0 the parents also indicated whether they smoked at home and when the answer was yes whether they smoked on the balcony/at an open window/under the fan, thus actively avoiding direct exposure of the child.
Classification of outcome
Recurrent wheezing up to two years of age.
Three episodes of wheezing or more after three months of age in combination with the use of inhaled glucocorticoids and/or signs of bronchial hyperreactivity (wheezing or severe coughing when playing or being excited, or disturbed coughing at night not associated with common cold).
Doctor's diagnosed asthma
Reported "asthma" diagnosed by a doctor during the first and/or second year of life of the child.
Any wheezing
Wheezing and/or disturbing cough at night not associated with a common cold during the first and/or second year of life.
Odds ratios (ORs) and ninety-five percent confidence intervals (CIs) were calculated using logistic regression. To identify potential confounders several models including various covariates were tested (heredity, socioeconomy, maternal age, keeping of cat and/or dog, construction year of the home and duration of breastfeeding). Finally, adjustments were made for heredity (defined as doctor-diagnosed asthma and asthma medication and/or allergic rhino-conjunctivitis diagnosed by a doctor in combination with reported allergy to furred pets and/or pollen in one or both parents), exclusive breastfeeding during 4 months or more and maternal age ≥ 26 years, because these variables changed the OR estimates for smoking exposure. To test for interaction between smoking and other covariates an interaction term was included in the logistic regression model. The chi-square test and the Fisher exact test were used for statistical analyses of proportions.
Complete information on maternal smoking during pregnancy and answers on all three questionnaires were required to be included in the analyses and this was available for 3790 subjects.
Statistical analyses were made with the Stata Statistical Software: Release 8.0 (College Station, Texas, USA).
The study was approved by the ethical committee at the Karolinska Institutet, Stockholm, Sweden.
Short duration of breast-feeding, maternal age below 26 years, socio-economic status of the parents, the keeping of cat and/or dog and reported dampness were all associated with maternal smoking during pregnancy (table 1 ). In total, 469 infants were exposed to maternal smoking in utero. The prevalence of smoking decreased during pregnancy and reported smoking during the first, second and third trimester were 12%, 10 % and 9 % respectively. Twelve percent of the mothers reported to have smoked at least one cigarette daily during any part of or all through pregnancy. During the child's first two months the corresponding proportion was 8.0%, and when the child was one and two years old 9.4 and 10%, respectively. The corresponding reported postnatal exposure to paternal smoking was 16, 12 and 11%, respectively. Any exposure to ETS during the first two years of life of the children was reported for 25% of the children. In families with smoking fathers 34% of the mothers smoked compared to 8.3% in families with non-smoking fathers (p < 0.001). Most of the smoking parents (94%) reported in Q0 that they almost always smoked only outdoors, near open window or under the fan when at home.
The reported smoking of mothers with asthma or respiratory allergy (asthma requiring medication and/or doctor's diagnosed allergic rhino-conjunctivitis with reported allergy to furred pets and/or pollen) tended to be lower than that of mothers without such allergy both during pregnancy and the child's first two years (figure 1 ). This also held true for paternal smoking.

Smoking during pregnancy and the first two years of the child and parents with or without asthma and/or respiratory allergy.
The cumulative incidence of recurrent wheezing, doctor's diagnosed asthma and any wheezing up to two years of age were 8.5%, 6.5% and 27%, respectively. The reported smoking pattern of mothers of children with recurrent wheezing differed from that of the mothers with children without recurrent wheezing (figure 2 ). Maternal smoking of one cigarette daily or more was reported for 16 % of the children with recurrent wheezing at one year of age, compared to 8.7% for healthy children (p < 0.001). The corresponding proportions at two year's age were 17 and 9.4% (p < 0.001). Eleven percent of the mothers of the children with recurrent wheezing reported to have smoked ten cigarettes or more daily at one and 12% at two years age. The corresponding figures were 6.3% and 7.0% for mothers with healthy children.
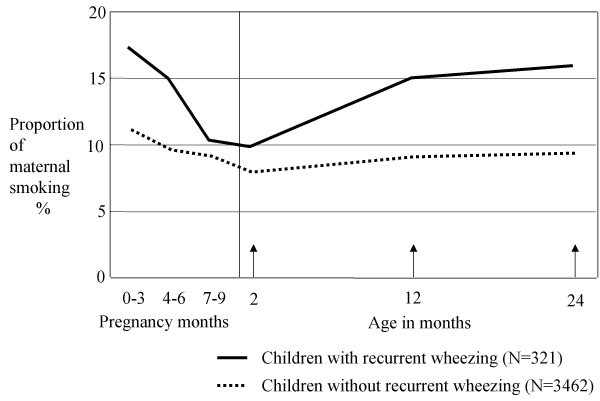
Proportion of maternal smoking of one or more cigarettes daily during pregnancy and during the first two years of the child among children with and without recurrent wheezing.
A large majority of infants (85%) were reported neither to have been exposed to maternal smoking during pregnancy, nor to any maternal smoking during the first two months of life and/or at one year of age, and these constituted the reference group. One-hundred and thirty-three children (3.6%) had been exposed in utero, but not after being born. Eleven percent of the children were exposed to ETS with or without maternal smoking in utero. Only 2.4% of the children were reported to have been exposed exclusively to ETS.
Maternal smoking during any period of pregnancy, but not after giving birth was associated with an increased risk of recurrent wheezing at two years of age, (OR adj = 2.2, 95% CI 1.3–3.6), (table 2 ). The effect appeared most pronounced when there was maternal smoking during the first and/or second trimester, (OR adj = 2.5, 95 % CI 1.5–4.0), but not thereafter in a separate analysis using the entire material and adjusting for the effect of ETS (data not shown).
Exposure to ETS alone or in combination with exposure in utero tended to be associated with an increased risk of recurrent wheezing (OR adj = 1.6, 95 % CI 1.2 – 2.3). The risk estimates were similar in the different exposure groups for doctor's diagnosed asthma and any wheezing up to two years of age, respectively (table 2 ). These effects were independent of gender of the infant (data not shown).
Exposure to cigarette smoking during pregnancy and of maternal smoking during the child's first year of life increased the risk of recurrent wheezing as well as of doctor's diagnosed asthma and any wheezing, respectively, at one year of age, in a similar way as reported in table 2 . Reported paternal smoking during the child's first year of life had no additional effect on any of the outcomes under study (data not shown).
The results of dose-response analyses were not conclusive i.e. neither confirmed nor excluded a trend, mainly due to low numbers of subjects in the high exposure groups (data not shown). Furthermore, there was no clear evidence of interaction between smoking and heredity or gender (data not shown).
This study provides strong evidence that exposure in utero to maternal smoking is important for development of recurrent wheezing during the first two years of life, irrespective of exposure to ETS after birth. Similar results have been published by others, but generally without separating the effects of exposure in utero exposure to ETS during the first few years of life [ 20 , 21 ]. The study by Lux and coworkers, however, clearly indicates that maternal smoking restricted to pregnancy causes wheezing [ 11 ]. The design of their study is similar to ours and allows for separation of the effects of different exposure periods but data about smoking during pregnancy were only obtained for gestational weeks 30–32. In the present study information about maternal smoking during pregnancy encompassed the various trimesters in detail. Our data suggest an effect with exposure particularly during early pregnancy. If so, this is possibly a consequence of an effect on intra-uterine growth [ 1 ].
An effect of maternal smoking on the foetus has also been documented by several studies of pulmonary function in neonates [ 4 , 6 , 22 , 23 ]. Most of these studies indicate hampered expiratory flows as indices of a detrimental effect. In a study by Hoo and co-workers prematurely born infants, in average seven weeks, were investigated and maternal smoking was associated with reduced pulmonary function [ 24 ]. The spirometric data in neonates only give indirect evidence of a reduction in airway diameter. For obvious reasons no direct studies of morphological consequences of exposure to smoking in the neonate lung have been carried out in healthy term babies. However, in children with sudden infant death increased airway thickness has been associated with maternal smoking of more than 20 cigarettes daily [ 25 ]. To which extent this effect stems from exposure prior to or after birth is not clear.
In many studies the role of ETS, as a determinant of childhood asthma, has been investigated but in most of them without due consideration of the separate influence of maternal smoking during pregnancy [ 8 , 26 ]. In a meta-analysis by Strachan and Cook a pooled risk estimate of 1.57 was found for lower respiratory illness in relation to smoking by either parent [ 7 ]. The relative contributions of pre- and postnatal smoking were not disentangled. In the study by Lux, an OR of 1.3 was found for exposure to ETS exclusively [ 11 ]. Possibly, the effect of exposure in utero may be the more important which is also supported by our data.
In Sweden exposure of children to tobacco smoking has been reduced to levels which are low in an international perspective. This is probably a consequence of a very active health policy and an effective maternal and child health care. During the study there was also a campaign "Smokefree children" through the Child Health Centres which reached almost all (99.5%) of the families when the baby was new-born (Statistics from Child Health Centres, Stockholm County Council, 1995). The effects of ETS are possibly diminished because of an overall awareness of the detrimental effects of exposure. This is supported by the finding that 94% of the parents reportedly never exposed their children to ETS. Exposure of the foetus, on the other hand, cannot be avoided by the pregnant mothers who are active smokers.
Participation in the study is most likely to have been affected by parental awareness of health hazards associated with cigarette smoking. Thus, smokers may to a higher extent than non-smokers have chosen not to join the study. A study of non- responders and actively excluded families of the BAMSE study showed that these parents smoked more than those included in the cohort [ 15 ]. This would render the study base less representative of the population, but in relation to tobacco smoke exposure probably not affect the risk estimate of smoking related health effects. Furthermore, parents with allergic diseases would possibly be more willing to join the original cohort but we found no such selection. We had the advantage of a large sample, allowing for the assessment of effects of exposures in subgroups of infants. Yet, possible biases must be taken into account. Smoking tobacco was found to be associated with a negative family history of allergic disease. Furthermore, we based the risk estimation on maternal smoking only, for obvious reasons regarding smoking in pregnancy, but this may lead to some misclassification of exposure postnatally. The effects of the role of ETS will be studied more in detail in the future follow up if the cohort.
The main implication of this study is that smoking cessation programmes need to be targeted on childbearing ages. In maternal health care such efforts should focus not only on those who are already pregnant, but also on women who plan to conceive.
Horta BL, Victora CG, Menezes AM, Halpern R, Barros FC: Low birthweight, preterm births and intrauterine growth retardation in relation to maternal smoking. Paediatr Perinat Epidemiol 1997, 11: 140–51.
Article CAS PubMed Google Scholar
Foundas M, Hawkrigg NC, Smith SM, Devadason SG, Le Souef PN: Urinary cotinine levels in early pregnancy. Aust N Z J Obstet Gynaecol 1997, 37: 383–6.
Lodrup Carlsen KC, Jaakkola JJ, Nafstad P, Carlsen KH: In utero exposure to cigarette smoking influences lung function at birth. Eur Respir J 1997, 10: 1774–9.
Stick SM, Burton PR, Gurrin L, Sly PD, Le Souef PN: Effects of maternal smoking during pregnancy and family history of asthma on respiratory function in newborn infants. Lancet 1996, 348: 1060–4.
Elliot JG, Carroll NG, James AL, Robinson PJ: Airway alveolar attachment points and exposure to cigarette smoke in utero. Am J Respir Crit Care Med 2003, 167 (1) : 45–9.
Article PubMed Google Scholar
Gilliland FD, Berhane K, Li YF, Rappaport EB, Peters JM: Effects on early onset asthma and in utero exposure to maternal smoking on childhood lung function. Am J Respir Crit Care Med 2003, 167 (6) : 917–24.
Strachan DP, Cook DG: Parental smoking and lower respiratory illness in infancy and early childhood. Thorax 1997, 52: 905–914.
Article CAS PubMed PubMed Central Google Scholar
Nafstad P, Kongerud J, Botten G, Hagen JA, Jaakkola JJ: The role of passive smoking in the development of bronchial obstruction during the first 2 years of life. Epidemiology 1997, 8: 293–297.
Wennergren G, Amark M, Amark K, Oskarsdottir S, Sten G: Wheezing bronchitis reinvestigated at the age of 10 years. Acta Paediatrica 1997, 86: 351–5.
Rylander E, Pershagen G, Eriksson M, Nordvall L: Parental smoking and other risk factors for wheezing bronchitis in children. Eur J Epidemiol 1993, 9: 517–526.
Lux AL, Henderson AJ, Pocock SJ, the ALSPAC Study Team: Wheeze associated with prenatal tobacco smoke exposure: a prospective, longitudinal study. Arch Dis Child 2000, 83: 307–12.
Von Mutius E: Environmental factors influencing the development and progression of pediatric asthma. J Allergy Clin Immunol 2002, 109 (Suppl 6) : S525–32.
Lannero E, Kull I, Wickman M, Pershagen G, Nordvall SL: Environmental risk factors for allergy and socioeconomic status in a birth cohort (BAMSE). Pediatr Allergy Immunol 2002, 13: 1–8.
Article Google Scholar
Kull I, Wickman M, Lilja G, Nordvall SL, Pershagen G: Breastfeeding and allergic diseases in infants – a prospective birth cohort study. Arch Dis Child 2002, 87 (6) : 478–81.
Wickman M, Kull I, Pershagen G, Nordvall SL: The BAMSE Project: presentation of a prospective longitudinal birth cohort study. Pediatr Allergy Immunol 2002, 13 (Suppl 15) : 11–13.
Occupations in Population and Housing Census 1985 (FoB 85) according to Nordic standard occupational classification (Nordisk yrkesklassificering, NYK) and Swedish socio-economic classification (Socioekonomisk indelning, SEI) [Swedish]. Statistics Sweden, Stockholm. 1989) .
Wickman M, Nordvall SL, Pershagen G: Riskfactors in early childhood for sensitization to airborne allergens. Pediatr Allerg Immunol 1992, 3: 128–133.
Lindfors A, Wickman M, Hedlin G, Pershagen G, Rietz H, Nordvall SL: Indoor environmental risk factors in young asthmatics: a case-control study. Arch Dis Child 1995, 73: 408–412.
Asher MI, Weiland SK: The International Study of Asthma and Allergies in Childhood (ISAAC) ISAAC Steeriing Committee. Clin Exp Allergy 1998, (Suppl 5) : 52–66.
Gilliland FD, Li YF, Peters JM: Effects of maternal smoking during pregnancy and environmental tobacco smoke on asthma and wheezing in children. Am J Respir Crit Care Med 2001, 2: 429–36.
Li YF, Gilliland FD, Berhane K, McConnell R, Gauderman WJ, Rappaport EB, Peters JM: Effects of in utero and environmental tobacco smoke exposure on lung function in boys and girls with and without asthma. Am J Respir Crit Care Med 2000, 162: 2097–2104.
Gilliland FD, Berhane K, McConnell R, Gaudrman WJ, Vora H, Rappaport EB, Avol E, Peters JM: Maternal smoking during pregnancy, environmental tobacco smoke exposure and childhood lung function. Thorax 2000, 55: 271–6.
Young S, Sherrill DL, Arnott J, Diepeveen D, LeSouef PN, Landau LI: Parental factors affecting respiratory function during the first year of life. Pediatric Pulmonology 2000, 29: 331–40.
Hoo A-F, Henschen M, Dezateux C, Costeloe K, Stocks J: Respiratory function among preterm infants whose mothers smoked during pregnancy. Am J Respir Crit Care Med 1998, 158 (3) : 700–705.
Elliot J, Vullermin P, Robinson P: Maternal cigarette smoking is associated with increased inner airway wall thickness in children who die from sudden infant death syndrome. Am J Respir Crit Care Med 1998, 158: 802–06.
Cunningham J, O'Connor GT, Dockery DW, Speizer FE: Environmental tobacco smoke, wheezing, and asthma in children in 24 communities. Am J Respir Crit Care Med 1996, 153: 218–226.
Download references
Acknowledgements
Assistance by epidemiology assistant Lena Tollin, research nurse Inger Kull, research secretary Eva Hallner and data co-ordinator André Lauber, Department of Environmental Health, Stockholm County Council, and statistical support from Niklas Berglind. Institute of Environmental Medicine, Karolinska Institutet, are gratefully acknowledged.
The study was supported by: The Swedish Asthma and Allergy Association, Swedish Council for Building Research, Stockholm County Council, The Swedish Foundation for Health Care Sciences and Allergy Research (Vårdalstiftelsen), Sven Jerring Foundation and 3MPharma.
Author information
Authors and affiliations.
Institute of Environmental Medicine, Karolinska Institutet, Stockholm, Sweden
Eva Lannerö, Magnus Wickman & Goran Pershagen
Department of Paediatrics, Karolinska University Hospital, Huddinge, Sweden
Eva Lannerö
Department of Occupational and Environmental Health, Stockholm County Council, Stockholm, Sweden
Centre for Allergy Research, Karolinska Institutet, Stockholm, Sweden
Magnus Wickman
Department of Women's and Children's Health, Uppsala University, Uppsala, Sweden
Lennart Nordvall
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Eva Lannerö .
Additional information
Competing interests.
The author(s) declare that they have no competing interests.
Authors' contributions
All four authors have made substantial intellectual contributions to this study and have also been involved in the BAMSE project since it started.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Reprints and permissions
About this article
Cite this article.
Lannerö, E., Wickman, M., Pershagen, G. et al. Maternal smoking during pregnancy increases the risk of recurrent wheezing during the first years of life (BAMSE). Respir Res 7 , 3 (2006). https://doi.org/10.1186/1465-9921-7-3
Download citation
Received : 31 May 2005
Accepted : 05 January 2006
Published : 05 January 2006
DOI : https://doi.org/10.1186/1465-9921-7-3
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Environmental Tobacco Smoke
- Maternal Smoking
- Tobacco Smoke Exposure
- Postnatal Exposure
Respiratory Research
ISSN: 1465-993X
- General enquiries: [email protected]
Maternal prenatal smoking linked to poor academic outcomes in children
- Download PDF Copy
Smoking harms almost every part of your body. But if you smoke when pregnant, the toxic chemicals in tobacco will also harm your unborn baby, with new research showing that it could lead to reduced academic outcomes at school.
In a systematic review of 19 studies and 1.25 million participants, researchers at the University of South Australia along with a team at Curtin University, SAHMRI, Harvard University and others found that 79% of studies reported reduced academic achievement in children exposed to maternal prenatal smoking.
An additional meta-analysis of eight primary studies with 723,877 participants showed that children exposed to maternal prenatal tobacco smoking were 49% more likely to struggle with poor academic achievement in comparison to those who had not been exposed to smoking in utero.
In Australia, 8.7% (or 26,433) of all mothers who gave birth in 2021 smoked at some time during their pregnancy.
Lead researcher, UniSA's Dr. Bereket Duko, says that despite what is already known about smoking, research is still uncovering additional negative effects.
For decades, agencies across the globe have pushed anti-smoking campaigns about the dangers of smoking. But despite these efforts, tobacco smoking remains a pervasive global public health issue. Prenatal smoking is known to cause multiple pregnancy complications, including a higher risk of miscarriage, stillbirth, restricted growth and development, and serious birth defects. It is also linked with adverse mental health outcomes and behavioral issues. Our new research adds to this, by showing that maternal prenatal smoking has a significant risk of limiting a child's academic performance, putting them well behind their peers at school. We all want children to have the best start in life. But clearly, we must do better to educate mothers and families about the noxious effects of smoking while pregnant on mother and baby. Remember, the fight against smoking is not one we have already won. Yes, we have made big steps to reduce the number of people smoking, and we have made many aware of the health risks. But this is an ongoing battle, and we must continue to educate people about the dangers of tobacco so that the next generations do not unnecessarily suffer." Dr. Bereket Duko, Lead Researcher, UniSA
University of South Australia
Duko, B., et al . (2024). The effect of maternal prenatal tobacco smoking on offspring academic achievement: A systematic review and meta-analysis. Addictive Behaviors . doi.org/10.1016/j.addbeh.2024.107985 .
Posted in: Child Health News | Medical Research News | Women's Health News
Tags: Alcohol , Baby , Birth Defects , Chemicals , Children , Cigarette , Drugs , In Utero , Mental Health , Miscarriage , Pregnancy , Prenatal , Public Health , Research , Smoking , Stillbirth , Tobacco
Suggested Reading

Cancel reply to comment
- Trending Stories
- Latest Interviews
- Top Health Articles

Global and Local Efforts to Take Action Against Hepatitis
Lindsey Hiebert and James Amugsi
In this interview, we explore global and local efforts to combat viral hepatitis with Lindsey Hiebert, Deputy Director of the Coalition for Global Hepatitis Elimination (CGHE), and James Amugsi, a Mandela Washington Fellow and Physician Assistant at Sandema Hospital in Ghana. Together, they provide valuable insights into the challenges, successes, and the importance of partnerships in the fight against hepatitis.

Addressing Important Cardiac Biology Questions with Shotgun Top-Down Proteomics
In this interview conducted at Pittcon 2024, we spoke to Professor John Yates about capturing cardiomyocyte cell-to-cell heterogeneity via shotgun top-down proteomics.

A Discussion with Hologic’s Tim Simpson on the Future of Cervical Cancer Screening
Tim Simpson
Hologic’s Tim Simpson Discusses the Future of Cervical Cancer Screening.

Latest News

Newsletters you may be interested in
Your AI Powered Scientific Assistant
Hi, I'm Azthena, you can trust me to find commercial scientific answers from News-Medical.net.
A few things you need to know before we start. Please read and accept to continue.
- Use of “Azthena” is subject to the terms and conditions of use as set out by OpenAI .
- Content provided on any AZoNetwork sites are subject to the site Terms & Conditions and Privacy Policy .
- Large Language Models can make mistakes. Consider checking important information.
Great. Ask your question.
Azthena may occasionally provide inaccurate responses. Read the full terms .
While we only use edited and approved content for Azthena answers, it may on occasions provide incorrect responses. Please confirm any data provided with the related suppliers or authors. We do not provide medical advice, if you search for medical information you must always consult a medical professional before acting on any information provided.
Your questions, but not your email details will be shared with OpenAI and retained for 30 days in accordance with their privacy principles.
Please do not ask questions that use sensitive or confidential information.
Read the full Terms & Conditions .
Provide Feedback


- News Releases
Prenatal smoking risks academic achievement of unborn babies
University of South Australia
Maternal prenatal smoking has a significant risk of limiting a child’s academic performance.
Credit: "Cigarette" by Sudipto_Sarkar is licensed under CC BY-NC-ND 2.0.
Smoking harms almost every part of your body. But if you smoke when pregnant, the toxic chemicals in tobacco will also harm your unborn baby, with new research showing that it could lead to reduced academic outcomes at school.
In a systematic review of 19 studies and 1.25 million participants, researchers at the University of South Australia along with a team at Curtin University , SAHMRI , Harvard University and others* found that 79% of studies reported reduced academic achievement in children exposed to maternal prenatal smoking.
An additional meta-analysis of eight primary studies with 723,877 participants showed that children exposed to maternal prenatal tobacco smoking were 49% more likely to struggle with poor academic achievement in comparison to those who had not been exposed to smoking in utero.
In Australia , 8.7% (or 26,433) of all mothers who gave birth in 2021 smoked at some time during their pregnancy .
Lead researcher, UniSA’s Dr Bereket Duko , says that despite what is already known about smoking, research is still uncovering additional negative effects.
“For decades, agencies across the globe have pushed anti-smoking campaigns about the dangers of smoking. But despite these efforts, tobacco smoking remains a pervasive global public health issue,” Dr Bereket says.
“Prenatal smoking is known to cause multiple pregnancy complications, including a higher risk of miscarriage, stillbirth, restricted growth and development, and serious birth defects. It is also linked with adverse mental health outcomes and behavioural issues.
“Our new research adds to this, by showing that maternal prenatal smoking has a significant risk of limiting a child’s academic performance, putting them well behind their peers at school.
“We all want children to have the best start in life. But clearly, we must do better to educate mothers and families about the noxious effects of smoking while pregnant on mother and baby.
“Remember, the fight against smoking is not one we have already won. Yes, we have made big steps to reduce the number of people smoking, and we have made many aware of the health risks. But this is an ongoing battle, and we must continue to educate people about the dangers of tobacco so that the next generations do not unnecessarily suffer.”
Notes or editors:
*Additional partners include the University of Iceland, the University of Queensland, the Norwegian Institute of Public Health, and the University of Sydney.
…………………………………………………………………………………………………………………………
Contact for interview: Dr Bereket Duko M: +61 410 350 140 E: [email protected] Media contact: Annabel Mansfield M: +61 479 182 489 E: [email protected]
Addictive Behaviors
10.1016/j.addbeh.2024.107985
Method of Research
Meta-analysis
Subject of Research
Not applicable
Article Title
The effect of maternal prenatal tobacco smoking on offspring academic achievement: A systematic review and meta-analysis
Article Publication Date
15-Jun-2024
COI Statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of news releases posted to EurekAlert! by contributing institutions or for the use of any information through the EurekAlert system.
Original Source
- Open access
- Published: 01 November 2022
Maternal factors during pregnancy influencing maternal, fetal, and childhood outcomes
- Louis J. Muglia ORCID: orcid.org/0000-0002-0301-8770 1 , 2 ,
- Katrien Benhalima 3 ,
- Stephen Tong 4 , 5 &
- Susan Ozanne 6
BMC Medicine volume 20 , Article number: 418 ( 2022 ) Cite this article
14k Accesses
25 Citations
36 Altmetric
Metrics details
Enhancing pregnancy health is known to improve the mother’s and offspring’s life-long well-being. The maternal environment, encompassing genetic factors, impacts of social determinants, the nutritional/metabolic milieu, and infections and inflammation, have immediate consequences for the in utero development of the fetus and long-term programming into childhood and adulthood. Moreover, adverse pregnancy outcomes such as preterm birth or preeclampsia, often attributed to the maternal environmental factors listed above, have been associated with poor maternal cardiometabolic health after pregnancy. In this BMC Medicine article collection, we explore a broad spectrum of maternal characteristics across pregnancy and postnatal phenotypes, anticipating substantial cross-fertilization of new understanding and shared mechanisms around diverse outcomes. Advances in the ability to leverage ‘omics across different platforms (genome, transcriptome, proteome, metabolome, microbiome, lipidome), large high-dimensional population databases, and unique cohorts are generating exciting new insights: The first articles in this collection highlight the role of placental biomarkers of preterm birth, metabolic influences on fetal and childhood growth, and the impact of common pre-existing maternal disorders, obesity and smoking on pregnancy outcomes, and the child’s health. As the collection grows, we look forward to seeing the connections emerge across maternal, fetal, and childhood outcomes that will foster new insights and preventative strategies for women.
The extraordinary, foundational months of pregnancy are a time of emergence of a new life for the conceptus and remarkable physiological and psychological adaptation by the mother. The orchestration of mutual communication between the mother and fetus is the driver of long-term health. It is shaped primarily by the maternal environment, with its genetic, physiologic, nutritional, inflammatory/infection, and psychological components. It has been repeatedly recognized that the in utero environment programs the fetus for lifelong health—the Barker Hypothesis—and pregnancy complications such as preeclampsia and preterm birth impact maternal cardiovascular health [ 1 , 2 ].
This special collection of articles by BMC Medicine seeks to synthesize information related to maternal and offspring outcomes associated with in utero exposures across pregnancy phenotypes and complications. Among the most common maternal traits that impact multiple aspects of fetal outcomes are maternal undernutrition and, more often, maternal overnutrition/obesity, associated with complications from development in an obesogenic environment and influences of gestational diabetes mellitus. In addition, the mechanisms leading to abnormalities in gestational duration and an increased risk for adverse outcomes such as preterm birth are central research targets [ 3 ]. The growing opportunity to interrogate “big data” with artificial intelligence or machine learning tools will accelerate this research and help to determine pregnancy interventions that are both safe and effective [ 4 , 5 , 6 , 7 ].
The editors believe that providing novel insights on exposures and outcomes across pregnancy phenotypes will be mutually informative as many driving determinants are shared. In this editorial, we will highlight some initial contributions to this collection and the new information that has been revealed.
Towards better health of mother and child—novel insights and potential pathways for intervention
Revealing underlying mechanisms in preterm birth and potential links to polycystic ovary syndrome.
Adverse pregnancy outcomes are common and have generally been refractory to interventions designed to reduce their incidence. Of all obstetric complications, preterm birth towers above nearly all others as the most severe. Affecting 8–10% of all pregnancies, it is depressingly common and can leave the newborn with a lifelong legacy of health deficits [ 8 ]: from subtle decrements in developmental outcomes for those born “late preterm” to profound disabilities for those born “extremely preterm” (cerebral palsy, chronic lung conditions, major learning problems) [ 9 ].
Spontaneous idiopathic preterm birth has been among the greatest challenges. Until now, there are no generally effective therapeutic interventions, and predictive biomarkers, while beginning to emerge, are limited. The lack of mechanistic insight has resulted in preterm birth being a long-standing leading cause of infant mortality and mortality in children under 5 years of age [ 10 ]. However, research of the underlying mechanisms in preterm birth has been greatly accelerated by using hypothesis-free interrogation of large data sets across ‘omics platforms and medical record information using advanced bioinformatic strategies [ 4 , 5 , 6 , 7 ].
As part of this collection, Tiensuu and colleagues [ 11 ] present new data for a candidate biomarker for preterm birth that may also help unravel the underlying mechanisms and is a potential target for interventions. In this study, the investigators evaluated whether placental proteins associated with spontaneous preterm birth can be identified using proteomics. Intriguingly, protein and mRNA levels of alpha-1 antitrypsin (AAT)/SERPINA1 were found to be downregulated on both the maternal and fetal sides of the placenta. This finding served as a basis to investigate whether damaging genetic DNA variations in AAT were also associated with spontaneous preterm birth through whole exome sequencing—and indeed, they were. After revealing this association, the authors performed functional studies, indicating that the downregulation of AAT affects the actin cytoskeletal pathways and extracellular matrix organization.
Beyond identifying relevant biomarkers, there is a strong need for interventions to prevent adverse pregnancy outcomes. The Tiensuu study moves forward with one strong candidate for such an intervention. Moreover, their approach of utilizing multiple association strategies to provide further evidence for a particular finding can be applied across various disease phenotypes.
Major risk factors for preterm birth have long been elucidated [ 8 , 9 ], such as prior preterm birth, early rupture of membranes, or co-existing medical conditions such as polycystic ovary syndrome (PCOS), as reported by others in this special collection [ 12 ]. Rocha and colleagues address an interesting question: for those in their second pregnancy and birth preterm, do risk factors associated with their preterm birth differ depending on whether or not their first infant was born preterm?
To address this question, the authors examined a large retrospective dataset from Brazil representing 1.7 million births [ 13 ]. They focused on women who had a preterm birth in their second pregnancy and split them according to whether their first pregnancy was delivered at full term (> 37 weeks gestation, “incident preterm birth” cohort) or they previously had a preterm birth (< 37 weeks gestation, “recurrent preterm birth” cohort).
Interestingly, the incident but not the recurrent preterm birth cohort had significant associations with household overcrowding, variations in ethnicity (being black, mixed-race, or indigenous had raised risks), being a younger mother (14–19 years), and having had a prior cesarean section, with odds ratios ranging from 1.04 to 1.34. Both cohorts were associated with reduced prenatal visits with higher odds ratios in the incidence preterm birth cohort. Notably, many of these risk factors likely reflect socioeconomic deprivation, stress, low educational attainment, and smoking—established risk factors for preterm birth [ 8 ].
Surprisingly, in both cohorts, being single or a widow conferred a 10–15% reduced risk of preterm birth compared to those who were married or in a civil union. While interesting, this finding is difficult to explain, and we do not suggest encouraging women to be single is a promising public health strategy to reduce preterm birth rates.
In another contribution to this collection, Subramanian and colleagues [ 12 ] present data indicating a convincing link between preterm birth and PCOS, a condition affecting 10% of women. While defined by a varied constellation of signs and symptoms—cysts on the ovary, biochemical or clinical evidence of androgen excess, oligo/anovulation [ 14 ]—PCOS is, at its heart, a metabolic disorder [ 15 ]. As a chronic condition that never retreats, those affected incur the risk of developing metabolic-related conditions as they age, especially diabetes and obesity [ 15 ].
Given PCOS is the most common endocrine disorder among women of reproductive age, it will invariably intersect with many pregnancies. In their retrospective study, Subramanian et al. examined the link between a pre-specified set of serious obstetric complications, including preterm birth, fetal size, mode of birth and stillbirth, and PCOS based on just under 140,000 pregnancies in the UK, of which 27,586 were affected by PCOS. While the lift in preterm birth risk in women affected by PCOS was modest (an 11% relative rise on the adjusted odds ratio), it could be substantiated by further sub-analyses. These findings concur with a recent study in a Swedish population, indicating an apparent doubling in the risk of extreme preterm birth < 28 weeks gestation in women suffering from PCOS and, thus, an even larger effect size [ 16 ].
But how is the link between PCOS and preterm birth explained? The authors muse over potential causes such as a shared genetic polymorphism between preterm birth and PCOS or a dysregulated hypothalamic-pituitary-adrenal axis tipping off a biological cascade that ends in spontaneous preterm birth. However, as the team did not adjust for important pregnancy-induced complications strongly associated with both PCOS and preterm birth (such as gestational diabetes and hypertensive disorders), more likely, the presence of such complications led to the excess in preterm births. They also found PCOS associated with a modestly increased risk of a cesarean section but no apparent link with stillbirth. While this finding seems reassuring, the study was likely underpowered to explore this outcome.
Finally, there may be a fascinating biological message buried within their apparently plain finding that PCOS is not associated with the birth of babies that are either small or large for gestational age. It suggests placental function may be surprisingly resistant to the multiple metabolic perturbations occurring within the mother, which would be a reassuring finding.
The impact of environmental exposures—metabolic in utero environment, obesity, and smoking during pregnancy
In addition to adverse obstetric outcomes such as preterm birth and associated risk factors, obesity during pregnancy is of great concern. Obesity rates continue to increase across the globe in all age groups in the population, including women of childbearing age [ 17 ]. Consequently, in a growing number of countries, over half of the pregnant women are now either obese or overweight.
Obesity is associated with immediate detrimental consequences for the mother and baby, including increased risk of gestational diabetes, preeclampsia, and the need for a caesarian section [ 18 ]. In addition, it is established that children born to obese women are at increased risk of becoming obese and developing type 2 diabetes and cardiovascular diseases. Furthermore, evidence suggests that at least part of this transmission of poor cardio-metabolic health from mother to child is driven by non-genetic factors. Notably, the latter provides an opportunity for early intervention before disease pathology is established [ 19 ].
Currently, it is not known which children born to obese mothers will follow a higher-than-normal body mass index growth trajectory and become overweight and ultimately obese. In this article collection, Gomes and colleagues address this knowledge deficit using the mother-child cohort study Programming of Enhanced Adiposity Risk in Childhood–Early Screening (PEACHES), which comprised 1671 mothers with pre-conception obesity and without (controls) and their offspring. They identified a “high-risk” subpopulation of offspring born to obese mothers susceptible to early upper deviations from healthy weight gain trajectories present within the first few months of life and progressing to overweight/obesity by 5 years of age. Hence, they established a novel sequential prediction system to allow early-risk stratification and re-evaluation to prevent a “higher-than-normal BMI growth pattern” at a subclinical stage preceding overweight. Maternal smoking and excessive gestational weight gain were the strongest predictors of a higher-than-normal BMI growth pattern by 3 months of age. Importantly, they validated these findings in the independent Perinatal Prevention of Obesity (PEPO) cohort, comprising 11,730 mother-child pairs recruited around 6 years of age. These findings take us a step closer to developing cost-effective and personalized advice and measures, counteracting the risk of early excess weight gain and ultimately preventing childhood obesity.
Several articles in this collection have focused on the metabolic environment in utero and the impact of environmental exposures in pregnancy on the mother’s and offspring’s long-term metabolic health. For example, the large mother-offspring Asian cohort study Growing Up in Singapore Towards healthy Outcomes (GUSTO), consisting of 1247 women from Singapore, studied the changes of 480 lipid species in the plasma of women during pregnancy (antenatal vs postnatal) and their offspring during development (cord blood and 6-year-old child plasma) [ 20 ]. This study demonstrated that around 36% of the profiled lipids increased in circulation during pregnancy, with phosphatidylethanolamine levels changing the most compared to pre-pregnancy. Compared to the gestating mother, the cord blood showed a lower concentration of most lipids, and a higher concentration of lysophospholipids, suggesting the specific developmental needs of the growing fetus. Pre-pregnancy BMI was specifically associated with a decrease in the levels of phospholipids, sphingomyelin, and several triacylglycerol species in pregnancy, highlighting the importance of managing obesity before pregnancy. Notably, lipid species associated with the child’s BMI were very similar to those associated with the adult’s BMI. This overlapping effect of adiposity, as observed in 6-year-old children and postnatal mothers in this study, may be influenced by the similarities in the diet apart from other factors such as genetics and shared lifestyle. The findings of this study were validated in an independent Caucasian birth cohort and provide an important resource for future research targeting early nutritional interventions to benefit maternal and child metabolic health.
Also focusing on the influencing factors on metabolic health, a Swedish nationwide register-based study investigated the association between maternal smoking during pregnancy and type 1 diabetes in the offspring [ 21 ]. The cohort consisted of nearly three million children born between the years 1983 and 2014, with follow-up until 2020. In addition, a nested case-control study was performed comparing children with type 1 diabetes to their age-matched siblings. A total of 18,617 children developed type 1 diabetes. Interestingly, maternal smoking during pregnancy was associated with a 22% lower risk of offspring type 1 diabetes in the full cohort. Although these data suggest a protective effect of maternal smoking on the risk for offspring to develop type 1 diabetes, mechanistic studies are needed to elucidate the underlying pathways behind this link. Despite these findings, we emphasize that smoking during pregnancy should be strongly advised against since it has severe effects on fetal and childhood health [ 21 ].
For example, a longitudinal study by Howell and colleagues in this collection provides evidence that maternal smoking is also associated with shorter offspring telomere length [ 22 ]. Acting as a mitotic clock to the cell, these hexameric repeat sequences found at the ends of chromosomes shorten with cell division [ 23 ] and, as shown recently, as a consequence of oxidative damage. Therefore, they represent good biomarkers of cellular aging and also exposure to oxidative damage [ 23 ]. Accelerated aging has been suggested as one potential mechanism linking suboptimal in utero exposures to long-term health. However, most evidence has primarily come from studies of suboptimal in utero nutritional exposures [ 24 ].
Howell et al. demonstrated that maternal prenatal smoking predicted greater telomere shortening by measuring the telomere length in buccal cells in infants from 4 to 18 months of age. They also showed that the relationship between maternal prenatal smoking and offspring attention-deficit/hyperactivity disorder (ADHD) was modulated by telomere length. Paradoxically, ADHD was associated with less telomere attrition for children whose mothers smoked. It is not known if these differences in buccal cell telomere length are reflective of the differences in other cell types, such as those in the brain. However, if similar differences are also present in brain tissue, this finding could indicate delayed maturation of cortical cells, which could provide a mechanistic link to ADHD.
Conclusions
As demonstrated by the initial series of articles published in this collection, the ability to utilize now more refined technologies to elucidate the underlying mechanisms that drive adverse pregnancy outcomes, such as preterm birth and metabolic risks for both the mother and fetus, has revealed new insights and potential pathways for intervention. Moreover, a deeper understanding of how these diverse outcomes are influenced by maternal co-morbidities such as maternal PCOS or smoking status is emerging.
However, to have a real impact on public health, these robust, reliable data and their implications need to be implemented in physician practice and be used for therapy development for a historically under-explored and invested group—pregnant women.
It will be essential to figure out how these findings can be used to tackle challenges related to lifestyle factors such as maternal obesity or smoking that have been refractory to preventive strategies and interventions. As the recognition of these influencing factors on maternal, fetal, and childhood outcomes across the lifespan emerges, we are encouraged that it will ultimately benefit the mother’s and child’s health.
We have enjoyed learning from this initial set of articles and look forward to future contributions to this collection.
Availability of data and materials
All data discussed in this editorial are included in this published article.
Parrettini S, Caroli A, Torlone E. Nutrition and metabolic adaptations in physiological and complicated pregnancy: focus on obesity and gestational diabetes. Front Endocrinol (Lausanne). 2020;11:611929.
Article Google Scholar
Sadovsky Y, Mesiano S, Burton GJ, Lampl M, Murray JC, Freathy RM, et al. Advancing human health in the decade ahead: pregnancy as a key window for discovery: a Burroughs Wellcome Fund Pregnancy Think Tank. Am J Obstet Gynecol. 2020;223(3):312–21.
Article PubMed PubMed Central Google Scholar
Jain VG, Monangi N, Zhang G, Muglia LJ. Genetics, epigenetics, and transcriptomics of preterm birth. Am J Reprod Immunol. 2022;88:e13600.
Article PubMed CAS Google Scholar
Tarca AL, Pataki BA, Romero R, Sirota M, Guan Y, Kutum R, et al. Crowdsourcing assessment of maternal blood multi-omics for predicting gestational age and preterm birth. Cell Rep Med. 2021;2(6):100323.
Article PubMed PubMed Central CAS Google Scholar
Espinosa C, Becker M, Marić I, Wong RJ, Shaw GM, Gaudilliere B, et al. Data-driven modeling of pregnancy-related complications. Trends Mol Med. 2021;27(8):762–76.
Marić I, Tsur A, Aghaeepour N, Montanari A, Stevenson DK, Shaw GM, et al. Early prediction of preeclampsia via machine learning. Am J Obstet Gynecol MFM. 2020;2(2):100100.
Article PubMed Google Scholar
Stevenson DK, Wong RJ, Aghaeepour N, Marić I, Angst MS, Contrepois K, et al. Towards personalized medicine in maternal and child health: integrating biologic and social determinants. Pediatr Res. 2021;89(2):252–8.
Frey HA, Klebanoff MA. The epidemiology, etiology, and costs of preterm birth. Semin Fetal Neonatal Med. 2016;21(2):68–73.
Colvin M, McGuire W, Fowlie PW. Neurodevelopmental outcomes after preterm birth. BMJ. 2004;329(7479):1390–3.
Liu L, Oza S, Hogan D, Perin J, Rudan I, Lawn JE, Cousens S, Mathers C, Black RE. Global, regional, and national causes of child mortality in 2000-13, with projections to inform post-2015 priorities: an updated systematic analysis. Lancet. 2015;385(9966):430–40. https://doi.org/10.1016/S0140-6736(14)61698-6 . Epub 2014 Sep 30.
Tiensuu H, Haapalainen AM, Tissarinen P, Pasanen A, Maatta TA, Huusko JM, et al. Human placental proteomics and exon variant studies link AAT/SERPINA1 with spontaneous preterm birth. BMC Med. 2022;20(1):141.
Subramanian A, Lee SI, Phillips K, Toulis KA, Kempegowda P, O’Reilly MW, et al. Polycystic ovary syndrome and risk of adverse obstetric outcomes: a retrospective population-based matched cohort study in England. BMC Med. 2022;20(1):1–3.
Rocha AS, de Cassia R-SR, Fiaccone RL, Paixao ES, Falcao IR, Alves FJO, et al. Differences in risk factors for incident and recurrent preterm birth: a population-based linkage of 3.5 million births from the CIDACS birth cohort. BMC Med. 2022;20(1):111.
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod. 2004;19(1):41–7.
Anagnostis P, Tarlatzis BC, Kauffman RP. Polycystic ovarian syndrome (PCOS): long-term metabolic consequences. Metabolism. 2018;86:33–43.
Valgeirsdottir H, Sundstrom Poromaa I, Kunovac Kallak T, Vanky E, Akhter T, Roos N, et al. Polycystic ovary syndrome and extremely preterm birth: a nationwide register-based study. PLoS One. 2021;16(2):e0246743.
Website of World Obesity. https://www.worldobesity.org/ . Accessed 22 Oct 2022.
Poston, et al. Preconceptional and maternal obesity: epidemiology and health consequences. Lancet Diabetes Endocrinol. 2016;4:1025–36.
Inzani I, Ozanne SE. Programming by maternal obesity: a pathway to poor cardiometabolic health in the offspring. Proc Nutr Soc. 2022;29:1–6.
Mir SA, Chen L, Burugupalli S, Burla B, Ji S, Smith AA, et al. Population-based plasma lipidomics reveals developmental changes in metabolism and signatures of obesity risk: a mother-offspring cohort study. BMC Med. 2022;20(1):1–8.
Wei Y, Andersson T, Edstorp J, Löfvenborg JE, Talbäck M, Feychting M, Carlsson S. Maternal smoking during pregnancy and type 1 diabetes in the offspring: a nationwide register-based study with family-based designs. BMC Med. 2022;20(1):240. https://doi.org/10.1186/s12916-022-02447-5 .
Salihu HM, Pradhan A, King L, Paothong A, Nwoga C, Marty PJ, et al. impact of intrauterine tobacco exposure on fetal telomere length. Am J Obstet Gynecol. 2015;212(2):205.
Blackburn, et al. Human telomere biology: a contributory and interactive factor in ageing, disease risks and protection. Science. 2015;350:1193–8.
Tarry-Adkins JL, Ozanne SE. The impact of early nutrition on the ageing trajectory. Proc Nutr Soc. 2014;73(2):289–301.
Download references
Acknowledgements
We thank Dr. Susanne Kröncke for her outstanding editorial support.
KB is the recipient of a “Fundamenteel Klinisch Navorserschap FWO Vlaanderen.”
None of the authors received funding for this editorial.
Author information
Authors and affiliations.
Burroughs Wellcome Fund, Research Triangle Park, Durham, NC, USA
Louis J. Muglia
Cincinnati Children’s Hospital Medical Center, Department of Pediatrics, University of Cincinnati College of Medicine, Cincinnati, OH, USA
University Hospital Gasthuisberg, KU Leuven, Leuven, Belgium
Katrien Benhalima
University of Melbourne, Melbourne, Australia
Stephen Tong
Mercy Perinatal, Heidelberg, Australia
University of Cambridge, Cambridge, UK
Susan Ozanne
You can also search for this author in PubMed Google Scholar
Contributions
All authors contributed equally and read and approved the final manuscript.
Corresponding author
Correspondence to Louis J. Muglia .
Ethics declarations
Ethics approval and consent to participate.
Not applicable; individual studies described in this editorial have appropriate ethical approvals.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Muglia, L.J., Benhalima, K., Tong, S. et al. Maternal factors during pregnancy influencing maternal, fetal, and childhood outcomes. BMC Med 20 , 418 (2022). https://doi.org/10.1186/s12916-022-02632-6
Download citation
Received : 22 October 2022
Accepted : 24 October 2022
Published : 01 November 2022
DOI : https://doi.org/10.1186/s12916-022-02632-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
BMC Medicine
ISSN: 1741-7015
- General enquiries: [email protected]

Annals of Global Health

- Download PDF (English) XML (English)
- Alt. Display
Original Research
Active and passive maternal smoking during pregnancy and birth outcomes: a study from a developing country.
- Shereen Hamadneh
- Jehan Hamadneh
Background: Smoking is one of the modifiable risk factors for adverse maternal and neonatal outcomes and is associated with low birth weight, preterm birth, respiratory, antepartum and intrapartum stillbirth, and perinatal death as well as long-term morbidity in offspring and sudden unexpected infant death. The rate of smoking in low- and middle-income countries is still relevantly high, and Jordan is no exception.
Objective: To investigate the effect of active and passive smoking during pregnancy on adverse pregnancy outcomes.
Methods: The case-control study was conducted in Jordan in June 2020. Healthy women with full-term singleton pregnancy (n = 180) were interviewed and stratified into three groups: Group I, active smokers; Group II, passive smokers; and Group III, nonsmokers. The study variables included demographic data, current pregnancy history, cotinine level of mothers and newborns, and perinatal outcomes. Statistical analysis was performed using the application package IBM SPSS 25. Various algorithms of statistical analysis were used depending on the type of distribution of feature and data quality. The threshold for statistical significance was set at p < 0.05.
Results: Active smokers had significantly lower gestational age at delivery compared to passive and nonsmoking women ( p = 0.038 and p = 0.003, respectively). Neonates from active smoking mothers had significantly lower birth weight compared to neonates from passive and nonsmoking women ( p = 0.016 and p = 0.019, respectively), significantly lower head and chest circumferences compared to babies from passive smokers ( p < 0.001 and p = 0.036, respectively), and significantly lower first-minute Apgar score compared to those from nonsmoking women ( p = 0.023). The urine cotinine level was significantly higher in both active and passive smoking women (both p < 0.01), and it was significantly higher in newborns who had been exposed to smoking in utero despite maternal active or passive smoking status (both p < 0.001). There was a weak negative correlation between urine cotinine level and birth weight: r = –0.14 for maternal cotinine level and r = –0.15 for neonate cotinine level.
Introduction
Smoking is a modifiable risk factor for adverse maternal and neonatal outcomes and is associated with maternal, fetal, and infant morbidity and mortality [ 7 ].
As shown in previous research, active and passive maternal smoking during pregnancy increases the risk of having a child with low birth weight [ 27 , 34 ] and significantly increases other negative pregnancy outcomes, such as preterm birth [ 17 , 21 ], respiratory distress [ 1 ], antepartum and intrapartum stillbirth [ 5 ], perinatal death [ 24 ], long-term morbidity in offspring [ 32 ], and sudden unexpected infant death [ 3 ].
Birth weight, length, and head and chest circumference at birth are the main indicators of fetal growth suppressed by maternal smoking [ 15 ]. It is not clear from the current body of evidence whether maternal smoking specifically affects head growth, though maternal smoking during pregnancy negatively affects fetal brain development [ 16 ].
The Apgar score is used as a standardized index of newborn health at birth and is strongly associated with the risk of neonatal and infant death [ 4 ]. According to [ 35 ], babies of smoking mothers had lower Apgar scores compared to those of nonsmokers. However, smoking during pregnancy is not an independent predictor of the Apgar score. It remains unclear whether quitting smoking during pregnancy affects the Apgar score [ 35 ].
The rate of smoking in low- and middle-income countries is still relevantly high [ 22 , 23 , 29 ]. The prevalence of active and passive smoking in Jordan is high [ 9 , 10 , 11 , 13 ], and it mirrors the husbands’ active smoking patterns [ 11 , 29 ]. Paternal smoking is considered an independent risk factor for fetal growth restriction [ 6 ] and stillbirth despite the maternal smoking status [ 28 ].
This study aimed to investigate the effects of active and passive smoking during pregnancy on adverse pregnancy outcomes.
The case-control study was conducted at the main hospital in Jordan (King Abdullah University Hospital (KAUH)) using a semistructured questionnaire in June 2020.
Inclusion criteria for the study were as follows: full-term singleton pregnancy, absence of chronic diseases (e.g., cardiovascular, kidney, endocrine diseases) and pregnancy complications (gestational hypertension and diabetes mellitus), accommodation at the north of Jordan, prenatal car, and delivery and postnatal care at KAUH Obstetrics and Gynecology Department as well as an agreement to participate in research and completion of at least 75% of the questionnaire.
The Institutional Research Board (IRB) at Jordan University of Science and Technology (University Review Committee for Research on Human) approved all study activities. All participants in this research were voluntary, and anyone could stop participating in the interview at any time. The consent form was prepared to get the agreement of the participants to be interviewed, and only those who agreed to participate in this study were interviewed. The consent form excluded the possibility of unjustified deception, undue influence, and intimidation. The agreement was signed only after prospective subjects were adequately informed. Their decision on whether to participate did affect the doctor-patient relationship or any other benefits to which they are entitled. Personal information about subjects will never be disclosed, and the data collected will remain confidential.
All women included in the study (n = 180) were interviewed according to the questionnaire designed and developed by our team and included yes/no questions, select from a list questions, and short answer questions in the Arabic language. All participants were stratified into 3 groups according to their smoking status: Group I, current active smokers; Group II, women with current exposure to secondhand smoke; and Group III, nonsmoking women, neither active nor passive.
The study variables included demographic information (maternal age, education, work status, environment status, smoking behavior and attitudes), current pregnancy history (parity, mode of delivery), and perinatal outcomes (birth weight, length, head and chest circumferences, Apgar score at 1 and 5 minutes, NICU admission).
Data for this study came from a face-to-face survey with pregnant women and laboratory reports. Data was entered into a unified computer database and analyzed by our team.
Statistical analysis was performed using the application package IBM SPSS 25 (SPSS: An IBM Company, New York, USA). The character of data distribution was evaluated with the W-criterion of Shapiro-Wilk. Various algorithms of statistical analysis were used depending on the type of distribution of feature. Absolute and relative indicators (%) were used to represent the qualitative characteristics. Quantitative data were presented by central tendency and dispersion: the mean value (M) with a 95% confidence interval (CI). A comparison of three independent groups on one or several signs, having at least one of the groups of distribution different from the normal or if the type of distribution was not analyzed, was carried out by checking the statistical hypotheses about the equality of middle rank using the Mann-Whitney U-test. Analysis of contingency tables (χ2) was used to assess the differences in relative values. Fisher exact test ( p ) was applied at frequencies less than 5. The threshold for statistical significance was set at p < 0.05.
Brief Characteristic of the Studied Group
All women who participated in the study were married. The average maternal age was 30.23 ± 1.47, 28.85 ± 1.50, and 31.1 ± 1.34 years among active smokers, passive smokers and nonsmokers, respectively. It was significantly lower in the group of passive women smokers compared with women who do not smoke ( p = 0.027). Active smokers were significantly less educated than passive smokers and nonsmokers ( p < 0.001), and most of them did not work compared to nonsmokers ( p = 0.037). Additionally, their family monthly income was significantly lower than in the group of passive smokers ( p = 0.006). Approximately 13% of active smoking women were nulliparous, while 43% of passive smokers were nulliparous women ( p < 0.001). Both active and passive smoking groups were less likely to follow up with perinatal care clinics compared to women who did not smoke ( p = 0.029). Brief characteristics of the studied groups are represented in Appendices Table 1.
Smoking Behavior and Attitudes in Studied Groups
Most smoking women (both active and passive) reported their husband is also a smoker and smokes at home (63% [50.68–74.38] and 80% [68.22–88.17], respectively), while only 2% (0.30–8.86) of nonsmoking women declared about a smoking husband ( p < 0.001).
We found that most women started to smoke at the age of 20, and more (43% [31.57–55.89], 33% [22.73–45.94]) classified themselves as moderate and heavy smokers. All of them smoke at home, and 20% (11.83–31.78) smoke at the workplace.
Our study revealed that many smoking women received information about the hazardous effects of smoking during perinatal visits (43% [31.57–55.89]), and 83% (71.96–90.68) were aware of the effects of smoking on perinatal outcomes. However, only 13% [6.91–24.16] of women quit smoking cigarettes before or in the early termination of pregnancy. Approximately 23% (14.44–35.43) decreased the frequency of smoking during their pregnancy, 13% (6.91–24.16) continued to smoke with the same frequency as before, and 23% (14.44–35.43) of women tried to stop their habit, but without success.
Pregnancy Outcomes
Active smoking women had significantly lower gestational age at delivery compared to passive smoking and nonsmoking women ( p = 0.038 and p = 0.003, respectively). There were no differences in the rate of cesarean section between study groups ( p > 0.05).
Neonates from active smoking mothers had significantly lower weight at birth compared to neonates from passive smoking and nonsmoking women ( p = 0.016 and p = 0.019, respectively) as well as significantly lower head and chest circumferences compared to babies from passive smoking mothers ( p < 0.001 and p = 0.036, respectively). Additionally, we found that neonates from active smoking women had a significantly lower 1-minute Apgar score compared to those from nonsmoking women ( p = 0.023), while there were no differences in the 5th-minute Apgar score. The rate of NICU admission did not differ among the 3 groups.
The main pregnancy outcomes are represented in Appendices Table 2.
Urine Cotinine Level in Mothers and Newborns
The urine cotinine level was significantly higher in both active and passive smoking women compared to nonsmoking women—43.27 ± 15.57 ng/mL, 1.08 ± 0.68 ng/mL, and 0.17 ± 0.05 ng/mL, respectively (both p < 0.01). Additionally, the urine cotinine level was significantly higher in newborns who had been exposed in utero (despite maternal active or passive smoking status)—42.53 ± 15.81 ng/mL, 0.42 ± 0.14 ng/mL, 0.15 ± 0.04 ng/mL for the active, passive, and nonsmoking group, respectively (both p < 0.001). A Pearson correlation test revealed there was a weak negative correlation between urine cotinine level and birth weight— r = –0.14 for maternal cotinine level and r = –0.15 for neonate cotinine level.
Smoking is considered one of the preventable risk factors for poor perinatal outcomes. Over the past several decades, many studies related to this issue have been conducted worldwide. Moreover, most of them have shown that smoking is a serious threat to public health, and this problem needs to be addressed at the level of the health care system [ 6 , 7 ].
This study, aimed at identifying the effects of smoking on perinatal outcomes, underscores the importance of smoking cessation interventions, especially in the context of high smoking rates among men and women of childbearing age [ 10 , 11 , 13 ].
Our study revealed that active smoking women were less educated and had less monthly family income compared to passive smoking and nonsmoking women. These findings replicate those of a similar study, where pregnant women with less than a high school education were more likely to smoke as compared to women with bachelor’s degree or higher [ 19 ].
The current study showed that active smoking women were likely to deliver an earlier gestational age compared to passive smoking and nonsmoking women. This finding is also suggested by several studies on the risk of preterm birth in smoking mothers [ 17 , 21 ]; however, we cannot equate these studies, since, in our study, the inclusion criterion was a full-term pregnancy.
According to Li and colleagues (2019), smoking women were more likely to have a caesarean section for nonreassuring fetal status [ 20 ]. However, in our study, there were no differences in the mode of delivery between women despite their smoking status.
We found a significantly lower 1-minute Apgar score in newborns from active smoking mothers compared to those from nonsmoking women, while the 5-minute Apgar score did not differ by maternal smoking status. The rate of NICU admission was similar between the study groups. Similar results were shown by Kharkova and colleagues (2017) [ 18 ], though another study found a significant increase in NICU admission in the smoking cohort [ 20 ]. Thus, we cannot accept smoking as the only factor that affects the Apgar score and the need for NICU admission [ 35 ].
Our study revealed the birth weights of newborns from active smoking mothers were significantly less than those from passive smoking and nonsmoking women, and secondhand exposure did not influence birth weight. Additionally, we found a tendency for smaller length and head and chest circumferences in newborns from active smoking women compared to those from nonsmoking mothers. These findings are strongly supported by similar studies, where maternal active smoking was associated with a lower mean birth weight [ 14 , 25 ], smaller length and head circumference [ 1 , 2 , 33 ], and abnormal body proportions [ 18 , 30 ].
We found that urinary cotinine levels in women and newborns were negatively associated with birth weight. However, this association was very weak. The only study that assessed the relationship between cotinine levels and anthropometric data in newborns found that the level of urine cotinine in newborns had a strong negative association with birth weight [ 8 ].
Furthermore, there is a high rate of smoking among Jordanian pregnant women, approximately 18% [ 9 ], and many were exposed to tobacco smoking by their husbands [ 9 ]. The smoking rate in Jordan is among the highest rates in the world, with a high exposure of smoking indoors and a lack of policy restrictions in developing countries [ 9 , 11 , 12 , 13 , 31 ]. This is a leading factor for stillbirths and sudden deaths among infants, which need more intervention programs [ 9 , 11 , 13 , 29 ]. Moreover, comprehensive smoke-free policies must be ensured in developing countries. In particular, for some health outcomes that are strongly influenced by active and passive smoking, keep in mind that smoking is a common habit and rates are higher among developing country communities [ 9 , 11 , 13 , 26 ].
Strengths and Limitations
The small sample size and the fact that the study sample excluded high-risk pregnant women and women with preterm birth means our results cannot be extrapolated to all pregnant women in general or other regions of Jordan. These groups of women require additional study.
We discussed limited perinatal outcomes as per our exclusion criteria and did not take into account pregnancy complications such as preterm delivery, gestational diabetes mellitus, gestational hypertension, placental abruption, and fetal malformations, which can be caused by smoking. So, further studies should be directed to these issues too.
Our information on smoking behavior during pregnancy was based on self-reporting. Consequently, underreporting of maternal smoking may have occurred, resulting in potential for misclassification. However, to address this, we measured cotinine in mother-baby couples to assess the accuracy of self-reported data. This was a major strength of our study.
Take into consideration the necessary contribution to the literature on prenatal smoke exposure and risks on obstetric outcomes for women and children in Jordan. The main strength is the inclusion of laparotomy cotinine levels. Furthermore, this study was the first of its kind in Jordan and therefore provides necessary baseline information for further improvement on tobacco control research in the developing countries and the Middle Eastern regions.
Conclusions and Recommendations
The current study illustrated that smoking during pregnancy leads to offspring with reduced birth weight, birth length, and head and chest circumference as well as reduced delivery gestational age and lower Apgar score. Moreover, we may conclude that argileh smoking (e.g., hookah) during pregnancy can also contribute to a reduction in newborns’ anthropometric measurements.
Our study findings highlight the need for continued study of the effects of smoking on perinatal outcomes and the need for targeted interventions to reduce and prevent tobacco smoking and tobacco smoke exposure during pregnancy. Further studies are needed to increase awareness of these adverse effects, to develop cessation interventions in the preconception period, and to evaluate useful interventions to enhance a smoking-free environment during pregnancy.
Additional File
The additional file for this article can be found as follows:
Appendix Table 1 and Appendix Table 2. DOI: https://doi.org/10.5334/aogh.3384.s1
Acknowledgements
The authors acknowledge the Jordan University of Science and Technology support for this research. Thanks to all the mothers participating in this study. Thanks and appreciation to King Abdullah University Hospital.
Funding Information
This study was partially funded by the Jordan University of Science and Technology Scientific Research Fund.
Competing Interests
The authors have no competing interests to declare.
Author Information
Dr. Shereen Hamadneh:
- Associate professor, Department of Maternal and Child Health, Princess Salma College of Nursing, Al-Bayt University, Mafraq, Jordan
- A member of the Union of Jordanian Universities for Smoking and Tobacco Control
- A scientific member of the executive council of the Center of Excellence for Scientific Research at the Scientific Society of Arab Nursing Faculties
- A clinical nutritionist, a member of the Jordanian Diet Association
- The chairman of the board of directors of the Happy Baby—Child and Family Association, Ministry of Social Development, Jordan
- Director of Safe Child Care Program, Australian Aid Program, Jordan
- A member of the board of directors of the Community Women Support Committee, Ramtha, Jordan
Adibelli D, Kirca N. The relationship between gestational active and passive smoking and early postpartum complications. Journal of Maternal-Fetal & Neonatal Medicine . 2020; 33(14): 2473–2479. DOI: https://doi.org/10.1080/14767058.2020.1763294
Al-Sheyab NA, Al-Fuqha RA, Kheirallah KA, Khabour OF, Alzoubi KH. Anthropometric measurements of newborns of women who smoke waterpipe during pregnancy: a comparative retrospective design. Inhalation Toxicology . 2016; 28(13): 629–635. DOI: https://doi.org/10.1080/08958378.2016.1244227
Anderson TM, Lavista Ferres JM, Ren SY, et al. Maternal smoking before and during pregnancy and the risk of sudden unexpected infant death. Pediatrics . 2019; 143(4): e20183325. DOI: https://doi.org/10.1542/peds.2018-3325
Apgar V. A proposal for a new method of evaluation of the newborn infant. Current Researches in Anesthesia & Analgesia . 1953; 32(4): 260–267. DOI: https://doi.org/10.1213/00000539-195301000-00041
Bjørnholt SM, Leite M, Albieri V, Kjaer SK, Jensen A. Maternal smoking during pregnancy and risk of stillbirth: results from a nationwide Danish register-based cohort study. Acta Obstetricia et Gynecologica Scandinavica . 2016; 95(11): 1305–1312. DOI: https://doi.org/10.1111/aogs.13011
Chen MM, Chiu CH, Yuan CP, Liao YC, Guo SE. Influence of environmental tobacco smoke and air pollution on fetal growth: a prospective study. International Journal of Environmental Research and Public Health . 2020; 17(15): 5319. DOI: https://doi.org/10.3390/ijerph17155319
Committee opinion no. 721 summary: smoking cessation during pregnancy. Obstetrics and Gynecology . 2017; 130(4): 1. DOI: https://doi.org/10.1097/AOG.0000000000002348
Ein-Mor E, Berman T, Barnett-Itzhaki Z, et al. Newborn infant urinary cotinine and birth outcomes in the Jerusalem Environment Mother and Child Cohort Study. International Journal of Hygiene and Environmental Health . 2019; 222(7): 1054–1058. DOI: https://doi.org/10.1016/j.ijheh.2019.07.006
Hamadneh J, Hamadneh S, Amarin Z, Al-Beitawi S. Knowledge, attitude and smoking patterns among pregnant women: a Jordanian perspective. Ann Glob Health . 2021; 87(1): 36. DOI: https://doi.org/10.5334/aogh.3279
Hamadneh S. The Impact of Implementing a Sudden Infant Death Syndrome Education Package in Jordan . Doctoral thesis. WA, Australia: Edith Cowan University; 2014. https://ro.ecu.edu.au/theses/1086
Hamadneh S, Al-Shdayfat N, Al Omari, O, et al. Sudden infant death syndrome in the Middle East: an exploration of the literature on rates, risk factors, high-risk groups and intervention programs. Research Journal of Medical Sciences . 2016a; 10(4): 199–204. DOI: https://doi.org/10.3923/rjmsci.2016.199.204
Hamadneh S, Kassab M, Eaton A, Wilkinson A, Creedy DK. Sudden unexpected infant death. In: Laher I, ed. Handbook of Healthcare in the Arab World . Cham: Springer; 2020. DOI: https://doi.org/10.1007/978-3-319-74365-3_144-1
Hamadneh S, Kassab M, Hamadneh J, Amarin A. Sudden unexpected infant death in Jordan and home environments. Pediat Inter . 2016b; 58(12): 1333–1336. DOI: https://doi.org/10.1111/ped.13016
Huang SH, Weng KP, Huang SM, et al. The effects of maternal smoking exposure during pregnancy on postnatal outcomes: a cross-sectional study. JCMA . 2017; 80(12): 796–802. DOI: https://doi.org/10.1016/j.jcma.2017.01.007
Inoue S, Naruse H, Yorifuji T, et al. Impact of maternal and paternal smoking on birth outcomes. Journal of Public Health (England, Oxford) . 2017; 39(3): 1–10. DOI: https://doi.org/10.1093/pubmed/fdw050
Källén K. Maternal smoking during pregnancy and infant head circumference at birth. Early Human Development . 2000; 58(3): 197–204. DOI: https://doi.org/10.1016/S0378-3782(00)00077-3
Kelkay B, Omer A, Teferi Y, Moges Y. Factors associated with singleton preterm birth in Shire Suhul General Hospital, Northern Ethiopia, 2018. Journal of Pregnancy . 2019; 4629101. DOI: https://doi.org/10.1155/2019/4629101
Kharkova OA, Grjibovski AM, Krettek A, Nieboer E, Odland JØ. Effect of smoking behavior before and during pregnancy on selected birth outcomes among singleton full-term pregnancy: a Murmansk County birth registry study. International Journal of Environmental Research and Public Health . 2017; 14(8): 867. DOI: https://doi.org/10.3390/ijerph14080867
Kondracki AJ. Prevalence and patterns of cigarette smoking before and during early and late pregnancy according to maternal characteristics: the first national data based on the 2003 birth certificate revision, United States, 2016. Reproductive Health . 2019; 16(1): 142. DOI: https://doi.org/10.1186/s12978-019-0807-5
Li R, Lodge J, Flatley C, Kumar S. The burden of adverse obstetric and perinatal outcomes from maternal smoking in an Australian cohort. Australian & New Zealand Journal of Obstetrics & Gynaecology . 2019; 59(3): 356–361. DOI: https://doi.org/10.1111/ajo.12849
Liu B, Xu G, Sun Y, et al. Maternal cigarette smoking before and during pregnancy and the risk of preterm birth: a dose-response analysis of 25 million mother-infant pairs. PLoS Medicine . 2020; 17(8): e1003158. DOI: https://doi.org/10.1371/journal.pmed.1003158
Madureira J, Camelo A, Silva AI, et al. The importance of socioeconomic position in smoking, cessation and environmental tobacco smoke exposure during pregnancy. Scientific Reports . 2020; 10(1): 15584. DOI: https://doi.org/10.1038/s41598-020-72298-8
Mahmoodabad S, Karimiankakolaki Z, Kazemi A, Mohammadi NK, Fallahzadeh H. Exposure to secondhand smoke in Iranian pregnant women at home and the related factors. Tobacco Prevention & Cessation . 2019; 5(February): 7. DOI: https://doi.org/10.18332/tpc/104435
McInerney C, Ibiebele I, Ford JB, et al. Benefits of not smoking during pregnancy for Australian Aboriginal and Torres Strait Islander women and their babies: a retrospective cohort study using linked data. BMJ Open . 2019; 9(11): e032763. DOI: https://doi.org/10.1136/bmjopen-2019-032763
Moore BF, Starling AP, Magzamen S, et al. Fetal exposure to maternal active and secondhand smoking with offspring early-life growth in the Healthy Start study. Int J Obes . 2019; 43(4): 652–662. DOI: https://doi.org/10.1038/s41366-018-0238-3
Nazar G, Tayu Lee J, Arora M, Millett C. Socioeconomic inequalities in secondhand smoke exposure at home and at work in 15 low- and middle-income countries. Nicotine Tob Res . 2016; 18(5): 1230–1239. DOI: https://doi.org/10.1093/ntr/ntv261
Philips, EM, Santos S, Trasande L, et al. Changes in parental smoking during pregnancy and risks of adverse birth outcomes and childhood overweight in Europe and North America: an individual participant data meta-analysis of 229,000 singleton births. PLoS Medicine . 2020; 17(8): e1003182. DOI: https://doi.org/10.1371/journal.pmed.1003182
Qu Y, Chen S, Pan H, et al. Exposure to tobacco smoke and stillbirth: a national prospective cohort study in rural China. Journal of Epidemiology and Community Health . 2020; 74(4): 315–320. DOI: https://doi.org/10.1136/jech-2019-213290
Reece S, Morgan C, Parascandola M, Siddiqi K. Secondhand smoke exposure during pregnancy: a cross-sectional analysis of data from Demographic and Health Survey from 30 low-income and middle-income countries. Tobacco Control . 2019; 28(4): 420–426. DOI: https://doi.org/10.1136/tobaccocontrol-2018-054288
Rumrich I, Vähäkangas K, Viluksela M, Gissler M, de Ruyter H, Hänninen O. Effects of maternal smoking on body size and proportions at birth: a register-based cohort study of 1.4 million births. BMJ Open . 2020; 10(2): e033465. DOI: https://doi.org/10.1136/bmjopen-2019-033465
Safi M, al-Tahat J. Jordan smoking rates highest in world amid claims of big tobacco interference. The Guardian . July 20, 2020. Accessed December 9, 2020. https://www.theguardian.com/world/2020/jun/23/jordan-smoking-rates-highest-in-world-amid-claims-of-big-tobacco-interference .
Snodgrass AM, Tan PT, Soh SE, et al. Tobacco smoke exposure and respiratory morbidity in young children. Tobacco Control . 2016; 25(e2): e75–e82. DOI: https://doi.org/10.1136/tobaccocontrol-2015-052383
Sturrock S, Williams E, Ambulkar H, Dassios T, Greenough A. Maternal smoking and cannabis use during pregnancy and infant outcomes. Journal of Perinatal Medicine . 2020; 48(2): 168–172. DOI: https://doi.org/10.1515/jpm-2019-0422
Tong VT, England LJ, Rockhill KM, D’Angelo DV. Risks of preterm delivery and small for gestational age infants: effects of nondaily and low-intensity daily smoking during pregnancy. Paediatric and Perinatal Epidemiology . 2017; 31(2): 144–148. DOI: https://doi.org/10.1111/ppe.12343
Walfisch A, Nikolovski S, Talevska B, Hallak M. Fetal growth restriction and maternal smoking in the Macedonian Roma population: a causality dilemma. Archives of Gynecology and Obstetrics . 2013; 287(6): 1131–1136. DOI: https://doi.org/10.1007/s00404-013-2731-1 .
- Apply now Enquire now
Study at UniSA
Accounting and Finance Business Management and HRM Marketing Property Tourism, Sports and Events
Architecture Communication Contemporary Art Creative Industries Design Film, Television and Visual Effects Journalism Urban and Regional Planning
Biomedical Science Health Science Human Movement, Sport, Clinical Exercise Physiology Laboratory Medicine Medical Radiation Science Nursing or Midwifery Nutrition and Food Sciences Occupational Therapy Pharmacy Physiotherapy Podiatry Speech Pathology
- Law & Criminal Justice
Aboriginal and Australian Studies Arts Human Services Languages Psychology Social Work
Aviation Construction Management Defence Engineering Environmental Science Information Technology Mathematics Science
- Teaching & Education
- What can I study
- Entry pathways
- Find an education agent
- Sponsorships
- Living in Adelaide
- UniSA Online
- Scholarships
- Postgraduate study
- Research degrees
- Executive Education
- Regional students
- Aboriginal students
- Discover our research Partner with us
- Research projects
- How to apply
- Check your eligibility
- Research scholarships
- Concentrations
- Cooperative Research Centres
- Research studies & volunteers
- Research experts
- Industry & collaboration
- Mawson Lakes
- Mount Gambier
- Psychology Clinic
- Health Clinics
- Disability Hub
- Wirringka Student Services
- Museums and galleries
- Industry & partnerships
- Enterprise Hub
About UniSA
- Our history
- Vice Chancellor
- Achievements
- Mission, values, vision
- Strategic action plan
- Staff directory
- UniSA Allied Health & Human Performance
- UniSA Business
- UniSA Clinical & Health Sciences
- UniSA Creative
- UniSA Education Futures
- UniSA Justice & Society
- Working at UniSA
- Publications
- Aboriginal engagement
- Login Staff Students Alumni
- Media centre
Prenatal smoking risks academic achievement of unborn babies
28 August 2024
In a systematic review of 19 studies and 1.25 million participants, researchers at the University of South Australia along with a team at Curtin University , SAHMRI , Harvard University and others* found that 79% of studies reported reduced academic achievement in children exposed to maternal prenatal smoking.
An additional meta-analysis of eight primary studies with 723,877 participants showed that children exposed to maternal prenatal tobacco smoking were 49% more likely to struggle with poor academic achievement in comparison to those who had not been exposed to smoking in utero.
In Australia , 8.7% (or 26,433) of all mothers who gave birth in 2021 smoked at some time during their pregnancy .
Lead researcher, UniSA’s Dr Bereket Duko , says that despite what is already known about smoking, research is still uncovering additional negative effects.
“For decades, agencies across the globe have pushed anti-smoking campaigns about the dangers of smoking. But despite these efforts, tobacco smoking remains a pervasive global public health issue,” Dr Bereket says.
“Prenatal smoking is known to cause multiple pregnancy complications, including a higher risk of miscarriage, stillbirth, restricted growth and development, and serious birth defects. It is also linked with adverse mental health outcomes and behavioural issues.
“Our new research adds to this, by showing that maternal prenatal smoking has a significant risk of limiting a child’s academic performance, putting them well behind their peers at school.
“We all want children to have the best start in life. But clearly, we must do better to educate mothers and families about the noxious effects of smoking while pregnant on mother and baby.
“Remember, the fight against smoking is not one we have already won. Yes, we have made big steps to reduce the number of people smoking, and we have made many aware of the health risks. But this is an ongoing battle, and we must continue to educate people about the dangers of tobacco so that the next generations do not unnecessarily suffer.”
Notes or editors:
*Additional partners include the University of Iceland, the University of Queensland, the Norwegian Institute of Public Health, and the University of Sydney.
…………………………………………………………………………………………………………………………
Contact for interview: Dr Bereket Duko M: +61 410 350 140 E: [email protected] Media contact: Annabel Mansfield M: +61 479 182 489 E: [email protected]
Other articles you may be interested in
It only takes 15 minutes to change your health
Say ‘aah’ and get a diagnosis on the spot: is this the future of health?
Swipe up! Health apps deliver real results en masse
Australian students
Phone: +61 8 8302 2376 Enquiry: unisa.edu.au/enquiry
International students
Phone: +61 8 9627 4854 Enquiry: unisa.edu.au/enquiry

- Research article
- Open access
- Published: 26 March 2021
Health outcomes of smoking during pregnancy and the postpartum period: an umbrella review
- Tuba Saygın Avşar ORCID: orcid.org/0000-0002-4143-3852 1 ,
- Hugh McLeod 2 , 3 &
- Louise Jackson 1
BMC Pregnancy and Childbirth volume 21 , Article number: 254 ( 2021 ) Cite this article
18k Accesses
63 Citations
23 Altmetric
Metrics details
Smoking during pregnancy (SDP) and the postpartum period has serious health outcomes for the mother and infant. Although some systematic reviews have shown the impact of maternal SDP on particular conditions, a systematic review examining the overall health outcomes has not been published. Hence, this paper aimed to conduct an umbrella review on this issue.
A systematic review of systematic reviews (umbrella review) was conducted according to a protocol submitted to PROSPERO ( CRD42018086350 ). CINAHL, EMBASE, MEDLINE, PsycINFO, Web of Science, CRD Database and HMIC databases were searched to include all studies published in English by 31 December 2017, except those focusing exclusively on low-income countries. Two researchers conducted the study selection and quality assessment independently.
The review included 64 studies analysing the relationship between maternal SDP and 46 health conditions. The highest increase in risks was found for sudden infant death syndrome, asthma, stillbirth, low birth weight and obesity amongst infants. The impact of SDP was associated with the number of cigarettes consumed. According to the causal link analysis, five mother-related and ten infant-related conditions had a causal link with SDP. In addition, some studies reported protective impacts of SDP on pre-eclampsia, hyperemesis gravidarum and skin defects on infants. The review identified important gaps in the literature regarding the dose-response association, exposure window, postnatal smoking.
Conclusions
The review shows that maternal SDP is not only associated with short-term health conditions (e.g. preterm birth, oral clefts) but also some which can have life-long detrimental impacts (e.g. obesity, intellectual impairment).
Implications
This umbrella review provides a comprehensive analysis of the overall health impacts of SDP. The study findings indicate that while estimating health and cost outcomes of SDP, long-term health impacts should be considered as well as short-term effects since studies not including the long-term outcomes would underestimate the magnitude of the issue. Also, interventions for pregnant women who smoke should consider the impact of reducing smoking due to health benefits on mothers and infants, and not solely cessation.
Peer Review reports
Smoking during pregnancy (SDP) is a significant public health concern due to adverse health outcomes on mothers and infants, such as miscarriage, low birth weight (LBW), preterm birth, and asthma [ 1 , 2 , 3 , 4 ]. The prevalence of SDP is around 10% in high-income countries (HICs) [ 5 , 6 , 7 ] and 3% in low- and middle-income countries (LMICs) [ 8 ].
Smoking during pregnancy generates a considerable cost burden and the annual cost of smoking-related pregnancy complications has been estimated to be between £8 and £64 million in the UK, depending on the estimation method chosen [ 9 ]. In addition, the costs associated with the health problems experienced by the infant during the first year following the birth were found to be between £12 and £23 million [ 9 ]. Smoking during pregnancy poses a considerable economic burden in the USA as well, since smoking-attributable neo-natal costs were estimated to be nearly $228 million in total [ 10 ]. When long-term impacts on the infant are considered, the actual figures are likely to be higher. Therefore, to have a comprehensive estimate of the health and cost impacts of SDP to inform policy decisions and ensure that scarce health resources are allocated optimally, it is necessary to review the evidence on the overall health effects for mothers and infants over the longer term.
A scoping review and a review of reviews by Godfrey and colleagues [ 9 ], and a scoping review by Jones [ 11 ] provided a picture of the health and cost outcomes associated with SDP, and several narrative reviews about the health outcomes have been published [ 12 , 13 , 14 , 15 ]. However, none of these papers were fully systematic and comprehensive. Moreover, a considerable number of systematic reviews have been published more recently on the impact of maternal SDP on separate health outcomes, which makes this overall review of the current evidence timely.
The present study aimed to investigate the overall health impacts of maternal smoking during pregnancy and the postpartum period on mothers and infants. Additionally, the evidence on the impact of the number cigarettes consumed and second-hand smoking (SHS) by partner during pregnancy was assessed [ 16 , 17 ].
The guideline provided by the Cochrane Handbook for Systematic Reviews of Interventions [ 18 ] was followed. The review was carried out according to a protocol which included a detailed description of the methodology [ 19 ]. Umbrella reviews have been increasingly used to summarise the existing evidence on an issue by analysing all systematic reviews conducted [ 18 , 20 ]. Considering the large number of original studies about health outcomes of SDP, an umbrella review was the appropriate design for this research.
Searches were undertaken of CINAHL, EMBASE, MEDLINE, PsycINFO, Web of Science, CRD Database (includes DARE, NHSEED and HTA) and HMIC databases. The search strategy for MEDLINE is presented in the Additional file 1 . All systematic reviews published in English and by December 2017. Two independent reviewers conducted the study selection and quality assessment. The data extraction toll is provided in the Additional file 1 : Table S1. The quality of included studies was assessed with a tool developed from the Centre for Reviews and Dissemination (CRD) checklist, which covers a range of issues including prior protocol use, bias in study selection, and consideration of publication bias and inclusion of a quality assessment [ 21 ]. Main outcome measures were odds ratios and relative risks for smoking women and their children compared to non-smoking women and their children.
To evaluate the causal link between SDP and the identified conditions which were found to have an association with SDP, a causal link analysis was conducted using established methods [ 11 ]. The evidence on the identified conditions was assessed and categorised using the following criteria:
Strong evidence - one systematic review with ≥8 studies (group 1) or more than one systematic review (group 2);
Weak evidence – more than one systematic review reported conflicting findings (group 3) or one systematic review reported limited number of studies (< 8) which found a relationship (group 4).
A validity assessment was conducted by reducing the threshold of eight studies to seven, and increasing it to 10 and 12. As discussed by Jones [ 11 ], this strength of evidence analysis fulfilled five of the nine items proposed by Hill [ 22 ] as conditions of a causal link (strength, consistency, specificity, temporality, and plausibility). In addition, the dose-response association was also considered. The remaining requirements (coherence, experiment, and analogy) of the Hill [ 22 ] criteria were irrelevant to this review as laboratory studies were not included and no causes other than smoking of the identified conditions were considered.
The database search yielded 744 studies and an additional five studies were found through hand searching the references of included studies. Following the removal of duplicates and abstract screening, 64 studies were selected for full-text analysis Fig. 1 .
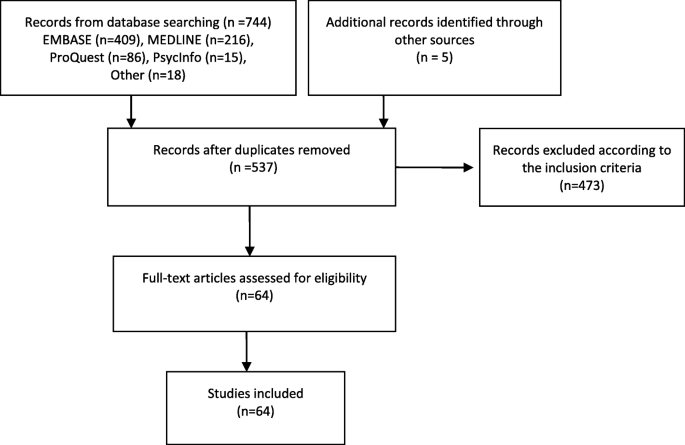
PRISMA Diagram for Study Selection
Characteristics of the included studies
Most reviews ( n = 46) were published since 2010. Only 13 reviews investigated a health condition related to mothers; the other 49 reviews analysed infant-related conditions, except two [ 23 , 24 ] which evaluated the impacts on both. Key characteristics of the included reviews are provided in Additional file 1 : Table S2.
In most reviews ( n = 27 reviews), the included studies were predominantly from HICs, and 22 of the included reviews covered studies from HIC only. In two reviews [ 3 , 25 ] most of the included studies were concerned with upper-middle-income countries.
In 12 reviews, the country of focus of the included studies was not provided. However, one of them [ 26 ] conducted a meta-analysis of the studies from Europe only, and in five reviews, the language of the included studies was either only English [ 27 , 28 , 29 ] or languages [ 30 , 31 ] which are only spoken by HICs. In the remaining six reviews [ 32 , 33 , 34 , 35 , 36 ] there was no indication of whether the studies focussed on LMICs or HICs. Nevertheless, when interpreting the results of these reviews, the possibility that studies which were conducted in LMICs have been included in addition to HICs should be born in mind.
Quality of the included studies
The quality scores of the reviews are provided in Additional file 1 : Table S2. The highest achievable score was 16, and most reviews ( n = 46) scored between nine and 14 while two reviews [ 25 , 32 ] achieved very low scores of 4 and 5. Therefore, most of the included reviews were moderate or high quality studies according to the criteria used.
Study selection was made by two reviewers independently in almost half of the reviews ( n = 31) to minimise bias. The majority of the studies ( n = 50) assessed publication bias. Heterogeneity was measured in all reviews although causes of heterogeneity were not analysed in some ( n = 17). However, only seven reviews reported protocol publication [ 3 , 26 , 33 , 37 , 38 , 39 , 40 ].
Impacts of smoking during pregnancy on mothers
Overall, of the 14 reviews that reported the impact of smoking on mothers, all except two [ 41 , 42 ] conducted meta-analyses (Additional file 1 : Table S3). The reviews presented consistent findings, suggesting a significantly increased risk associated with smoking and seven health conditions. The highest risks were reported for spontaneous miscarriage in assisted reproduction (OR = 2.65, 95% CI, 1.33–5.30, 28) and ectopic pregnancy (OR = 2.30, 95% CI, 2.02–2.80, 30). Two conditions (preeclampsia and hypremesis gravidarum) were found to be negatively associated with SDP. Hence, women who smoked whilst pregnant were less likely to experience these two conditions.
Impacts of smoking during pregnancy on infants
Studies found a smoking-related increased risk for 20 conditions and the highest impact was observed for sudden infant death syndrome (SIDS) (OR = 2.98, 95% CI, 2.51–3.54) [ 24 ], asthma (OR = 1.85, 95% CI, 1.35–2.53) [ 1 ], LBW (OR = 1.75, 95% CI, 1.42–2.10), stillbirth (OR = 1.55, 95% CI, 1.36–1.78) [ 38 ] and obesity (OR = 1.60, 95% CI 1.37–1.88) [ 43 ]. Studies did not find any significant association between 15 conditions and SDP, including autism, brain tumors, breast cancer in daughters and testicular cancer in sons. On the other hand, a protective impact on skin defects was observed in one review [ 44 ].
Most studies ( n = 42) investigating the impacts of SDP on infants conducted a meta-analysis (Additional file 1 : Table S4), and only nine did not include this (Additional file 1 : Table S5). In these studies, there was no significant relationship between maternal SDP and lung functions, or Tourette’s syndrome.
The age group of study participants varied between studies; for example, some conditions were assessed amongst infants while some were measured in adults. In some reviews, participants were both infants and adults. Table 1 lists health conditions by the life stage they were assessed.
The reviews included in this study indicated that maternal smoking increased the risk of death for the child during the prenatal period, neonatal period and infancy. The evidence showed maternal SDP did not only have short-term impact but also some long-term outcomes which could be detrimental for offspring. Moreover, some of the conditions measured in early life stages could continue later in life. For instance, some birth defects and intellectual disability would affect later stages of life.
Dose-response association
To understand the impact of reductions in smoking, the relationship between the number of cigarettes consumed and the health implications for infants or mothers were investigated. Although a dose-response impact of SDP was reported in 27 reviews (22 related to infant conditions), it was statistically tested in just 17 studies. Among them, four found no significant impact of SDP and their dose-response tests showed similar results. In addition, one review [ 62 ] reported a dose-response association for SIDS but did not provide the odds ratios. Findings of the remaining 12 studies are summarised in the Additional file 1 : Table S6.
To define light, moderate and heavy smokers, most studies [ 38 , 39 , 46 , 62 , 63 , 64 ] chose smoking 10 cigarettes daily as a cut-off point to distinguish light smokers from moderate and heavy smokers. In some studies [ 4 , 39 , 46 , 61 , 64 ], both 10 cigarettes daily and 20 cigarettes daily were utilised as the thresholds. In one review the number of cigarettes consumed daily for each category was inconsistent [ 65 ]. All studies estimated the risk ratios compared to non-smokers [ 66 ], except for one review, in which light smokers were compared to moderate smokers.
Included reviews showed that the risk of stillbirth, birth defects, preterm birth and perinatal death elevated as the number of cigarettes increased [ 4 , 38 , 39 , 46 ]. In contrast, smoking not only protected against pre-eclampsia but the risk reduced as exposure increased [ 67 ].
A dose-response relationship was found in five reviews although a pooled estimation was not calculated. They reported an increased risk for placental abruption [ 68 ], and for the offspring the risk of being overweight [ 57 ], having oral clefts [ 29 , 50 ], or a decrease in cognitive abilities [ 53 ] increased along with the number of cigarettes that the mothers consumed. Five reviews included studies reporting a dose-response relationship along with others that did not find any relationship [ 1 , 41 , 51 , 56 , 69 ]. Therefore, it was not clear whether or not the risk for some conditions (pre-eclampsia, and in the offspring asthma, attention deficit hyperactivity disorder, and vision difficulties) was affected by the number of cigarettes consumed.
Six reviews observed no significant association between the number of cigarettes consumed and the risk of health conditions for the children exposed to maternal SDP, although overall they reported a significantly increased risk. These studies covered congenital heart diseases [ 65 ], central nervous system tumors [ 64 ], childhood neuroblastoma [ 63 ], lower respiratory infections (LRI) [ 37 ] and lymphoblastic leukaemia [ 66 ], and reduced menarche age in daughters [ 61 ].
Impacts of postnatal maternal smoking on infants
The main findings of the reviews which investigated the impact of postnatal smoking on the infants are shown in Additional file 1 : Table S7. The reviews showed an increased impact on asthma, LRI, SIDS and wheezing but not on leukaemia and obesity. However, in some studies, it was not clear whether or not the mothers included in the studies smoked during the whole pregnancy as well as the postpartum period. This is a significant consideration as one study reported by Oken et al. [ 57 ] found no increase in the prevalence of obesity when the mother smoked only after birth, whereas smoking before and throughout pregnancy were found to be related with an increased risk [ 70 ].
Impact of second-hand smoking by partners
In addition to active smoking, SHS during pregnancy could have health implications. It was important to understand whether the health-related risks were higher when partners smoked during pregnancy. Therefore, partner-related findings of the included reviews were analysed. Partner smoking was considered in only 12 reviews of which six did not assess the impact of SHS specific to the pregnancy period (Additional file 1 : Table S8). None of the studies reported the combined impact of SDP and SHS by the partner during pregnancy. Two reviews reported an increased risk of SIDS [ 71 ] and delay in mental development [ 25 ] when the partners of non-smoking women smoked during pregnancy, while no association was found for brain tumors [ 72 ] or breast cancer risk in daughters [ 73 ].
Sub-group analyses in the included reviews
The reviews conducted sub-group analyses to assess the impact of study design, sample size, the duration of the infant exposure to smoking (i.e. pre-pregnancy, first trimester or the whole pregnancy) and adjustments for confounding factors. The study findings did not differ significantly in most of the analyses except for adjustments for confounding and study quality. The evidence was not sufficient to make a comparison based on country income groups because most studies were from high-income countries.
Although the included meta-analyses utilised the most adjusted estimations of observational studies when pooling their results, only 10 of the included reviews provided risk ratios for adjusted and unadjusted estimations (Additional file 1 : Table S9). Studies with unadjusted ratios estimated greater values for miscarriage, perinatal death, SDIS, overweight and obesity.
Sub-group analyses based on quality appraisal of the included studies were conducted in 14 reviews (Additional file 1 : Table S10). The results showed that high-quality studies reported higher ratios for some conditions (overweight, obesity, placenta previa) as opposed to lower or insignificant ratios for some others (e.g. LBW, miscarriage, stillbirth).
Two reviews [ 46 , 74 ] compared the type of smoking status data and found similar results for biochemical and self-reported data. The exposure period was researched in five reviews [ 40 , 41 , 46 , 64 , 75 ], and the results showed no significant difference between women who quit early in pregnancy and those who did not smoke.
Causal link analysis
The causal link analysis identified a range of health conditions found to have strong association with SDP; these are presented in Table 2 , grouped according to the strength of evidence.
Nearly all of the conditions for which a strong association was identified fulfilled the criteria for a causal link. The health conditions were largely reported by moderate- or high-quality reviews and there were consistent findings in the sub-group analyses. There was not a sufficient biological explanation to the correlation found between hyperemesis gravidarum and SDP, hence although there was a strong association, a causal link could not be confirmed.
This study analysed the health impacts of smoking during pregnancy and during the postpartum period on mothers and infants. The 64 included reviews covered 1744 studies relating to SDP or smoking during the postpartum period. The review found that maternal SDP has short-term and long-term health consequences, suggesting a positive association between 20 infant-related and seven mother-related conditions, and a negative association with two maternal conditions. The review did not find a statistically significant impact of SDP on 15 infant-related conditions while conflicting findings were reported for leukaemia and lymphoma.
The causal link analysis of the conditions that were found to have an association with SDP suggested that five mother-related and 10 infant-related conditions had a causal link with SDP. PPROM and intellectual disability in children did not fulfil the criteria for the casual link although meta-analyses reported a statistically significant relationship with SDP.
Health conditions with conflicting results
Some health conditions were assessed in multiple meta-analyses and they reported conflicting results. For instance, the increased risk of having any type of birth defect was statistically significant despite being small in the effect size (OR = 1.18, 95% CI, 1.14–1.22) in one review [ 39 ] as opposed to a borderline ratio (OR = 1.01, 95% CI, 0.96–1.07) reported in another [ 44 ]. The main difference was the reduced risk of skin defects (OR = 0.82, 95% CI, 0.75–0.89) which was included in the latter [ 44 ] while omitted in the former [ 39 ] without any justification. All five studies included in this meta-analysis reported a negative relationship and the heterogeneity was low ( P = 0.00001, I 2 = 0%). Therefore, the evidence suggested an increased risk of birth defects except for skin defects amongst SDP exposed children. However, there was no biological explanation for the potential protective impact of SDP on skin defects.
Another health condition with mixed findings was leukaemia. One meta-analysis [ 64 ] including 19 studies indicated an insignificant decreased risk (OR = 0.99, 95% CI, 0.92–1.06) whereas another review [ 66 ] of 21 studies found an increased risk (OR = 1.10, 95% CI, 1.02–1.19). The difference could be explained by the different studies included, since there were only five studies common to both, and the association between SDP and leukaemia is unclear.
Similarly, the reviews reported different results for lymphoma. One meta-analysis [ 55 ] found an insignificant association between any lymphoma and SDP based on eight studies (OR = 1.10, 95% CI, 0.96–1.27), although positive relationship for non-Hodgkin lymphoma was reported (OR = 1.22, 95% CI, 1.03–1.45, n = 8). Another review [ 64 ] which included six studies found an increased risk for any lymphoma (OR = 1.21, 95% CI, 1.05–1.34). Hence, SDP increases the risk of non-Hodgkin lymphoma but for other types of lymphoma the impact is unclear.
Strengths and limitations of the umbrella review
To the best of the authors’ knowledge, this is the first umbrella review on the topic and provides the most systematic and comprehensive assessment of the current evidence. The criteria to assess any causal links are an important consideration. The tool developed by Hill [ 22 ] is widely recognised for assessing causation. In addition to these criteria, this study considered the quality of reviews and the findings of sub-group analyses. Hence, the conditions identified by the causal link analysis are very likely to have a causal link with SDP.
The review has some limitations. Firstly, although systematic reviews are accepted as the highest in the evidence hierarchy [ 76 , 77 ], the focus on systematic reviews alone meant some health conditions were not covered. Some original studies have indicated the impact of SDP on other infant-related conditions, such as diabetes [ 78 ], hypomania [ 79 ], otitis [ 80 ] and pervasive development disorder [ 81 ], which were not assessed in a systematic review, and as a result were not included in this study. Furthermore, SDP has been shown to be related to the smoking uptake of the offspring [ 82 , 83 ]. There are also some maternal health conditions found to be related to smoking whilst pregnant in one study; vein thrombosis, myocardial infarction, influenza or pneumonia, bronchitis, gastrointestinal ulcers [ 84 ]. However, the current study focused on the conditions for which there was strong evidence from systematic reviews.
The methodological limitations of the original studies covered in the included reviews should be born in mind when interpreting the results of the current review. First, long-term implications of SDP were often tested retrospectively by asking mothers whether or not they had smoked during pregnancy. This clearly has limitations as these studies were not designed to compare the offspring of smoking mothers with the children of non-smoking mothers to determine differences in their health, but rather to compare the exposure in children with particular conditions and those without these conditions. The second issue is the usual reliance on mother’s memory and openness about their smoking behaviour is unsatisfactory. The third issue is the impact of confounding factors. For example, a seven-year-old child with diagnosed asthma could have a mother who smoked during pregnancy only and a father who smoked during pregnancy and the postpartum period. To minimise the impact of this the most adjusted estimations were reported in this review.
The review in the context of literature
Two previous scoping reviews were conducted to define the health outcomes of SDP although they did not focus on systematic reviews [ 9 , 11 ]. The scoping review by Jones et al. [ 11 ] was more comprehensive and included 32 health conditions. A quality assessment was not conducted but specific criteria were used to assess the strength of the evidence. According to the criteria, Jones et al. suggested that the evidence for a link between obesity and SDP was not strong [ 11 ]. However, the current analysis suggests a causal link due to the inclusion of two subsequently published systematic reviews [ 32 , 43 ].
Some of the health conditions covered in this study were also included in the review by Godfrey et al. but often higher ratios were reported [ 9 ]. This might be because they included narrative reviews which did not separate maternal SDP and postnatal passive smoke exposure while estimating the summary risk ratios [ 24 , 85 , 86 , 87 ]. Moreover, none of the previous reviews analysed the impact of the number of cigarettes consumed, partners’ smoking and postpartum smoking on infants. Therefore, the current review is more comprehensive and more systematic than previous studies.
Gaps in the literature
The study identified important gaps in the literature which warrant further research. In particular, there is a need to further our understanding of dose-response association, the impact of postnatal smoking, and SHS during pregnancy. Current evidence on the impact of number of cigarettes consumed suggests that even low amounts of cigarette consumption during pregnancy have significant health outcomes and there is a clear gradient for some conditions. This indicates the importance of smoking cessation during pregnancy and if reduction in smoking which is often not addressed in smoking cessation interventions designed for pregnant women.
Only two studies assessed the impact of SHS by partners during pregnancy when the mother was a non-smoker. There was no review reporting the combined impact of SDP and SHS by partners during pregnancy while two reviews reported increased risks for SID [ 43 ] and delay in mental development [ 25 ] when only the partner smoked during pregnancy. Hence, more research is needed to understand the impacts of having a smoking partner during pregnancy.
This study has shown that smoking during pregnancy and the postpartum period has significant health consequences for mothers and infants. It is important to encourage pregnant smokers to quit smoking or reduce the number of cigarettes consumed if they are not prepared to quit entirely since the existing evidence indicates a dose-response association. Similarly, the impact of SHS needs to be considered to promote a smoke-free environment for the mother and infant.
Availability of data and materials
Not applicable.
Abbreviations
- Smoking during pregnancy
Low birth weight
Lower respiratory infections
High-income countries
Low and middle-income countries
Centre for Reviews and Dissemination
Second-hand smoking
Sudden infant death syndrome
Burke H, Leonardi-Bee J, Hashim A, Pine-Abata H, Chen Y, Cook DG, et al. Prenatal and passive smoke exposure and incidence of asthma and wheeze: systematic review and Meta-analysis. Pediatrics. 2012;129(4):735–44. https://doi.org/10.1542/peds.2011-2196 .
Castles A, Adams EK, Melvin CL, Kelsch C, Boulton ML. Effects of smoking during pregnancy. Five meta-analyses. Am J Prev Med. 1999;16(3):208–15. https://doi.org/10.1016/S0749-3797(98)00089-0 .
Article CAS PubMed Google Scholar
Pereira PP, Da Mata FA, Figueiredo AC, de Andrade KR, Pereira MG. Maternal active smoking during pregnancy and low birth weight in the Americas: a systematic review and Meta-analysis. Nicotine Tob Res. 2017;19(5):497–505. https://doi.org/10.1093/ntr/ntw228 .
Article PubMed Google Scholar
Shah NR, Bracken MB. A systematic review and meta-analysis of prospective studies on the association between maternal cigarette smoking and preterm delivery. Am J Obstet Gynecol. 2000;182(2):465–72. https://doi.org/10.1016/S0002-9378(00)70240-7 .
Article CAS PubMed PubMed Central Google Scholar
Australian Institute of Health and Welfare. Australia’s mothers and babies 2015. Canberra: AIHW; 2017.
Google Scholar
NHS. Statistics on Smoking. England: NHS; 2017.
Tong VT, Dietz PM, Farr SL, D'Angelo DV, England LJ. Estimates of smoking before and during pregnancy, and smoking cessation during pregnancy: comparing two population-based data sources. Public Health Rep (Washington, DC : 1974). 2013;128(3):179–88.
Article Google Scholar
Caleyachetty R, Tait CA, Kengne AP, Corvalan C, Uauy R, Echouffo-Tcheugui JB. Tobacco use in pregnant women: analysis of data from demographic and health surveys from 54 low-income and middle-income countries. Lancet Glob Health. 2014;2(9):e513–e20. https://doi.org/10.1016/S2214-109X(14)70283-9 .
Godfrey C, Pickett KE, Parrott S, Mdege ND, Eapen D. Estimating the costs to the NHS of smoking in pregnancy for pregnant women and infants. York: University of York; 2010.
CDC. Annual Smoking-Attributable Mortality, Years of Potential Life Lost, and Economic Costs, United States. 2002.
Jones M. The development of the economic impacts of smoking in pregnancy (ESIP) model for measuring the impacts of smoking and smoking cessation during pregnancy: University of Nottingham; 2015.
Banderali G, Martelli A, Landi M, Moretti F, Betti F, Radaelli G, et al. Short and long term health effects of parental tobacco smoking during pregnancy and lactation: a descriptive review. J Transl Med. 2015;13(1):327. https://doi.org/10.1186/s12967-015-0690-y .
Bruin JE, Gerstein HC, Holloway AC. Long-term consequences of fetal and neonatal nicotine exposure: a critical review. Toxicol Sci. 2010;116(2):364–74. https://doi.org/10.1093/toxsci/kfq103 .
Delpisheh A, Brabin L, Brabin BJ. Pregnancy, smoking and birth outcomes. Women Health. 2006;2(3):389–403. https://doi.org/10.2217/17455057.2.3.389 .
Article CAS Google Scholar
Cnattingius S. The epidemiology of smoking during pregnancy: Smoking prevalence, maternal characteristics, and pregnancy outcomes. Nicotine Tob Res. 2004;6(Suppl_2):S125–S40.
NICE. Smoking: stopping in pregnancy and after childbirth (PH26). NICE; 2010.
Baxter S, Blank L, Guillaume L, Messina J, Everson-Hock E, Burrows J. Systematic review of how to stop smoking in pregnancy and following childbirth: University of Sheffield; 2009.
Higgins J, Green S. Cochrane handbook for systematic reviews of interventions version 5.1.0. Wiley-Blackwell: England; 2011.
Saygın Avşar T, McLeod H, Jackson L. Health outcomes of maternal smoking during pregnancy and postpartum period for the mother and infant: protocol for an umbrella review. Syst Rev. 2018;7(1):235. https://doi.org/10.1186/s13643-018-0900-9 .
Article PubMed PubMed Central Google Scholar
Aromataris E, Fernandez R, Godfrey CM, Holly C, Khalil H, Tungpunkom P. Summarizing systematic reviews: methodological development, conduct and reporting of an umbrella review approach. Int J Evid-Based Health. 2015;13(3):132–40. https://doi.org/10.1097/XEB.0000000000000055 .
CRD. CRD’s guidance for undertaking reviews in health care: Centre for Reviews and Dissemination, University of York; 2009.
Hill AB. The environment and disease: association or causation? Proc R Soc Med. 1965;58(5):295–300. https://doi.org/10.1177/003591576505800503 .
Jayes L, Haslam PL, Gratziou CG, Powell P, Britton J, Vardavas C, et al. SmokeHaz: systematic reviews and Meta-analyses of the effects of smoking on respiratory health. Chest. 2016;150(1):164–79. https://doi.org/10.1016/j.chest.2016.03.060 .
Difranza JR, Lew RA. Effect of maternal cigarette-smoking on pregnancy complications and sudden-infant-death-syndrome. J Fam Pract. 1995;40(4):385–94.
CAS PubMed Google Scholar
Tsai MS, Chen MH, Lin CC, Ng S, Hsieh CJ, Liu CY, et al. Children’s environmental health based on birth cohort studies of Asia. Sci Total Environ. 2017;609:396–409. https://doi.org/10.1016/j.scitotenv.2017.07.081 .
Kantor R, Kim A, Thyssen JP, Silverberg JI. Association of atopic dermatitis with smoking: A systematic review and meta-analysis. J Am Acad Dermatol. 2016;75(6):1119–25.e1.
Latimer K, Wilson P, Kemp J, Thompson L, Sim F, Gillberg C, et al. Disruptive behaviour disorders: a systematic review of environmental antenatal and early years risk factors. Child Care Health Dev. 2012;38(5):611–28.
Waylen AL, Metwally M, Jones GL, Wilkinson AJ, Ledger WL. Effects of cigarette smoking upon clinical outcomes of assisted reproduction: a meta-analysis. Hum Reprod Update. 2009;15(1):31–44. https://doi.org/10.1093/humupd/dmn046 .
Xuan Z, Zhongpeng Y, Yanjun G, Jiaqi D, Yuchi Z, Bing S, et al. Maternal active smoking and risk of oral clefts: a meta-analysis. Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122(6):680–90. https://doi.org/10.1016/j.oooo.2016.08.007 .
Ankum WM, Mol BWJ, Van der Veen F, Bossuyt PMM. Risk factors for ectopic pregnancy: a meta-analysis. Fertil Steril. 1996;65(6):1093–9. https://doi.org/10.1016/S0015-0282(16)58320-4 .
Chao TK, Hu J, Pringsheim T. Prenatal risk factors for tourette syndrome: a systematic review. BMC Pregnancy Childbirth. 2014;14(1):53.
Huang JS, Lee TA, Lu MC. Prenatal programming of childhood overweight and obesity. Matern Child Health J. 2007;11(5):461–73.
Huncharek MS, Kupelnick B, Klassen H. Maternal smoking during pregnancy and the risk of childhood brain tumors: a meta-analysis of 6566 subjects from twelve epidemiological studies. J Neuro-Oncol. 2002;57(1):51–7. https://doi.org/10.1023/A:1015734921470 .
Ferrante G, Antona R, Malizia V, Montalbano L, Corsello G, La Grutta S. Smoke exposure as a risk factor for asthma in childhood: a review of current evidence. Allergy Asthma Proceed. 2014;35(6):454–61. https://doi.org/10.2500/aap.2014.35.3789 .
Little J, Cardy A, Munger RG. Tobacco smoking and oral clefts: a meta-analysis. Bull World Health Organ. 2004;82(3):213–8.
PubMed PubMed Central Google Scholar
Tuomisto J, Holl K, Rantakokko P, Koskela P, Hallmans G, Wadell G, et al. Maternal smoking during pregnancy and testicular cancer in the sons: a nested case-control study and a meta-analysis. Eur J Cancer. 2009;45(9):1640–8. https://doi.org/10.1016/j.ejca.2009.01.017 .
Jones LL, Hashim A, McKeever T, Cook DG, Britton J, Leonardi-Bee J. Parental and household smoking and the increased risk of bronchitis, bronchiolitis and other lower respiratory infections in infancy: systematic review and meta-analysis. Respir Res. 2011;12:1. https://doi.org/10.1186/1465-9921-12-5 .
Marufu TC, Ahankari A, Coleman T, Lewis S. Maternal smoking and the risk of still birth: systematic review and meta-analysis. BMC Public Health. 2015;15(1):239. https://doi.org/10.1186/s12889-015-1552-5 .
Nicoletti D, Appel LD, Siedersberger Neto P, Guimaraes GW, Zhang L. Maternal smoking during pregnancy and birth defects in children: a systematic review with meta-analysis. Cadernos de saude publica. 2014;30(12):2491–529. https://doi.org/10.1590/0102-311x00115813 .
Wendland EM, Pinto ME, Duncan BB, Belizán JM, Schmidt MI. Cigarette smoking and risk of gestational diabetes: a systematic review of observational studies. BMC Pregnancy Childbirth. 2008;8(1):53. https://doi.org/10.1186/1471-2393-8-53 .
England L, Zhang J. Smoking and risk of preeclampsia: a systematic review. Front Biosci. 2007;12(1):2471–83. https://doi.org/10.2741/2248 .
Lancaster CA, Gold KJ, Flynn HA, Yoo H, Marcus SM, Davis MM. Risk factors for depressive symptoms during pregnancy: a systematic review. Am J Obstet Gynecol. 2010;202(1):5–14. https://doi.org/10.1016/j.ajog.2009.09.007 .
Riedel C, Schonberger K, Yang S, Koshy G, Chen YC, Gopinath B, et al. Parental smoking and childhood obesity: higher effect estimates for maternal smoking in pregnancy compared with paternal smoking--a meta-analysis. Int J Epidemiol. 2014;43(5):1593–606. https://doi.org/10.1093/ije/dyu150 .
Hackshaw A, Charles R, Sadie B. Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update. 2011;17(5):5.
Flenady V, Koopmans L, Middleton P, Froen J, Smith GC, Gibbons K, et al. Major risk factors for stillbirth in high-income countries: a systematic review and meta-analysis. Lancet. 2011;377(9774):1331–40. https://doi.org/10.1016/S0140-6736(10)62233-7 .
Pineles BL, Hsu S, Park E, Samet JM. Systematic review and Meta-analyses of perinatal death and maternal exposure to tobacco smoke during pregnancy. Am J Epidemiol. 2016;184(2):87–97. https://doi.org/10.1093/aje/kwv301 .
Chrestani MA, Santos IS, Horta BL, Dumith SC, de Oliveira Dode MA. Associated factors for accelerated growth in childhood: a systematic review. Matern Child Health J. 2013;17(3):512–9. https://doi.org/10.1007/s10995-012-1025-8 .
Lee L, Lupo P. Maternal smoking during pregnancy and risk of congenital heart defects: a systematic review and a meta-analysis. Circ Conf. 2012;125(10 SUPPL. 1):398.
Wang M, Wang ZP, Zhang M, Zhao ZT. Maternal passive smoking during pregnancy and neural tube defects in offspring: a meta-analysis. Arch Gynecol Obstet. 2014;289(3):513–21. https://doi.org/10.1007/s00404-013-2997-3 .
Wyszynski DF, Duffy DL, Beaty TH. Maternal cigarette smoking and oral clefts: a meta-analysis. Cleft Palate Craniofac J. 1997;34(3):206–10. https://doi.org/10.1597/1545-1569_1997_034_0206_mcsaoc_2.3.co_2 .
Zhang D, Cui H, Zhang L, Huang Y, Zhu J, Li X. Is maternal smoking during pregnancy associated with an increased risk of congenital heart defects among offspring? A systematic review and meta-analysis of observational studies. J Matern-Fetal Neonatal Med. 2017;30(6):645–57. https://doi.org/10.1080/14767058.2016.1183640 .
Brion M-JA, Leary SD, Lawlor DA, Smith GD, Ness AR. Modifiable maternal exposures and offspring blood pressure: a review of epidemiological studies of maternal age, diet, and smoking. Pediatr Res. 2008;63(6):593–8. https://doi.org/10.1203/PDR.0b013e31816fdbd3 .
Clifford A, Lang L, Chen R. Effects of maternal cigarette smoking during pregnancy on cognitive parameters of children and young adults: a literature review. Neurotoxicol Teratol. 2012;34(6):560–70. https://doi.org/10.1016/j.ntt.2012.09.004 .
Ino T. Maternal smoking during pregnancy and offspring obesity: Meta-analysis. Pediatr Int. 2010;52(1):94–9. https://doi.org/10.1111/j.1442-200X.2009.02883.x .
Antonopoulos CN, Sergentanis TN, Papadopoulou C, Andrie E, Dessypris N, Panagopoulou P, et al. Maternal smoking during pregnancy and childhood lymphoma: a meta-analysis. Int J Cancer. 2011;129(11):2694–703. https://doi.org/10.1002/ijc.25929 .
Linnet KM, Dalsgaard S, Obel C, Wisborg K, Henriksen TB, Rodriquez A, et al. Maternal lifestyle factors in pregnancy risk of attention deficit hyperactivity disorder and associated behaviors: review of the current evidence. Am J Psychiatry. 2003;160(6):1028–40. https://doi.org/10.1176/appi.ajp.160.6.1028 .
Oken E, Levitan E, Gillman M. Maternal smoking during pregnancy and child overweight: systematic review and meta-analysis. Int J Obes. 2008;32:10.
Rayfield S, Plugge E. Systematic review and meta-analysis of the association between maternal smoking in pregnancy and childhood overweight and obesity. J Epidemiol Commun Health. 2017;71(2):162–73. https://doi.org/10.1136/jech-2016-207376 .
Weng SF, Redsell SA, Swift JA, Yang M, Glazebrook CP. Systematic review and meta-analyses of risk factors for childhood overweight identifiable during infancy. Arch Dis Child. 2012;97(12):1019–26. https://doi.org/10.1136/archdischild-2012-302263 .
Huang J, Zhu T, Qu Y, Mu D. Prenatal, perinatal and neonatal risk factors for intellectual disability: a systemic review and meta- Analysis. PLoS One. 2016;11(4):e0153655.
Yermachenko A, Dvornyk V. A meta-analysis provides evidence that prenatal smoking exposure decreases age at menarche. Reprod Toxicol. 2015;58:222–8. https://doi.org/10.1016/j.reprotox.2015.10.019 .
Zhang K, Wang X. Maternal smoking and increased risk of sudden infant death syndrome: a meta-analysis. Legal Med (Tokyo, Japan). 2013;15(3):115–21.
Chu P, Wang H, Han S, Jin Y, Lu J, Han W, et al. Maternal smoking during pregnancy and risk of childhood neuroblastoma: systematic review and meta-analysis. J Cancer Res Ther. 2016;12(2):999.
Rumrich IK, Viluksela M, Vahakangas K, Gissler M, Surcel HM, Hanninen O. Maternal smoking and the risk of cancer in early life - a meta-analysis. PLoS ONE [Electronic Resource]. 2016;11(11):e0165040.
Lee LJ, Lupo PJ. Maternal smoking during pregnancy and the risk of congenital heart defects in offspring: a systematic review and metaanalysis. Pediatr Cardiol. 2013;34(2):398–407. https://doi.org/10.1007/s00246-012-0470-x .
Yan K, Xu X, Liu X, Wang X, Hua S, Wang C, et al. The associations between maternal factors during pregnancy and the risk of childhood acute lymphoblastic leukemia: a meta-analysis.[erratum appears in Pediatr blood Cancer. 2016 may;63(5):953-4; PMID: 26999072]. Pediatr Blood Cancer. 2015;62(7):1162–70. https://doi.org/10.1002/pbc.25443 .
Conde-Agudelo A, Althabe F, Belizan JM, Kafury-Goeta AC. Cigarette smoking during pregnancy and risk of preeclampsia: a systematic review. Am J Obstet Gynecol. 1999;181(4):1026–35. https://doi.org/10.1016/S0002-9378(99)70341-8 .
Ananth CV, Smulian JC, Vintzileos AM. Incidence of placental abruption in relation to cigarette smoking and hypertensive disorders during pregnancy: a meta-analysis of observational studies. Obstet Gynecol. 1999;93(4):622–8. https://doi.org/10.1016/s0029-7844(98)00408-6 .
Fernandes M, Yang X, Li JY, Cheikh IL. Smoking during pregnancy and vision difficulties in children: a systematic review. Acta Ophthalmol. 2015;93(3):213–23. https://doi.org/10.1111/aos.12627 .
Toschke AM, Beyerlein A, von Kries R. Children at high risk for overweight: a classification and regression trees analysis approach. Obes Res. 2005;13(7):1270–4. https://doi.org/10.1038/oby.2005.151 .
Mitchell EA, Milerad J. Smoking and the sudden infant death syndrome. Rev Environ Health. 2006;21(2):81–103. https://doi.org/10.1515/reveh.2006.21.2.81 .
Huang Y, Huang J, Lan H, Zhao G, Huang C. A meta-analysis of parental smoking and the risk of childhood brain tumors. PLoS One. 2014;9(7):e102910.
Park SK, Kang D, McGlynn KA, Garcia-Closas M, Kim Y, Yoo KY, et al. Intrauterine environments and breast cancer risk: meta-analysis and systematic review. Breast Cancer Res. 2008;10(1):R8. https://doi.org/10.1186/bcr1850 .
Jones LL, Hassanien A, Cook DG, Britton J, Leonardi-Bee J. Parental smoking and the risk of middle ear disease in children: a systematic review and meta-analysis. Arch Pediatr Adolesc Med. 2012;166(1):18–27. https://doi.org/10.1001/archpediatrics.2011.158 .
Pineles BL, Park E, Samet JM. Systematic review and Meta-analysis of miscarriage and maternal exposure to tobacco smoke during pregnancy. Am J Epidemiol. 2014;179(7):807–23. https://doi.org/10.1093/aje/kwt334 .
Evans D. Hierarchy of evidence: a framework for ranking evidence evaluating healthcare interventions. J Clin Nurs. 2003;12(1):77–84. https://doi.org/10.1046/j.1365-2702.2003.00662.x .
Murad MH, Asi N, Alsawas M, Alahdab F. New evidence pyramid. Evid Based Med. 2016;21(4):125–7. https://doi.org/10.1136/ebmed-2016-110401 .
Montgomery SM, Ekbom A. Smoking during pregnancy and diabetes mellitus in a British longitudinal birth cohort. Br Med J. 2002;324(7328):26–7. https://doi.org/10.1136/bmj.324.7328.26 .
Mackay DF, Anderson JJ, Pell JP, Zammit S, Smith DJ. Exposure to tobacco smoke in utero or during early childhood and risk of hypomania: prospective birth cohort study. Eur Psychiatry. 2017;39:33–9. https://doi.org/10.1016/j.eurpsy.2016.06.001 .
Haberg SE, Bentdal YE, London SJ, Stigum H, Kvaerner KJ, Nystad W, et al. Pre- and postnatal parental smoking and acute otitis Media in Early Childhood. Am J Resp Crit Care. 2009;179:1.
Tran PL, Lehti V, Lampi KM, Helenius H, Suominen A, Gissler M, et al. Smoking during pregnancy and risk of autism spectrum disorder in a Finnish National Birth Cohort. Paediatr Perinat Epidemiol. 2013;27(3):266–74. https://doi.org/10.1111/ppe.12043 .
Biederman J, Martelon M, Woodworth KY, Spencer TJ, Faraone SV. Is maternal smoking during pregnancy a risk factor for cigarette smoking in offspring? A longitudinal controlled study of ADHD children grown up. J Atten Disord. 2017;21(12):975–85. https://doi.org/10.1177/1087054714557357 .
Niemela S, Raisanen A, Koskela J, Taanila A, Miettunen J, Ramsay H, et al. The effect of prenatal smoking exposure on daily smoking among teenage offspring. Addiction. 2016;112(1):134–43. https://doi.org/10.1111/add.13533 .
Roelands J, Jamison MG, Lyerly AD, James AH. Consequences of smoking during pregnancy on maternal health. J Women's Health. 2009;18(6):867–72. https://doi.org/10.1089/jwh.2008.1024 .
Andres RL, Day M-C. Perinatal complications associated with maternal tobacco use. Semin Neonatol. 2000;5(3):231–41. https://doi.org/10.1053/siny.2000.0025 .
Salihu HM, Wilson RE. Epidemiology of prenatal smoking and perinatal outcomes. Early Hum Dev. 2007;83(11):713–20. https://doi.org/10.1016/j.earlhumdev.2007.08.002 .
Walsh RA, Redman S, Brinsmead MW, Byrne JM, Melmeth A. A smoking cessation program at a public antenatal clinic. Am J Public Health. 1997;87(7):1201–4. https://doi.org/10.2105/AJPH.87.7.1201 .
Download references
Acknowledgements
No funding was received specifically for this review. It was conducted as part of the lead author’s PhD research at the University of Birmingham, which was funded by the Turkish Ministry of Education. Hugh McLeod’s time is supported by the National Institute for Health Research Applied Research Collaboration West (NIHR ARC West) at University Hospitals Bristol and Weston NHS Foundation Trust.
Author information
Authors and affiliations.
Health Economics Unit, University of Birmingham, Edgbaston, Birmingham, B15 2TT, UK
Tuba Saygın Avşar & Louise Jackson
Population Health Sciences, Bristol Medical School, University of Bristol, Bristol, BS1 2NT, UK
Hugh McLeod
National Institute for Health Research Applied Research Collaboration (NIHR ARC) West at University Hospitals Bristol and Weston NHS Foundation Trust, Bristol, UK
You can also search for this author in PubMed Google Scholar
Contributions
The search strategy was developed by the research team. TSA conducted the review, analysed the data, and drafted the manuscript. Second reviewing was done by HM and LJ. HM and LJ provided inputs in analysing the data and drafting the manuscript. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Tuba Saygın Avşar .
Ethics declarations
Ethics approval and consent to participate, consent for publication, competing interests.
The authors declare that there is no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1..
MEDLINE Search strategy and Tables S1-S10.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Avşar, T.S., McLeod, H. & Jackson, L. Health outcomes of smoking during pregnancy and the postpartum period: an umbrella review. BMC Pregnancy Childbirth 21 , 254 (2021). https://doi.org/10.1186/s12884-021-03729-1
Download citation
Received : 29 October 2020
Accepted : 17 March 2021
Published : 26 March 2021
DOI : https://doi.org/10.1186/s12884-021-03729-1
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Partner smoking
- Systematic review
- Umbrella review
- Health outcomes
BMC Pregnancy and Childbirth
ISSN: 1471-2393
- Submission enquiries: [email protected]
- General enquiries: [email protected]
- Health Tech
- Health Insurance
- Medical Devices
- Gene Therapy
- Neuroscience
- H5N1 Bird Flu
- Health Disparities
- Infectious Disease
- Mental Health
- Cardiovascular Disease
- Chronic Disease
- Alzheimer's
- Coercive Care
- The Obesity Revolution
- The War on Recovery
- Adam Feuerstein
- Matthew Herper
- Jennifer Adaeze Okwerekwu
- Ed Silverman
- CRISPR Tracker
- Breakthrough Device Tracker
- Generative AI Tracker
- Obesity Drug Tracker
- 2024 STAT Summit
- All Summits
- STATUS List
- STAT Madness
- STAT Brand Studio
Don't miss out
Subscribe to STAT+ today, for the best life sciences journalism in the industry
Our investigation of UnitedHealth, explainer on Oropouche and news on maternal health
By Theresa Gaffney Aug. 28, 2024

Want to stay on top of health news? Sign up to get our Morning Rounds newsletter in your inbox.
Good morning! Amid today’s news, two items on pregnancy that are interesting and eerie to read in tandem. And on top of that, two itchy pieces of mosquito-related news from STAT’s Helen Branswell.
advertisement
Health Care’s Colossus: UnitedHealth promised it would be hands off. It wasn’t.
Take our money, the company said. We’ll handle the business side while you look after your patients, it said. Together we can create a future where primary care leads, it said. Those were the promises UnitedHealth Group made in 2014 to ProHealth Physicians, a reputable Connecticut-based primary care network with more than 50 clinics across the state.
But doctors say the promises weren’t kept — 10 years later, the group is a shell of its former self. “People cannot get the care that they need,” said Sharon Maloney, whose husband couldn’t get into his doctor’s office for three days, precipitating a chain of events that led to his death.
In Part 3 of STAT’s investigative series on UnitedHealth Group’s physician empire, reporters recount in detail how the story unfolded at ProHealth. My colleagues spoke with more than 15 former doctors, current and former patients, and experts. They also obtained documents through public records requests.
Read the story . And if you haven’t already, go back to read Part 1 and Part 2 .
CDC announces $118.5 million to investigate maternal deaths
The Biden administration announced yesterday that CDC will invest $118.5 million over five years to help identify and prevent pregnancy-related deaths. Specifically, the funds will go toward expanding a system of Maternal Mortality Review Committees from representing 46 to 52 states and U.S. territories. The groups review deaths within each state or territory that occur within one year of the end of a pregnancy to determine if the deaths were preventable, and recommend how the deaths could be prevented in the future. Funds will also be used to implement the White House Blueprint for Addressing the Maternal Health Crisis, according to the announcement.
Maternal mortality is tricky to measure , but by all expert accounts, the U.S. is in the midst of a crisis . It’s yet to be seen how additional funding from the federal government will affect the work done by MMRCs. “Increasingly, the reporting of those findings have become a political issue, and there have been efforts to suppress their findings,” researcher Greg Roth told STAT’s Nalis Merelli last summer. Between 2017 and 2019, only 36 state groups reported their findings to the CDC.
Can you imagine having your tubes tied then getting pregnant anyway?
It happens — to about 3 to 5% of people who receive tubal sterilization, according to new estimates published yesterday in NEJM Evidence . Researchers used data from the National Survey of Family Growth collected in waves over the last two decades. Among women surveyed between 2013 and 2015, the researchers estimated that about 2.9% of those who had their tubes tied became pregnant within a year. But an estimated 8.4% had become pregnant within 10 years of the procedure.
The results indicate “nontrivial” rates of pregnancy after the purportedly permanent procedure, the authors write. It’s good to know, as data from the same survey show that about a third of women receive the surgery by the age of 44.
What is Oropouche virus, should I be worried about it, and how do I pronounce it?
Twenty-one people in the U.S. have contracted the Oropouche virus during travels to Cuba over the summer, the CDC reported yesterday. The island country is experiencing its first ever recorded outbreak of the virus, which has also been spreading in several South American countries. But what exactly is Oropouche virus?
As usual, STAT’s Helen Branswell has us covered. The virus is spread through biting insects — specifically one species of midge, which is a small fly, and one type of mosquito. About 60% of people who become infected will develop symptoms like fever, severe headache, chills, muscle aches, and joint pains. And there isn’t a vaccine or specific drug treatment for the virus, so the best way to avoid it, as Helen writes, is to not get bit.
Read more on everything you need to know about the virus including how to pronounce its name.
Rare mosquito-borne encephalitis is found in four states, with one death
In more mosquito news: New Hampshire has recorded the country’s first death this year from eastern equine encephalitis, a rare but dangerous disease spread through the bite of infected mosquitoes. The CDC says it has been informed of four human infections so far in 2024, with Wisconsin, Massachusetts, and New Jersey also reporting cases. All four involve neuroinvasive disease, meaning the virus moved into the brain. About 30% of Triple E cases are fatal, and survivors often have long-term neurological problems. Massachusetts has begun spraying mosquitoes in some communities, and is urging people to consider remaining indoors from dusk to dawn to avoid being bitten.
Triple E infections typically occur in late summer and early fall, before cool temperatures kill off the mosquitoes that spread it. The virus is most commonly found along the Gulf Coast, in the Atlantic states and around the Great Lakes. The number of cases varies by year, though most years fewer than 10 cases are reported. In 2019, however, a record 38 cases were reported.
—Helen Branswell
NIH empowers Native Americans to study substance use in their own communities
The National Institutes of Health has announced plans to devote over a quarter-billion dollars to researching substance use and drug overdose among indigenous tribes across the country. But there’s a community-driven twist: Tribes and tribal-serving organizations get to design and conduct the research themselves.
Of course, major initiatives promising to serve indigenous people can be tricky to execute, given the federal government’s track record of violence and broken promises — and broader mistrust of research projects aimed at historically marginalized communities, STAT’s Lev Facher reports.
But that context is the “foundation of this program,” said Kathy Etz, the director of Native American Programs at the National Institute on Drug Abuse. “Tribes want to lead research, they want to be directly funded to lead research, and they want the research to reflect the priorities of their communities,” she said. “What we’re doing here is supporting tribes and Native American-serving organizations to do what they want.” Read more from Lev.
What we’re reading
What mental health care protections exist in your state? Pro Publica
America is doubling down on sewer surveillance, The Atlantic
- Medicare’s new transitional coverage of emerging technologies program doesn’t go far enough, STAT
- Polio vaccines arrive in Gaza, but distributing them is the next challenge, New York Times
- Exclusive: Centene eliminates brokers’ commissions for Medicare drug plans, STAT
About the Author Reprints
Theresa gaffney.
Morning Rounds Writer and Podcast Producer
Theresa Gaffney is the lead Morning Rounds writer and a podcast producer at STAT.
infectious disease
reproductive health
STAT encourages you to share your voice. We welcome your commentary, criticism, and expertise on our subscriber-only platform, STAT+ Connect
To submit a correction request, please visit our Contact Us page .

Recommended

Recommended Stories

Glucose monitors, mpox vaccine inequity and effects of LGBTQ+ restrictions

A virus bodega, DNC takeaways, and free Covid tests

STAT Plus: UnitedHealth pledged a hands-off approach after buying a Connecticut medical group. Then it upended how doctors practice

STAT Plus: Gene therapy pioneer Jim Wilson believes he can commercialize rare-disease drugs in a tough climate

STAT Plus: AI drug firm Recursion seeks to move from survival to industry domination

IMAGES
VIDEO
COMMENTS
Background. Smoking during pregnancy (SDP) is a significant public health concern due to adverse health outcomes on mothers and infants, such as miscarriage, low birth weight (LBW), preterm birth, and asthma [ 1 - 4 ]. The prevalence of SDP is around 10% in high-income countries (HICs) [ 5 - 7] and 3% in low- and middle-income countries ...
Results. Nineteen thousand five hundred fifty-four pregnant women met the inclusion criteria and 2,714 (13.9%) of them were smokers. Even after adjusting for confounding factors, smoking during pregnancy was associated with preterm birth, birthweight < 2500 g, intrauterine growth restriction, neonatal respiratory and gastrointestinal diseases, transfer to the neonatal intensive care unit, and ...
Background It has been shown that active exposure to tobacco is associated with adverse pregnancy outcomes including, but not limited to, intrauterine fetal death, reduced fetal weight, and higher risk of preterm birth. We want to investigate these effects in a high-income country. Methods This cross-sectional study examined 20,843 pregnant women who delivered over 10 years at the Maternity ...
As shown in previous research, active and passive maternal smoking during pregnancy increases the risk of having a child with low birth weight [27,34] and significantly increases other negative pregnancy outcomes, such as preterm birth [17,21], respiratory distress , antepartum and intrapartum stillbirth , perinatal death , long-term morbidity ...
The maternal smoking status before childbirth was as follows: Never = 60.0%, Quit before recognising current pregnancy = 24.1%, Quit after finding out about current pregnancy = 12.3%, and Still ...
Research Article. Maternal cigarette smoking before and during pregnancy and the risk of preterm birth: A dose-response analysis of 25 million mother-infant pairs ... Maternal smoking during either the first or second trimester of pregnancy was associated with an increased risk of preterm birth. After adjustment for maternal age, race ...
Introduction. Maternal smoking during pregnancy is associated with a reduction in birth weight of approximately 250g and is known to adversely affect the health of both fetus and mother.[] Knowledge of the age at onset of faltering fetal growth in association with maternal smoking would be useful evidence to underpin public health advice for mothers not to smoke during pregnancy.
Associations of maternal smoking with fetal growth. Differences in mean fetal size across gestation by maternal smoking during pregnancy are presented in Fig 1, with estimates from multivariable adjusted analyses shown in S7 Table. Overall, trajectories of fetal growth varied according to maternal smoking status (p < 0.001 for each fetal ...
Maternal smoking is one of the most important preventable risk factors for infant morbidity and mortality and associated with increased risk of Sudden Infant Death Syndrome (SIDS). 1,2 SIDS is a ...
Smoking in pregnancy is known to be associated with a range of adverse pregnancy outcomes, yet there is a high prevalence of smoking among pregnant women in many countries, and it remains a major public health concern. We have conducted a systematic review and meta-analysis to provide contemporary estimates of the association between maternal smoking in pregnancy and the risk of stillbirth.
Background Maternal smoking during pregnancy may be associated with low birth weight (LBW) in offspring and global risk estimates have not been summarized previously. We aimed to systematically explore evidence regarding maternal smoking and the LBW risk in offspring globally and examine possible causes of heterogeneity across relevant studies. Methods Comprehensive search of PubMed, Ovid ...
The prevalence data are consistent with a recent analysis based on Demographic and Health Survey data from 54 countries, in which the global pooled estimate of tobacco smoking prevalence during pregnancy was 1·3% (95% CI 0·9-1·8). However, although this analysis included a higher number of reporting countries than in the study by Lange and ...
Articles were retained if they: consisted of original, quantitative research published in a peer-reviewed journal; reported the prevalence of smoking during pregnancy in the general population; and provided a measure of uncertainty (CI or SE) for the prevalence or at least two of the following: sample size, number of cases, or prevalence.
Here, we aim to investigate the effects of maternal prepregnancy smoking, reduction during pregnancy, and smoking during pregnancy on SUID rates. METHODS: We analyzed the Centers for Disease Control and Prevention Birth Cohort Linked Birth/Infant Death Data Set (2007-2011: 20 685 463 births and 19 127 SUIDs).
Research article. First published online October 2, 2018. Striving to Meet Healthy People 2020 Objectives: Trend Analysis of Maternal Smoking. ... (NHANES) to examine trends in maternal smoking and smoking cessation during pregnancy in the United States from 1985 through 2014 to (1) address the gap in data on national trends in pregnancy ...
In addition, more efforts should be made to investigate the causal nature of observed relations, given the confounding issues in research on maternal smoking during pregnancy and child outcomes. 8 Using paternal smoking as a negative control is a well-known approach to examine whether there is a direct intrauterine effect of a maternal exposure ...
Evidence regarding the negative effects of tobacco smoking on fetal development is widely documented in existing literature. The toxic effects vary from perinatal complications, such as low birth weight, to changes in adult behavior [1-5].Regarding the effects of maternal tobacco smoking on placental blood flow and vascular resistance, there still exists some controversy regarding which ...
3.2.3 Joint analyses of maternal smoking status during pregnancy and ASI with ACD. We found that for ACD, there was no interaction between MSDP and ASI (p values for interaction = 0.905). Then, according to the maternal smoking status around birth and the time starting to smoke, all participants were divided into eight groups (Groups 0 to 7).
Introduction. Maternal smoking during pregnancy (MSDP) is a major public health concern with nearly half of all women who smoke continuing to do so throughout their pregnancies (Centers for Disease Control (CDC), 2002, 2004; Ebrahim, Floyd, Merrit, Decoufle, & Holtzman, 2000).As a result, more than half a million infants per year are prenatally exposed to maternal smoking (CDC, 2004; Smith ...
The corresponding OR was 1.6, (95% CI 1.2 - 2.3) for reported exposure to ETS with or without maternal smoking in utero. Maternal smoking during pregnancy but no exposure to ETS also increased the risk of doctor's diagnosed asthma up to two years of age, ORadj = 2.1, (95% CI 1.2 - 3.7). Exposure to maternal cigarette smoking in utero is a ...
An additional meta-analysis of eight primary studies with 723,877 participants showed that children exposed to maternal prenatal tobacco smoking were 49% more likely to struggle with poor academic ...
image: Maternal prenatal smoking has a significant risk of limiting a child's academic performance. view more . Credit: "Cigarette" by Sudipto_Sarkar is licensed under CC BY-NC-ND 2.0.
Enhancing pregnancy health is known to improve the mother's and offspring's life-long well-being. The maternal environment, encompassing genetic factors, impacts of social determinants, the nutritional/metabolic milieu, and infections and inflammation, have immediate consequences for the in utero development of the fetus and long-term programming into childhood and adulthood. Moreover ...
Introduction. Smoking is a modifiable risk factor for adverse maternal and neonatal outcomes and is associated with maternal, fetal, and infant morbidity and mortality [].As shown in previous research, active and passive maternal smoking during pregnancy increases the risk of having a child with low birth weight [27, 34] and significantly increases other negative pregnancy outcomes, such as ...
28 August 2024 Smoking harms almost every part of your body. But if you smoke when pregnant, the toxic chemicals in tobacco will also harm your unborn baby, with new research showing that it could lead to reduced academic outcomes at school.. In a systematic review of 19 studies and 1.25 million participants, researchers at the University of South Australia along with a team at Curtin ...
More information: Bereket Duko et al, The effect of maternal prenatal tobacco smoking on offspring academic achievement: A systematic review and meta-analysis, Addictive Behaviors (2024). DOI: 10. ...
Smoking during pregnancy (SDP) is a significant public health concern due to adverse health outcomes on mothers and infants, such as miscarriage, low birth weight (LBW), preterm birth, and asthma [1,2,3,4].The prevalence of SDP is around 10% in high-income countries (HICs) [5,6,7] and 3% in low- and middle-income countries (LMICs) [].Smoking during pregnancy generates a considerable cost ...
Smoking even one or two cigarettes a day before or during pregnancy can lead to serious health problems for newborns, according to a new analysis of more than 12 million families.
Not so, found the new study, which tracked the health outcomes of more than 32,000 people diagnosed with heart disease over five years. Close to 15,000 had smoked at some point in their lives and ...
Read the story.And if you haven't already, go back to read Part 1 and Part 2.. CDC announces $118.5 million to investigate maternal deaths. The Biden administration announced yesterday that CDC ...